Submitted on: 2023, 6th November; revised on: 2023, 13th November; accepted on: 2025, 21th January
Growth and metamorphosis of Bufo gargarizans gargarizans larvae: effects of water volume and food diversity
College of Life Science, Xinyang Normal University, SD 464000, China
*Corresponding author. Email: yutonglei_00000@163.com
Abstract. Organisms can evolve and produce different phenotypes in respond to local environmental conditions. Abiotic factors, such as water volume and food diversity in breeding ponds, also play significant roles in the survival and age at metamorphosis of amphibians. Here, we experimentally examined the plasticity of growth rate, survival rate, larval mass, age, and body size at metamorphosis in Bufo gargarizans gargarizans in response to varying combinations of water volume and food diversity. The interaction between water volume and food diversity had no effect on larval growth and development. However, food diversity had a significant impact on the age at metamorphosis, with two food resources resulting in a shorter larval period and earlier metamorphosis, particularly in larger water volumes. Our findings demonstrated that tadpoles raised in larger water volumes had larger body sizes at metamorphosis compared to those in smaller water volumes. Additionally, tadpoles in larger water volumes exhibited higher growth rates and shorter larval periods than those in smaller water volumes. We suggest that the increased frequency of physical encounters between tadpoles and the vessel walls in smaller volumes, akin to high-density conditions, causes psychological stress due to crowding, which hinders larval growth and development.
Keywords. Bufo gargarizans gargarizans, water volume, food diversity, phenotypic plasticity, metamorphic size.
Organisms have the ability to adapt to specific environmental conditions by changing their phenotype (Pigliucci, 2001; DeWitt and Scheiner, 2004). Biotic factors such as population density (Kehr et al., 2014), competition, and predation can impact the composition of tadpole populations (e.g., tadpole size, developmental stage, distribution, richness, and diversity). Additionally, abiotic factors like hydroperiod, habitat size, or water volume can affect the survival and timing of metamorphosis in amphibians (Semlitsch et al., 1996; Wilbut, 1997).
For organisms with complex life cycles, such as amphibians, the growth and developmental rate of the larval stage can change according to different environmental conditions (Richter-Boix et al., 2011). Although excessive water volume can reduce the growth of certain species (Pearman, 1993; Kehr et al., 2014), growth rates increase with larger absolute water volume in most cases (Pearman, 1993; Montealegre-Delgado et al., 2013). Several studies have examined the potential impact of abiotic factors, such as habitat space or water volume, on the density effects (biotic factor) in organisms. As a result, the crowding effect caused by high population density often reduces the growth and development of larvae (Smith-Gill and Berven, 1979; Semlitsch and Caldwell, 1982; Relyea and Hoverman, 2003). To date, four primary factors have been suggested to explain the decrease in individual growth and the lengthening of the larval period. These factors include a decrease in food ration (Wilbur, 1977; Hota and Dash, 1981), competition for limited food resources (Brockelman, 1969; Wilbur and Collins, 1973; DeBenedictis, 1974), social interactions (Gromko et al., 1973), and stress (Lynn and Edelman, 1936; John and Fenster, 1975).
Most anuran tadpoles are herbivorous or omnivorous, but there are a few that are carnivorous and cannibalistic (Kamat, 1962; Costa and Balasubramaniam, 1965; Sabnis and Kolhatkar, 1977; Sabnis and Kuthe, 1978; Sekar, 1992; Altig et al., 2007). For instance, Bufo stomaticus larvae showed optimal growth when provided with a diet containing a combination of animal and plant proteins. However, when these larvae were only provided with goat meat, their growth rate declined, and the timing of metamorphosis was delayed (Saidapur, 2001). Similarly, Bufo melanostictus larvae showed comparable growth rates when fed a diet of spinach and Chironomous larvae. However, their growth was stunted and metamorphosis was delayed when provided with only Chironomous larvae (Sabnis and Kuthe, 1978). This suggests that competition among larvae may be reduced when multiple food sources are available in breeding sites, allowing individuals to utilize alternative resources (Martin and Garnett, 2013).
