Submitted on: 2024, 4th May; revised on: 2024, 4th June; accepted on: 2025, 6th May
New record and dietary ecology of a poorly known amphibian species, Micryletta menglienica (Yang and Su, 1980), from Vietnam
1 Faculty of Environmental Sciences, University of Science, Vietnam National University, Hanoi, 334 Nguyen Trai Road, Hanoi, Vietnam
2 Institute of Biology, Vietnam Academy of Science and Technology, 18 Hoang Quoc Viet Road, Hanoi 10072, Vietnam
3 Graduate University of Science and Technology, Vietnam Academy of Science and Technology, 18 Hoang Quoc Viet Road, Cau Giay, Hanoi 10072, Vietnam
4 Tay Bac University, Son La City, Son La Province, Vietnam
5 Central Institute for Natural Resources and Environmental Studies, Vietnam National University, 19 Le Thanh Tong, Hanoi, Vietnam
6 Department of Herpetology, American Museum of Natural History, Central Park West at 79th Street, New York, New York
*Corresponding authors. Email: ngohai2709@gmail.com; phamanh@hus.edu.vn
Abstract. Recent field surveys revealed a new population of the Menglian Paddy Frog (Micryletta menglienica) from Son La Province, northwestern Vietnam based on molecular and morphological analyses. The species has been listed as “Data Deficient” in the IUCN Red List. To date, knowledge about the natural history of this species is scarce, including data on its dietary ecology. Using the stomach-flushing method, we analyzed stomach contents of 45 individuals (21 males and 24 females) from a total of 52 captured frogs because stomachs of six males and one female (13.5%) were empty. The food spectrum of M. menglienica comprises 12 types of 691 prey items (687 invertebrate items and four unidentified items), belonging to six insect orders, larvae and Araneae. Similar to other species of the family Microhylidae, M. menglienica is also identified as an ant (Formicidae) – termite (Rhinotermitidae) eating specialist with distinctly higher percentages of occurrence frequency (F = 65 %), number of prey items (N = 80 %), and importance (Ix = 75 %) than those of remaining types. The Simpson diversity index of 0.939 shows a high abundance of food prey in the wild, and the Evenness index of 0.536 suggests biased consumption of feeding selection for M. menglienica.
Keywords. Formicidae, prey items, Rhinotermitidae, Son La Province, stomach contents.
Inhabiting both freshwater and terrestrial ecosystems, amphibians play a paramount role in maintaining the stability of food webs and energy flow between their habitats (Duellman and Trueb, 1994; Wells, 2007). In particular, amphibians transfer the energy intake by consuming many invertebrates, even small vertebrate species, to higher trophic levels by serving as an essential food source for several predatory animal groups (such as reptiles and mammals), accounting for a major part of animal biomass in ecosystems (Burton and Likens, 1975; Toledo et al., 2007; Oliveira et al., 2013). Thus, understanding the trophic niche may provide additional insights into species’ natural history and biotic interactions (Schoener, 1974; Toft, 1980; Prado et al., 2005; Wells, 2007; Duré et al., 2009). To investigate this matter in amphibians, the stomach content is often obtained to identify which prey was eaten and their relative importance in the species’ diet (Maneyro et al., 2004; Caldart et al., 2012; Le et al., 2018). In terms of conservation assessments, these analyses provide crucial data on conditions and resources required for species survival under human impacts (e.g., habitat loss, degradation, and overexploitation for trade and food consumption) that can imperil the stability in animal communities with intermittent trophic chains (Clavel et al., 2011). However, detailed information on the diet niche of many amphibian species is still lacking.
The paddy frogs of the genus Micryletta Dubois 1987 currently consist of 13 species, distributed from northeastern India and China through the Indochina region and expanding southward to west Malaysia and Sumatra (Frost, 2024). Remarkably, as many as eight species of Micryletta have been discovered in the last five years (Frost, 2024). The Menglian Paddy Frog, Micryletta menglienica (Yang and Su, 1980) was originally described based on type series collected in Menglien, Yunnan Province, southern China (Yang and Su, 1980; Liu et al., 2021) and subsequently recorded from Lang Son and Cao Bang provinces, northern Vietnam (Ohler and Grosjean, 2005). It has recently been reassigned to the genus Micryletta after having been placed in the genus Kalophrynus (Fei, 2020; Liu et al., 2021). The species is currently classified as Data Deficient in the IUCN Red List because little data on its population status and natural history are available (IUCN, 2023).
As a result of our field surveys in Son La Province, northwestern Vietnam, individuals of Micryletta were collected and their morphological characteristics closely resemble those of Micryletta menglienica as reported by Liu et al. (2021). We herein confirm the record of the species for the first time from Son La Province based on analyses of molecular and morphological data. In addition, to better understand the natural history of dietary ecology of the poorly known species, we analyzed the stomach content of 52 captured individuals to identify prey items and determined their relative importance in the species’ diet.
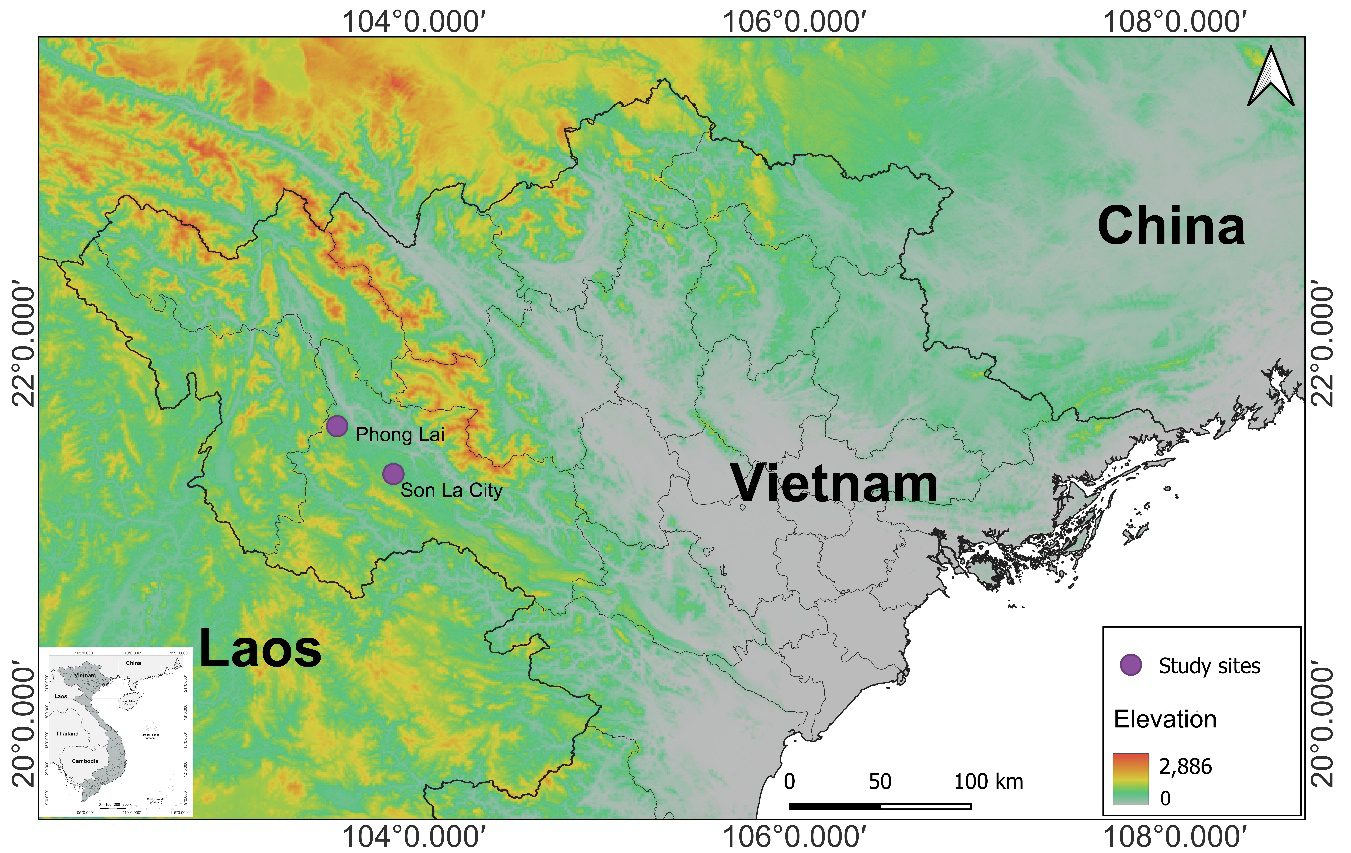
Field surveys were conducted in June, July, and September 2017 in two localities of Son La Province, northwestern Vietnam: the first site in Son La City (21°18.659’N, 103°46.956’E, at an elevation of 550 m) and the second one in Phong Lai Commune, Thuan Chau District (21o36.458’N, 103o33.980’E, at an elevation of 680 m) (Table 1, Fig. 1). The survey transects were set up with lengths ranging from 2.0 to 3.0 km, in limestone karst forests and open areas of grass and shrubs. We captured frogs by hand between 20:00 and 24:00h and used a stomach-flushing technique to obtain stomach contents without sacrificing them (Griffiths, 1986; Leclerc and Courtois, 1993; Solé et al. 2005; Norval et al., 2014). Prey items were preserved in 70% ethanol and deposited at the Faculty Environmental of Science, University of Science, Vietnam National University, Hanoi, Vietnam. Frogs were subsequently released at the collecting site after measurements of snout-vent length (SVL) and mouth width (MW) with a digital caliper to the nearest 0.01 mm taken. In total, 52 frogs, including 27 males and 25 females, were caught at two sites (Table 2).