Extensive studies have been conducted on the relationships between crowding and growth rates (Goetsch, 1924; Gromko et al., 1973; Steinwascher, 1978; Fixari III et al., 2017), but little attention has been paid to the impact of confining solitary tadpoles and the potential effects of water volume and food diversity on tadpole growth and development. In this study, we investigated the potential interactive effects of food diversity and water volume on the plasticity of metamorphic traits in Bufo gargarizans gargarizans, including the length of larval period, survival, the size at metamorphosis, and growth rate.
Bufo gargarizans gargarizans exhibits sexual dimorphism and has a wide distribution in East Asia. They are explosive breeders (Wells, 2007), typically breeding between 6 to 14 days (Yu and Sharma, 2012) and tadpoles hatch after two weeks. Ponds used as breeding sites are often rich in Spirogyra and pondweed (e.g., Potamogeton crispus), which serve as a food source for tadpoles (Wei et al., 2011).
During the peak breeding period in mid-February 2018, we captured 5 mating pairs of B. g. gargarizans in Shihe County (32°08’39”N, 114°02’37”E, altitude 84 m), central plains of China. Each pair was then placed individually in a plastic container (20 L) filled with approximately 12-15 cm of pond water until females laid their eggs. Once the oviposition process was completed, we collected 50 eggs from each of the 5 egg masses. All eggs were placed separately into five plastic containers (5 L) filled with approximately 10 cm of tap water. Tap water has been previously stored in two large buckets (100 L) for several days before use. After the breeding was finished, we transported all toads and the remaining eggs back to their original spawning site.
We employed a randomized block design to test the effect of food diversity (two food resources: Potamogeton crispus and fish food with high protein content (>45%), lipids (>12%), algae (>12%), fiber (>4%), and ash (<10%); single food resource: P. crispus), and water volume (250 mL, 500 mL, 2000 mL, 8000 mL) on the growth and development of B. g. gargarizans tadpoles. Each tadpole (Gosner stage 25, absorption of external gills and formed spiracle, Gosner, 1960) was placed individually in a series of numbered opaque tanks. A total of 120 tanks were used, with the tank diameters ranging from approximately 9.49 cm to 26.99 cm, representing the four different water volumes. Water temperature was recorded using a mercury thermometer.
The experiment was conducted using 120 tanks, with each of the eight treatment combinations replicated 15 times. In each treatment combination, three tadpoles from the same families were used to diminish intra-specific competition in accordance with the principles of kin selection theory. Then, olfactory cues were provided during the larval period (before Gosner stage 43) by adding 25-800 mL of water from other families when changing water, minimizing maternal influence. Throughout the study period, tadpoles were subjected to a 13L:11D photoperiod. The room temperature was maintained at 27.72 ± 1.12 °C, and any temperature variations were consistent across all parts of the experiment. To ensure cleanliness, we used a plastic straw to remove faeces and excess food from the tanks once a day prior to feeding. In smaller containers (250 mL and 500 mL), half of the water was replaced twice a week. For the 2000 mL containers, water replacement occurred once a week, while for the 8000 mL containers, it was done biweekly.
When the first metamorph (Gosner stage 42) was observed, tanks were checked daily. All metamorphs were then carefully collected and placed individually in plastic vials with sand and 1 mm of water until the end of metamorphosis (Gosner stage 46). We measured five variables. The length of the larval period was determined by counting the number of days from the beginning of the experiment until Gosner stage 42. Mass at metamorphosis was measured using a balance (to the nearest 0.001 g), and snout-vent length (SVL) was measured using a digital caliper (to the nearest 0.01 mm). Growth rate was calculated by dividing the mass at metamorphosis by the age at metamorphosis (Gosner stage 42), and the survival rate was determined by calculating the percentage of tadpoles that successfully underwent metamorphosis.