| Locality | Time | Number of individuals | ||
|---|---|---|---|---|
| Male | Female | Total | ||
| Son La City | 10-13 June 2017 | 5 | 3 | 8 |
| 6-9 July 2017 | 3 | 4 | 7 | |
| 5-7 September 2017 | 2 | 0 | 2 | |
| Phong Lai Commune | 16-19 June 2017 | 6 | 9 | 15 |
| 15-18 July 2017 | 8 | 6 | 14 | |
| 11-13 September 2017 | 3 | 3 | 6 | |
| Total | 27 | 25 | 52 | |
| Total | Female | Male | Test (Sex) | Test (Locality) | Test (Sex + Locality) | SVL | MW | |
|---|---|---|---|---|---|---|---|---|
| Wm (mm) | 1.33 ± 0.12 (0.2-5.0) |
1.11 ± 0.09 (0.5-2.67) |
1.58 ± 0.23 (0.2-5.0) |
F = 4.4; P = 0.04 < 0.05* |
F = 3.05; P = 0.09 > 0.05 |
F = 4.3; P = 0.04 < 0.05* |
r = -0.18; P = 0.25 > 0.05 |
r = 0.02; P = 0.92 > 0.05 |
| Wt (mm) | 15.7 ± 2.56 (0.5-66.0) |
18.5 ± 4.28 (0.5-66.0) |
12.5 ± 2.4 (1.0-50.7) |
F = 1.4; P = 0.24 > 0.05 |
F = 1.8; P = 0.19 > 0.05 |
F = 0.9; P = 0.3 > 0.05 |
r = 0.14; P = 0.34 > 0.05 |
r = 0.05; P = 0.72 > 0.05 |
| Lm (mm) | 2.8 ± 0.19 (0.64-7.23) |
2..81 ± 0.25 (1.07-7.23) |
2.78 ± 0.31 (0.64-5.8) |
F = 0.008; P = 0.9 > 0.05 |
F = 0.01; P = 0.91 > 0.05 |
F = 0.4; P = 0.55 > 0.05 |
r = 0.06; P = 0.69 > 0.05 |
r = 0.24; P = 0.11 > 0.05 |
| Lt (mm) | 35.24 ± 6.6 (2.0-184.8) |
46.32 ± 11.52 (2.0-184.8) |
22.6 ± 4.01 (2.0-72.0) |
F = 3.4; P = 0.07 > 0.05 |
F = 1.6; P = 0.2 > 0.05 |
F = 0.4; P = 0.54 > 0.05 |
r = 0.23; P = 0.14 > 0.05 |
r = 0.14; P = 0.35 > 0.05 |
| Volume (mm3) | 44.02 ± 8.41 (0.26-224.6) |
40.8 ± 10.9 (0.26-188.4) |
47.7 ± 13.3 (0.3-224.8) |
F = 0.16; P = 0.69 > 0.05 |
F = 0.27; P = 0.61 > 0.05 |
F = 1.9; P = 0.18 > 0.05 |
r = -0.06; P = 0.7 > 0.05 |
r = -0.02; P = 0.91 > 0.05 |
For taxonomic identification, four individuals were collected for voucher specimens. After having been photographed in life, animals were anesthetized and euthanized in a closed vessel with a piece of cotton wool containing ethyl acetate (Simmons, 2002), fixed in 85% ethanol and subsequently stored in 70% ethanol. Determination of species was based on morphology following Liu et al. (2021). We also sequenced two new samples of Micryletta menglienica collected from Son La Province. We used the protocols of Le et al. (2006) for DNA extraction, amplification, and sequencing. A fragment of 16S rRNA gene, approximately 480bp was amplified and sequenced using a primer pair 16Sar and 16Sbr (Palumbi et al. 1991). Sequences were compared with those available from GenBank using Basic Local Alignment Search Tool (BLAST) searches.
In the laboratory, prey items were identified under microscopes (Olympus SZ 700) following taxonomic literature of invertebrates (i.e., Naumann et al., 1991; Johnson and Triplehorn, 2005). The maximum length (L) and width (W) of each prey item were measured to the nearest 0.01 mm using either a digital caliper or a calibrated ocular micrometer fitted to a microscope (Hirai and Matsui, 2001). The volume (V, mm³) of prey items was calculated using the formula for a prolate spheroid (π = 3.14; Magnusson et al., 2003): V = 4π/3 × (L/2) × (W/2)2. The index of relative importance (Ix), was used to determine the importance of each food type, which was calculated following the formula: Ix = (%F + %N + %V)/3 (Caldart et al., 2012), where %F (F – Frequency of occurrence) is a percentage of stomach containing each prey type, %N (N – Number) is a percentage of number of each prey item in all.
We used the reciprocal Simpson’s heterogeneity index, 1-D, to calculate dietary heterogeneity: D = [ni(ni – 1)]/[N(N – 1)]; where ni is the number of food items in the ith taxon category and N is the total number of prey items (Krebs, 1999). We used another index to estimate prey evenness. The evenness index is calculated from the equation: J’ = H’/Hmax = H’/lnS; where S is the total number of prey taxa and H’ is the index of taxon diversity. The value of H’ is calculated from the equation: H’ = –(pi × lnpi); where pi is the proportion of food items belonging to the ith taxon for the total food items of the sample (Magurran, 2004; Muñoz-Pedreros and Merino, 2014).
Two-way ANOVA tests were performed to determine differences or similarities in morphological characteristics of the species, sizes, and volume of ingested food between males and females, between populations, and the combined effects of “Sex + Location”. Post hoc Tukey tests (TukeyHSD) were afterwards applied to check the difference between all pairs of sex groups in each population, and location groups in each sex after the ANOVA tests. Females were identified by having longer snout-vent length (SVL ≥ 21.0 mm), whereas males were characterized by a single internal vocal sac and shorter SVL (< 21.0 mm). All statistical analyses were performed by using the software environment R.3.1.2 (RStudio Team, 2018).
A new record of Micryletta menglienica in Son La Province
Pham et al. (2016) reported Micryletta inornata (Boulenger, 1890) from Co Ma Commune, Thuan Chau District, Son La Province, Vietnam. However, the specimens were re-identified herein as M. menglienica because two sequences (GenBank accession numbers PQ634813 and PQ634814) obtained from the collected samples in Son La Province were identical to each other and 99.35% similar to those with accession numbers OK335186 and OK335187 of M. menglienica available on GenBank. This is also the first record of M. menglienica from Son La Province, northwestern Vietnam.
Morphological characteristics of paddy frogs collected in Son La Province match well with the diagnosis of Micryletta menglienica (Liu et al., 2021): size small (SVL: 14.8-27.7 mm, n = 52); head wider than long; snout obtusely round in profile; the interorbital distance wider than upper eyelid; tympanum indistinct; supratympanic fold distinct; vomerine teeth absent; tibiotarsal articulation of adpressed limb reaching the level of eye; dorsal surface granular with small round tubercles; subtle longitudinal median ridge present on dorsum; supratympanic fold present; dorsolateral fold absent; outer metatarsal tubercle absent; dorsum coloration purple brown, blueish gray or dark brown; body flanks brown with dark-brown to black patches or spots edged with white, a large black streak extending on each side from tip of the snout to crotch; ventral surface greyish brow, whitish with light-gray marbling patterns on chest and belly (Fig. 2).

Regarding sexual dimorphism, males have a shorter snout-vent length (SVL: 19.03 ± 1.95 mm, ranging 14.8-20.8 mm, n = 27; ANOVA-test, F = 124.2, P-value < 0.001) and a narrower mouth width (MW: 5.6 ± 0.73 mm, ranging 4.4-6.7 mm, n = 27; F = 52.1, P-value < 0.001) than those of females (SVL: 24.73 ± 1.4 mm, ranging 21.5-27.7 mm and MW: 6.77 ± 0.49 mm, ranging 6.0-7.8 mm, n = 25, respectively). Together with the effect of locations, the tests did not approve the general difference in both snout-vent length and head width (P-values > 0.05). In particular, the intra-sexual variations in both morphological traits of M. menglienica were not presented between two populations of Son La City and Phong Lai Commune (Tukey’s HSD tests, P-values < 0.001), whereas males’ traits were significantly different to females’, noted in each population (Tukey’s HSD tests, P-values < 0.001).
In terms of natural history, paddy frogs were found between 19:00 and 24:00 at elevations between 550 and 680 m a.s.l. The surrounding habitat was the limestone karst forest of small hardwoods, shrubs and grasses (Fig. 2). The relative humidity was approximately 70-85% and the air temperature ranged from 25 to 30oC. Other sympatric amphibian species were also observed, including Leptobrachella sp., Microhyla butleri Boulenger 1900, M. heymonsi Vogt 1911, and Polypedates megacephalus Hallowell 1861.
A total of 52 individuals (27 males and 25 females) of M. menglienica were captured in Son La City (17 individuals) and Thuan Chau District (35 individuals) of Son La Province. All caught individuals were checked for stomach contents, except for seven frogs (13.5%), whose stomachs were empty. Based on the obtained contents, we identified a total of 691 ingested food items (687 invertebrates and four unidentified items) belonging to 12 different prey types of six insect orders: Blattodea, Coleoptera (Coccinellidae, Staphylinidae, Tenebrionidae, and Other Coleoptera), Diptera (Syrphidae, Other Diptera), Hymenoptera (Formicidae), Isoptera (Rhinotermitidae), and Orthoptera (Gryllidae), Insect larvae, and Spiders (Araneae).