We analyzed the length of the larval period, SVL, mass at metamorphosis, and growth rate using univariate two-way ANOVAs with type III sum of mean squares, treating water volume, food diversity, and their interaction as fixed factors. We also used a log-linear model to examine the survival rate. In cases where the univariate two-way ANOVAs yielded significant results, we performed ANOVAs or one-way ANOVAs with post-hoc comparisons (Fisher’s LSD) to assess differences between water volume or food diversity. However, given the non-significant findings from the log-linear model, no further analyses were performed to investigate differences in survival rates across water volume or food diversity. Statistical analyses were conducted using IBM SPSS Statistics 20.0 (IBM Corp, Armonk, NY, USA).
The effects of water volume and food diversity on the length of the larval period were significant (Table 1). However, their interaction was not significant (Table 1). Tadpoles raised at 8000 mL took less time to metamorphose than those raised at other volumes (Fisher’s LSD’s post hoc tests, all P < 0.001, Fig 1), while no difference was found across other water volume treatments (all P > 0.060). Tadpoles feeding on two resources reached metamorphosis earlier than those raised at single-resource treatments (P = 0.028 for all 1 of 4 LSD’s post hoc tests, Fig 1).
| Response variable | Source of variation | df | MS | F-value | P-value |
|---|---|---|---|---|---|
| Length of larval period | Water volume | 3 | 100.014 | 16.594 | <0.001 |
| Food diversity | 1 | 30.539 | 5.067 | 0.027 | |
| Water volume × Food diversity | 3 | 12.225 | 2.028 | 0.115 | |
| Error | 94 | 6.027 | |||
| SVL | Water volume | 3 | 4.92 | 7.306 | <0.001 |
| Food diversity | 1 | 0.5 | 0.742 | 0.391 | |
| Water volume × Food diversity | 3 | 0.532 | 0.79 | 0.502 | |
| Error | 94 | 0.673 | |||
| Body mass | Water volume | 3 | 0.015 | 19.447 | <0.001 |
| Food diversity | 1 | <0.001 | 0.044 | 0.835 | |
| Water volume × Food diversity | 3 | 0.0004 | 0.561 | 0.642 | |
| Error | 94 | 0.001 | |||
| Growth rate | Water volume | 3 | 20.244 | 32.709 | <0.001 |
| Food diversity | 1 | 0.391 | 0.631 | 0.429 | |
| Water volume × Food diversity | 3 | 0.46 | 0.743 | 0.529 | |
| Error | 94 | 0.619 |
The mass at metamorphosis was significantly influenced by water volume (Table 1, Fig. 1). However, there was no significant impact of food diversity or the interaction between water volume and food diversity (Table 1). Tadpoles reared in 8000 ml and 2000 ml had significantly larger mass at metamorphosis compared to those raised in 250 mL and 500 mL (all P < 0.001), but not between 8000 mL and 2000 mL (P = 0.807) or 250 mL and 500 mL (P = 0.992).
Water volume significantly affected SVL at metamorphosis (Table 1, Fig. 1), but food diversity, as well as water volume × food diversity interaction, did not (Table 1). The SVL of tadpoles reared at 8000 ml and 2000 ml was significantly larger at metamorphosis compared to those reared at 250 mL and 500 mL (all P < 0.003). However, there was no significant difference in SVL between tadpoles reared at 8000 mL and 2000 mL (P = 0.904), or between those reared at 250 mL and 500 mL (P = 0.777).
In addition to water volume (Table 1, Fig. 1), food diversity, as well as the interaction between water volume and food diversity did not significantly affect growth rate (Table 1). Tadpoles raised at 8000 mL had a greater growth rate than those raised at other treatments (all P < 0.038, Fig. 1), while tadpoles raised at 250 mL and 500 mL had a smaller growth rate than those raised at 2000 mL (both P < 0.001), but not between 250 mL and 500 mL (P = 0.809).
Water volume and food diversity did not affect survival at metamorphosis (water volume, Z = -0.97, P = 0.330; food diversity, Z = -0.57, P = 0.571), neither the interaction between water volume and food diversity (Z = 0.83, P = 0.405, Fig. 1).