In terms of size measurements, the item-mean width (Wm) of ingested prey was 1.33 ± 0.12 mm (0.2-5.0 mm), the item-total width (Wt) was 15.7 ± 2.56 (0.5-66.0 mm), the item-mean length (Lm) was 2.8 ± 0.19 mm (0.64-7.23 mm), and the item-total length (Lt) was 35.24 ± 6.6 (2.0-184.8 mm) (Fig. 3, Table 2). We calculated the mean volume of 44.02 ± 8.41 mm3 (0.26-224.6 mm3). There was only a significant difference in the item-mean width (Wm) of consumed prey between males and females, and the interaction of “Sex + Locality” (Two-way ANOVA tests, P-values = 0.04 < 0.05), whereas remaining prey characteristics (i.e., Wt, Lm, Lt and volume) were relatively similar between intraspecific sexes, localities and combined effect of “sex + locality” (P-values > 0.05; Fig. 3, Table 2). Our multiple linear regression analyses showed that the sizes and volume of ingested prey are not significantly correlated with morphological traits of SVL and MW of the species (Table 2).
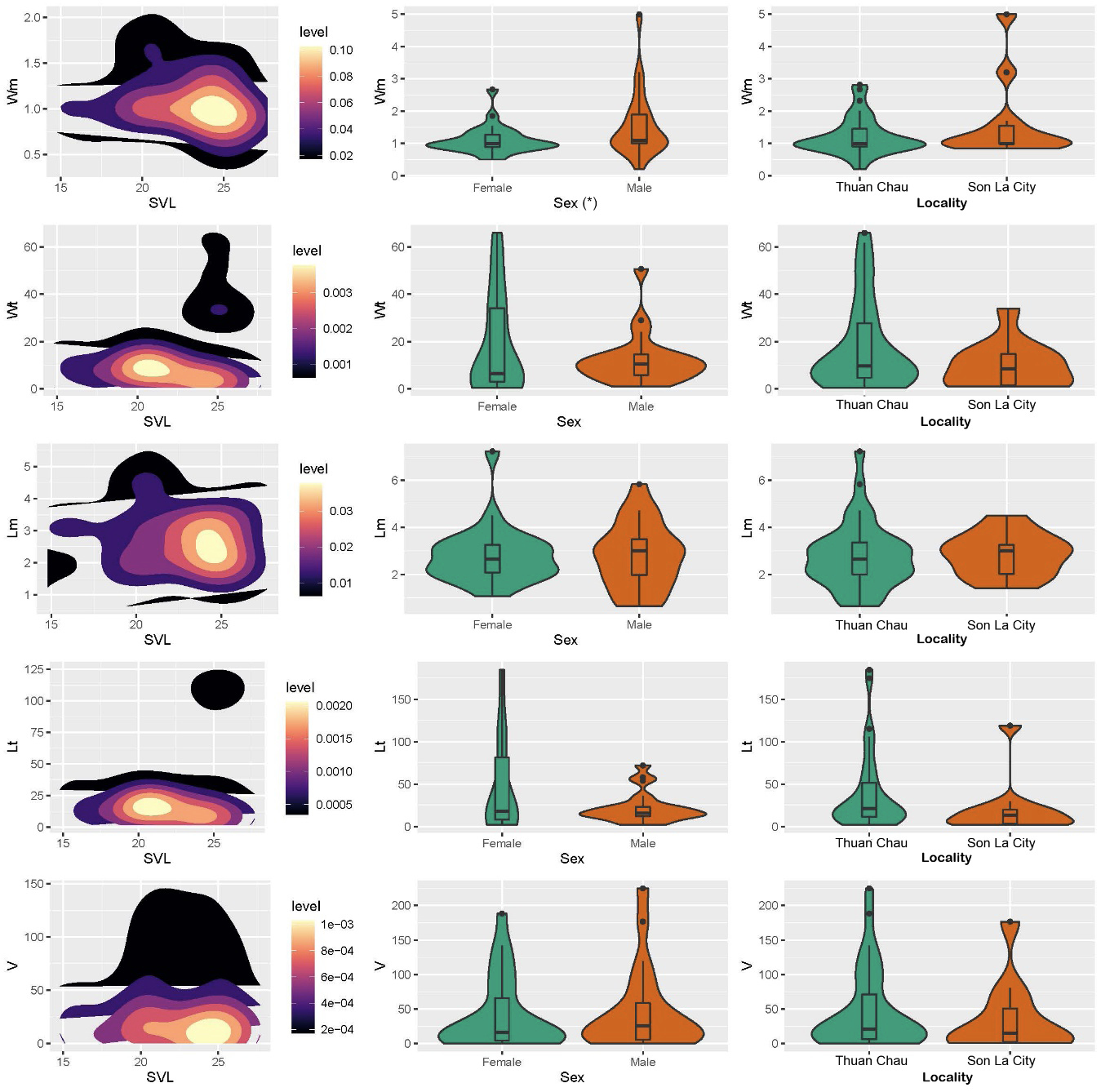
The prey of M. menglienica mainly comprised two invertebrate families of Formicidae and Rhinotermitidae, which accounted for more than 65% of the frequency of occurrence (F) and 80% of the number of prey items (N) (Table 3). The two invertebrate families were also the most important prey of the species, accounting for more than 75% of Ix (Table 3).