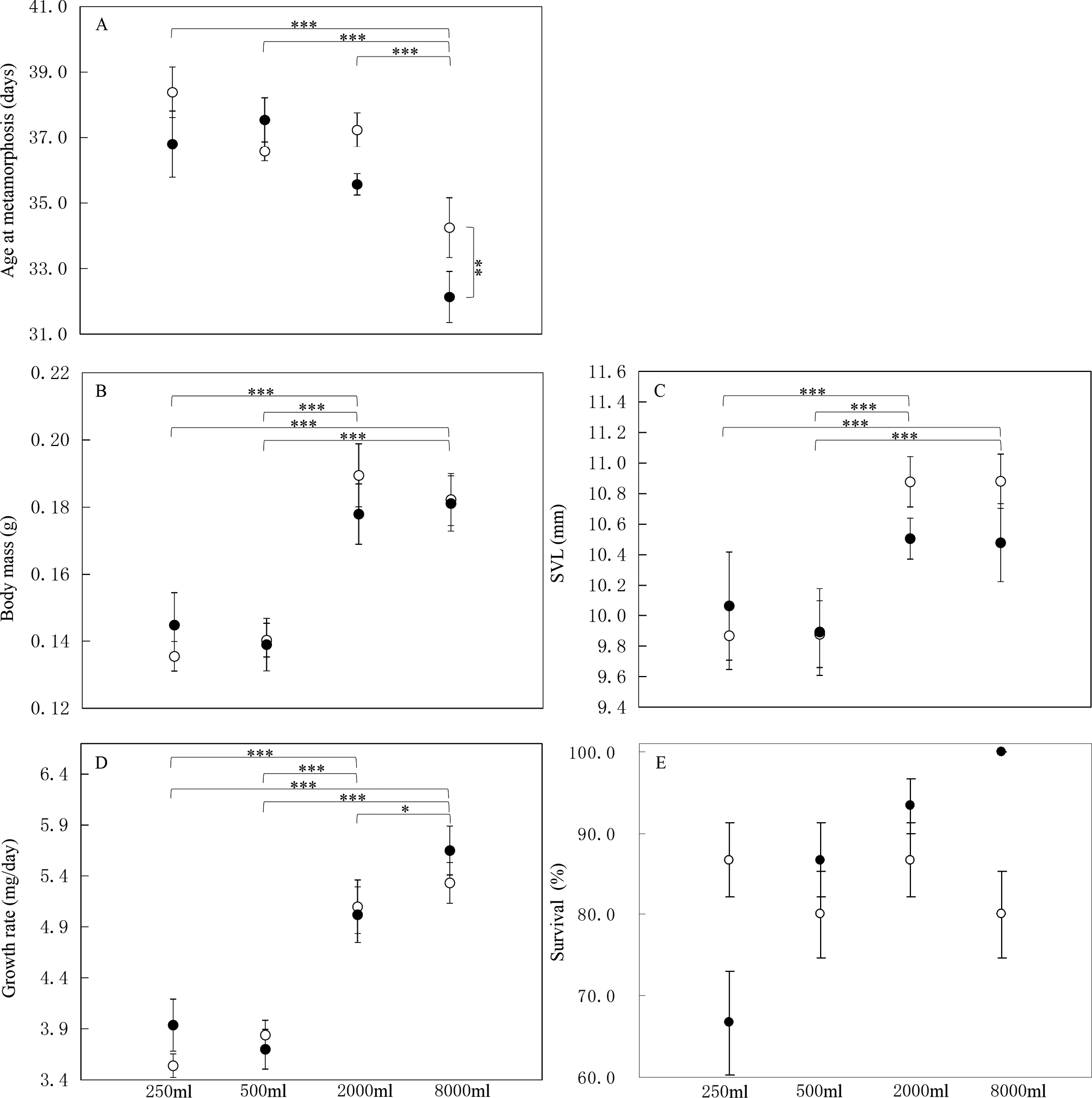
Increasing the absolute volume of water increases the growth and developmental rate of tadpoles (Gromko et al., 1973; Golay and Durrer, 1994; Smith, 1998; Durnin and Smith, 2001; Kehr et al., 2014). Our study found that single tadpoles reared in larger water volume had a larger body size at metamorphosis compared to those raised in smaller water volume. Additionally, our results demonstrated that single B. g. gargarizans tadpoles raised in larger water volume grew at a higher rate and had a shorter larval period than those raised in smaller water volume. Previous studies have also shown that smaller water volume decreased tadpole growth (Smith, 1998; Durnin and Smith, 2001), which is consistent with our results.
In this study, all tadpoles were individually placed in opaque glass beakers and received the same amount of food, suggesting that the effects of water volume could occur independently of food limitation, chemical accumulation, and social interactions. These findings support the idea that mechanisms operating in smaller water volumes can lead to crowding effects. One of the mechanisms responsible for the effects of water volume is an increase in the number of collisions among individuals confined to limited volumes, which in turn leads to a decrease in growth or development rates (John and Fenster, 1975; John and Fusaro, 1981; Rot-Nikcevic et al., 2005; Fixari III et al., 2017). Nevertheless, in our experiment there was an increased possibility of physical contact between tadpoles and vessel walls due to the smaller water volume. Additionally, we observed higher activity levels in tadpoles in smaller water volumes. This increased activity can be attributed to the increased collisions and agitation, resulting in an expenditure of energy (Rugh, 1934). Crowding diminishes the appetite of tadpoles and leads to slower growth (Adolph, 1931). Therefore, we suggest that the frequency of physical encounters between tadpoles and the vessel walls may cause psychological stress due to crowding (John and Fenster, 1975).
The surface area played a crucial role in restricting growth by regulating the availability of oxygen. Likewise, Yung (1885) discovered that tadpoles attained a larger size in tanks with a greater surface area. In this study, the surface and bottom areas of the container increase proportionally with the increase in container volume, but we speculated that the oxygen levels in smaller containers may not have been sufficient to restrict growth or had minimal inhibitory effects due to the increased frequency of water changes as the tank volume decreased. Additionally, in tanks with smaller water volumes, but with the same number of tadpoles, there was a higher accumulation of fecal material on the tank bottom, which could potentially impact growth (Podhradsky, 1932; Adolph, 1931). In this case, we employed plastic straws to clean up feces once a day throughout the experiments, thus minimizing the potential impact of feces on growth.
While the interaction between water volume and food diversity did not impact larval growth and development, we found that food diversity plays a significant role in determining the length of the larval period. Specifically, larger bodies of water with two food resources resulted in a shorter larval period and earlier metamorphosis. Within natural populations, tadpoles that undergo early metamorphosis can escape pond drying and predators (Loman, 2002). Additionally, they acquire a larger body size before hibernation, increasing their chances of survival during the longer winter period (Valenzuelasanchez et al., 2015).
In conclusion, both water volume and food diversity had an impact on larval growth and development, but these factors acted independently. The small volume of water had a more pronounced effect on limiting larval growth and development compared to food diversity. This could be attributed to the increased physical encounters between tadpoles and vessel walls, resulting in stress from crowding or psychological factors. Additionally, the crowded conditions in a small volume of water could lead to limited oxygen supply, which may be a secondary factor in reducing larval growth. Further studies are needed to explore the amount of available oxygen and gain a better understanding of its role in this process.
The field research on the plateau brown frog was funded by National Natural Science Foundation of China (Grant no. 31741019) and Natural Science Foundation of Henan Province of China (Grant no. 252300421429). This study was approved by the Animal Ethics Committee at Xinyang Normal University, Xinyang, Hunan, China, approval code: XYEC-2018-008, approval date: 1 January 2018.
Adolph, E.F. (1931): The size of the body and the size of the environment in the growth of tadpoles. Biol Bull. 61: 350-375.
Altig, R., Whiles, M.R., Taylor, C.L. (2007): What do tadpoles really eat? Assessing the trophic status of an understudied and imperiled group of consumers in freshwater habitats. Freshw. Biol. 52: 386-395.
Brockelman, W.Y. (1969): An analysis of density effects and predation in Bufo americanus tadpoles. Ecology 50: 632-644.