| Prey type | F | %F | N | %N | V | %V | Ix |
|---|---|---|---|---|---|---|---|
| Araneae | 1 | 1.41 | 1 | 0.14 | 1.57 | 0.08 | 0.54 |
| Blattodea | 1 | 1.41 | 1 | 0.14 | 20.12 | 1.02 | 0.86 |
| Coleoptera | |||||||
| Coccinellidae | 2 | 2.82 | 2 | 0.29 | 9.42 | 0.48 | 1.19 |
| Staphylinidae | 3 | 4.23 | 3 | 0.43 | 4.31 | 0.22 | 1.63 |
| Tenebrionidae | 1 | 1.41 | 1 | 0.14 | 2.74 | 0.14 | 0.56 |
| Other Coleoptera | 6 | 8.45 | 9 | 1.30 | 219.35 | 11.07 | 6.94 |
| Diptera | |||||||
| Syrphidae | 1 | 1.41 | 1 | 0.14 | 5.65 | 0.29 | 0.61 |
| Other Diptera | 1 | 1.41 | 3 | 0.43 | 36.24 | 1.83 | 1.22 |
| Hymenoptera | |||||||
| Formicidae | 36 | 50.70 | 476 | 68.89 | 632.57 | 31.94 | 50.51 |
| Isoptera | |||||||
| Rhinotermitidae | 11 | 15.49 | 91 | 13.17 | 901.84 | 45.53 | 24.73 |
| Orthoptera | |||||||
| Gryllidae | 2 | 2.82 | 2 | 0.29 | 12.04 | 0.61 | 1.24 |
| Insect larvae | 2 | 2.82 | 97 | 14.04 | 14.26 | 0.72 | 5.86 |
| Unidentified | 4 | 5.63 | 4 | 0.58 | 120.67 | 6.09 | 4.10 |
| Total | 71 | 100 | 691 | 100 | 1980.77 | 100 | 100 |
The dietary breadth of M. menglienica from Son La Province, expressed by the Simpson’s index of diversity was 0.939, and evenness was 0.536 (Table 4). Males of M. menglienica had higher values of both Simpson index (0.926) and evenness (0.783) than those of females (Table 4). The population of M. menglienica in Son La City consumed less diverse prey than the population in Thuan Chau District (Table 4).
| Simpson 1-D | Evenness | |
|---|---|---|
| Species (Micryletta menglienica) | 0.939 (0.930-0.9438) | 0.536 (0.495-0.573) |
| Male | 0.926 (0.909-0.933) | 0.783 (0.684-0.825) |
| Female | 0.8896 (0.877-0.899) | 0.50 (0.471-0.562) |
| Son La City | 0.761 (0.670-0.818) | 0.566 (0.466-0.697) |
| Phong Lai Commune | 0.9255 (0.9158-0.932) | 0.5647 (0.516-0.599) |
In this study, M. menglienica was recorded for the first time in Son La Province, which is approximately 350 km from the type locality of the species in Puer City, Yunnan Province, China. With the new record of M. menglienica in Son La Province, we recommend additional field surveys be conducted in other karst ecosystems in northern Vietnam to look for new populations of the species and better understand the species distribution range.
A total of 12 different prey types belonging to six invertebrate orders, larvae and spiders (Araneae) were identified as the food spectrum of M. menglienica, in which ants (Formicidae) and termites (Rhinotermitidae) were determined as the most dominant and important prey of the species. Anurans, especially in the family Microhylidae, have been widely recognized as ant or sometimes termite specialists (Toft, 1980; Isacch and Barg, 2002; Sole et al., 2002; Berazategui et al., 2007; Atencia et al., 2017; Lopes et al., 2017).
In this study, we did not find any intersexual difference in the sizes and volume of prey eaten by M. menglienica. Competitive interactions among conspecifics for sharing resources could take place between males and females (Schoener, 1974; Irschick et al., 2005; van Schingen et al., 2015). Our estimation based on the number of food items for indices of Simpson diversity (more than 0.88) showed a high abundance of prey at the studied sites, but not evenly among food types (0.536 – Evenness index). Furthermore, females’ prey (Evenness index = 0.5) was more biased than those in males (0.783). All of these can reduce intersexual competition. Pham et al. (2022) studied the diet niche of Microhyla butleri and M. heymonsi in Son La Province and showed that both sympatric species primarily consumed ants and termites as well. However, there were significant differences in the trophic niche between them to limit interspecific competition. In particular, 11 prey types of M. butleri were not found in the food spectrum of M. heymonsi and nine prey types were not presented in vice versa. Furthermore, they preferred to consume significantly different prey in the sizes and volume (Pham et al. 2022).