Costa, H.H., Balasubramaniam, S. (1965): The food of the tadpoles of Rhacophorus cruciger (Blyth). Ceylon J. Sci. 5: 105-109
Debenedictis, P.A. (1974): Interspecific competition between tadpoles of Rana pipiens and Rana sylvaticu: an experimental field study. Ecol. Monogr. 44: 129-151.
DeWitt, T.J., Scheiner, S.M. (2004): Phenotypic Plasticity: functional and conceptual, approaches. New York: Oxford University Press.
Durnin, K.B., Smith, G.R. (2001): Effects of changing water volume on the tadpoles of two anuran species (Pseudacris triseriata and Rana blairi). J. Freshw. Ecol. 16: 411-414.
Fixari, J.E. III., Mysiw, A.W., Sorenson, D.R., Smith, G.R., Rettig, J.E. (2017): Crowding effects on growth rate of Lithobates sphenocephalus tadpoles (Amphibia: Anura: Ranidae). Salamandra 53: 466-468.
Gillespie, G.R. (2002): Impacts of sediment loads, tadpole density, and food type on the growth and development of tadpoles of the spotted tree frog Litoria spenceri: an in-stream experiment. Biol. Conserv. 106: 141-150.
Goetsch, W. (1924): Lebensraum und Korpergrosse. Biol. Zentralbl. 44: 529-560.
Golay, N., Durrer, H. (1994): Volume d’eau effectif, volume d’eau accessible et effet de masse chez les têtards de Bufo calamita. Alytes 12: 64-74.
Gosner, K.L. (1960): A simplified table for staging anuran embryos and larvae with notes on identification. Herpetologica 16: 183-190.
Gromko, M.H., Mason, F. S., Smith-Gill, S. J. (1973): Analysis of the crowding effect in Runa pipiens tadpoles. J. Exp. Zool. 186: 63-72.
Hota, A.K., Dash, M.C. (1981): Growth and metamorphosis of Rana tigrina larvae: effects of food level and larval density. Oikos 37: 349-352.
John, K.R., Fusaro, J.M. (1981): Growth and metamorphosis of solitary Rana pipiens tadpoles in confined space. Copeia 1981: 737-741.
John, K.R., Fenster, D. (1975): The effects of partitions on the growth rate of crowded Rana pipiens tadpoles. Am. Midl. Nat. 93: 123-130.
Kamat, N.D. (1962): On the intestinal contents of tadpoles and algae of small ponds. Curr. Sci. 31: 300-301.
Kehr, A.I., Schaefer, E.F., Duré, M.I., Gómez, V.I. (2014): Influence of light intensity, water volume and density in tadpoles raised in mesocosm experiments. J. Zool. 293: 33-39.
Licht, L.E. (1967): Growth inhibition in crowded tadpoles: Intraspecific and interspecific effects. Ecology 48: 736-745.
Loman, J. (2002): Rana temporaria metamorph production and population dynamics in the field–effects of tadpole density, predation and pond drying. J. Nat. Conserv. 10: 95-107.
Lynn, W.G., Edelman, A. (1936): Crowding and metamorphosis in the tadpole. Ecology 17: 104-109.
Martin, R.A., Garnett, S.C. (2013): Relatedness and resource diversity interact to influence the intensity of competition. Biol. J. Linn. Soc. 110: 689-695.
Montealegre-Delgado, X.K., Avendaño-Casadiego, K., Bernal, M.H. (2013): Efecto del tamaño del habitat en la supervivencia, desarrollo y crecimiento en renacuajos de Engystomops pustulosus (Anura: Leiuperidae) y Rhinella humboldti (Anura: Bufonidae). Pap. Avulsos Zool. 53: 309-314.
Pearman, P.B. (1993): Effects of habitat size on tadpole populations. Ecology 74: 1982-1991.
Pigliucci, M. (2001): Phenotypic plasticity: beyond nature and nurture. Maryland: Johns Hopkins University Press.
Podhiradsky, J. (1932): tjber den Einfluss des Lebensriumes auf das Wachstum der Tiere. Arch. F. Entwickl. 127: 251-82.