Regarding human impacts, karstic forest degradation caused by infrastructure development (e.g., road expansion, development of residential areas, and limestone quarrying) has been documented in Son La City (Pham pers. obs), which could explain the low number of M. menglienica. Only 17 individuals of M. menglienica were captured in Son La City during three surveys, compared to half of captured animals (n = 35) in less disturbed habitats in Phong Lai Commune. Such human impacts particularly destabilize its wild populations and food availability. However, similar to what is happening in Son La Province, unsustainable human activities (particularly quarrying and tourist activities) over karst ecosystems in northern Vietnam, can extensively impact undiscovered populations of the species in unique karstic habitats (Clemens et al. 2006). Therefore, conservation measures are needed to protect the species, although M. menglienica has not been targeted for the pet trade and food consumption, and its geographical distribution is potentially extended. However, to date, no conservation plan has been developed to safeguard the poorly known species as well as its natural habitats. Besides the investigation of dietary ecology, we highly recommend further studies to identify the entire geographical extent, assess population status, and determine other ecological traits of M. menglienica. Only when the baseline data becomes available, appropriate conservation measures can be designed to better safeguard the species from current extinction risks.
We thank T.V. Dau and Q.T. Bui (Son La) for their assistance in the field. Fieldwork and collection of specimens were approved by the Forest Protection Department of Son La Province (permit No. 22/GT-SL).
Atencia, P., Conde, Ó., Solano, L. (2017): Use of biological resources by Elachistocleis pearsei (Ruthven, 1914) (Anura: Microhylidae) in a lentic ecosystem from the department of Sucre, Colombia. Acta Zool. Mex. 33: 130-132.
Berazategui, M., Camargo, A., Maneyro, R. (2007): Environmental and seasonal variation in the diet of Elachistocleis bicolor (Guérin-Méneville 1838) (Anura: Microhylidae) from northern Uruguay. Zool. Sci. 24: 225-231.
Burton, T.M., Likens, G.E. (1975): Energy flow and nutrient cycling in salamander populations in the Hubbard Brook Experimental Forest, New Hampshire. Ecology. 56: 1068-1080.
Caldart, V.M., Lop, S., Bertaso, T.R.N., Zanini, C. (2012): Feeding ecology of Crossodactylus schmidti (Anura: Hylodidae) in southern Brazil. Zool. Stud. 51: 484-493.
Clavel, J., Julliard, R., Devictor, V. (2011): Worldwide decline of specialist species: toward a global functional homogenization? Front. Ecol. Environ. 9: 222-228.
Clements, R., Sodhi, N. S., Schilthuizen, M., Ng, P. K. (2006): Limestone karsts of Southeast Asia: imperiled arks of biodiversity. Bioscience 56: 733-742
Duellman, W.E., Trueb, L. (1994): Biology of Amphibians. The John Hopkins University Press, Baltimore, USA.
Duré, M.I., Kehr, A.I., Schaefer, E.F. (2009): Niche overlap and resource partitioning among five sympatric bufonids (Anura, Bufonidae) from northeastern Argentina. J. Herpetol. 8: 27-39.
Fei, L. (2020): Atlas of Amphibians in China (Field Edition). Zhengzhou, Henan science and Technology Press.
Frost, Darrel. R. (2024): Amphibian Species of the World: an online reference. American Museum of Natural History, New York, USA. Available: https://amphibiansoftheworld.amnh.org/index.php. [Accessed: 02 February 2024]
Griffiths, R.A. (1986): Feeding niche overlap and food selection in Smooth and Palmate newts, Triturus vulgaris and T. helveticus, at a pond in mid-Wales. J. Anim. Ecol. 55: 201-214.
Hirai, T., Matsui, M. (2001): Diet composition of the Indian rice frog, Rana limnocharis, in rice fields of central Japan. Curr. Herpetol. 20: 97-103.
Irschick, D.J., Vanhooydonck, B., Herrel, A., Meyers, J.J. (2005): Intraspecific correlations among morphology, performance, and habitat use within a green anole lizard (Anolis carolinensis) population. Biol. J. Linn. Soc. 85: 211-221.
Isacch, J.P., Barg, M. (2002): Are bufonid toads specialized ant feeders? A case test from the Argentinian flooding pampa. J. Nat. Hist. 36: 2005-2012.
IUCN. (2023): The IUCN Red List of Threatened Species. Available: https://www.iucnredlist.org [Accessed: 10 January 2023]
Johnson, N.F., Triplehorn, C.A. (2005): Borror and Delong’s Introduction to the Study of Insects. Belmont, California, USA, Thomson Learning Inc.
Krebs, C.J. (1999): Ecological Methodology. Menlo Park, California, Addison Wesley Longman.
Le, D.M., Raxworthy, C.J., McCord, W.P., Mertz, L. (2006): A molecular phylogeny of tortoises (Testudines: Testudinidae) based on mitochondrial and nuclear genes. Mol. Phylogenet. Evol. 40: 517-531.
Le, T.T.D., Rowley, J.L.J., Tran, T.A.D., Vo, N.T., Hoang, D.H. (2018): Diet composition and overlap in a montane frog community in Vietnam. Herpetol. Conserv. Biol. 13: 205-215.
Leclerc, J., Courtois, D. (1993): A simple stomach flushing method for ranid frogs. Herpetol. Rev. 24: 142-143.