Relyea, R.A., Hoverman, J.T. (2003): The impact of larval predators and competitors on the morphology and fitness of juvenile tree frogs. Oecologia 134: 596-604.
Richter-Boix, A., Tejedo, M., Rezende, E.L. (2011): Evolution and plasticity of anuran larval development in response to desiccation. A comparative analysis. Ecol. Evol. 1: 15-25.
Rot-Nikcevic, I., Denver, R.J., Wassersug, R.J. (2005): The influence of visual and tactile stimulation on growth and metamorphosis in anuran larvae. Funct. Ecol. 19: 1008-1016.
Rugh, R. (1934): The space factor in the growth rate of tadpoles. Ecology 15: 407-411.
Sabnis, J.H., Kuthe, S.N. (1978): Observation on food and growth of Bufo melanostictus tadpole. J. Bombay Nat. Hist. Soc. 77: 21-25
Sabnis, J.H., Kolhatkar, B.L. (1977): Observations on the food preference of Rana cyanophlyctis tadpole. Comp. Physiol. Ecol. 2: 232-233.
Saidapur, SK. (2001): Behavioral ecology of anuran tadpoles: the Indian scenario. Proc. Indian Natn. Sci. Acad. (PINSA) 67: 311-322.
Sekar, A.G. (1992): A study of the food habits of six anuran tadpoles. J. Bombay Nat. Hist. Soc. 89: 9-16.
Semlitsch, R.D., Caldwell, J.P. (1982): Effects of density on growth, metamorphosis, and survivorship in tadpoles of Scuphiopus holbrooki. Ecology 63: 905-911.
Semlitsch, R.D., Scott, D.E., Pechmann, J.H.K., Gibbons, J.W. (1996): Structure and dynamics of an amphibian community: Evidence from a 16-year study of a natural pond. In: Long-term studies of vertebrate communities, pp. 217-247. Cody, M.L., Small-Wood, J.A., Eds, San Diego: Academic Press Inc.
Smith, G.R. (1998): Abundance and water volume effects in amphibian larvae. Bull. Maryland Herpetol. Soc. 34: 113-117.
Smith-Gill, S.J., Berven, K.A. (1979): Predicting amphibian metamorphosis. Am. Nat. 113: 563-585.
Steinwascher, K. (1978): Interference and exploitation competition among tadpoles of Rana utricularia. Ecology 59: 1039-1046.
Székely, P., Cogălniceanu, D., Tudor, M. (2010): Effect of habitat drying on the development of the Eastern spadefoot toad (Pelobates syriacus) tadpoles. Amphib.-Reptil. 31: 425-434.
Valenzuelasanchez, A., Cunningham, A.A., Sotoazat, C. (2015): Geographic body size variation in ectotherms: effects of seasonality on an anuran from the southern temperate forest. Front. Zool. 12: 37-37.
Wei, G., Xia P.H., Cheng, G., Wang, S., Zhang, J.Z., Lu, J.C., Xu, N. (2011): Predatory effect of tadpoles of Bufo gargarizans gargarizans and Microhyla pulchra on Algae. Guizhou Agr. Sci. 39: 150-151.
Wells, K.D. (2007): The ecology and behavior of amphibians. University of Chicago Press, Chicago.
Wilbur, H.M. (1977): Interactions of food level and population density in Rana sylvatica. Ecology 58: 206-209.
Wilbur, H.M. (1997): Experimental ecology of food webs: complex systems in temporary ponds. Ecology 78: 2279-2303.
Wilbur, H. M., Collins, J. P. (1973): Ecological aspects of amphibian metamorphosis. Science 58: 1305-1314.
Yu, T.L., Sharma, M.D. (2012): Sex recognition and mate choice by male Bufo gargarizans in Central China. Zool. Sci. 29: 347-350.
Yung, E. (1885): Influence du nombre des individus contenus dans un meme vase, et de la forme de ce vase, sur le dleveloppment des larues de Grenouille. Comp. Rend. A cad. Sci. 101: 1018-1020.