Liu, S., Yang, B., Wang, Q., Hou, M. (2021): Taxonomic reassessment of the poorly known microhylid, Kalophrynus menglienicus Yang & Su, 1980. Herpetozoa 34: 223-232.
Lopes, M.S., Bovendorp, R.S., Moraes, G.J.D., Percequillo, A.R., Bertoluci, J. (2017): Diversity of ants and mites in the diet of the Brazilian frog Chiasmocleis leucosticta (Anura: Microhylidae). Biota Neotrop. 17: 1-5.
Magnusson, W.E., Lima, A.P., Silva, W.A., Araújo, M.C. (2003): Use of geometric forms to estimate volume of invertebrates in ecological studies of dietary overlap. Copeia 1: 13-19.
Magurran, A.E. (2004): Measuring Biological Diversity. Malden, Massachusetts, USA, Blackwell Science.
Maneyro, R.D.E., Naya, I.R., Canavero, A., Camargo, A. (2004): Diet of the South American frog Leptodactylus ocellatus (Anura, Leptodactylidae) in Uruguay. Iheringia Sér. Zool. 94: 57-61.
Muñoz-Pedreros, A., Merino, C. (2014): Diversity of aquatic bird species in a wetland complex in southern Chile. J. Nat. Hist. 48: 1453-1465.
Naumann, I.D., Came, P.B., Lawrence, J.F., Nielsen, E.S., Spradbery, J.P., Taylor, R.W., Whitten, M.J., Littlejohn, M.J., Eds. (1991): The Insects of Australia. 2 Vols, Division of Entomology, CSIRO, Australia. Melbourne University Press, Melbourne, Australia, and Cornell University Press, Ithaca, New York.
Norval, G., Huang, S.C., Mao, J.J., Goldberg, S.R., Yang, Y.J. (2014): Notes on the diets of five amphibian species from southwestern Taiwan. Alytes. 30: 69-77.
Ohler, A., Grosjean, S. (2005): Color pattern and call variation in Kalophrynus from south-east Asia. Herpetozoa 18: 99-106.
Oliveira, M.C.L.M., Santos, M.B., Loebmann, D., Hartman, A., Tozetti, A.M. (2013): Diversity and associations between coastal habitats and anurans in southernmost Brazil. An. Acad. Bras. Cienc. 85: 575-584.
Palumbi, S.R., Martin, A., Romano, S., McMillan, W.O., Stice, L., Grabowski, G. (1991): The simple fool’s guide to PCR. Department of Zoology and Kewalo Marine Laboratory, Hawaii, USA.
Pham, V.A., Ngo, D.C., Nguyen, Q.T., Ngo, V.B. (2022): Diet of Microhyla butleri and M. heymonsi from Son La Province, Northwest, Vietnam. Herpetol. Conserv. Biol. 17: 217-224.
Pham, V.A., Nguyen, Q.T., Nguyen, L.H.S., Tu, V.H., Song, B.N., Giang, A. H. (2016): Diversity of the Narrow-mouth frogs (Amphibia: Anura: Microhylidae) from Son La Province. Proceedings of the 2st National scientific conference on Biological research and teaching in Vietnam. 2: 37-42.
Prado, C.P.A., Uetanabaro, M., Haddad, C.F.B. (2005): Breeding activity patterns, reproductive modes, and habitat use by anurans (Amphibia) in a seasonal environment in the Pantanal, Brazil. Amphib.-Reptil. 26: 211-221.
RStudio Team. (2018): RStudio: Integrated Development for R. RStudio, Inc., Boston, MA, USA. Available at: https://www.rstudio.com/.
Schoener, T.W. (1974): Resource partitioning in ecological communities. Science 185: 27-39.
Simmons, J.E. (2002): Herpetological Collecting and Collections Management, 2nd ed. Lawrence, Kansas: Society for the Study of Amphibians and Reptile.
Solé, M., Beckmann, O., Pelz, B., Kwet, A., Egels, W. (2005): Stomach-flushing for diet analysis in anuran: an improved protocol evaluated in a case study in Araucaria forests, southern Brazil. Stud. Neotrop. Fauna Environ. 40: 23-28.
Solé, M., Ketterl, J., Di-Bernardo, M., Kwet, A. (2002): Ants and termites are the diet of the microhylid frog Elachistocleis ovalis (Schneider, 1799) at an Araucaria forest in Rio Grande do Sul, Brazil. Herpetol. Bull. 79: 14-17.
Toft, C.A. (1980): Feeding ecology of thirteen syntopic species of anuran in a seasonal tropical environment. Oecologia 45: 131-141.
Toledo, L.F., Silva, R.R., Haddad, C.F.B. (2007): Anurans as prey: an exploratory analysis and size relationships between predators and their prey. J. Zool. 271: 170-177.
van Schingen, M., Pham, C.T., An, H.T., Nguyen, T.Q., Bernardes, M., Bonkowski, M., Ziegler, T. (2015): First ecological assessment of the endangered crocodile lizard Shinisaurus crocodilurus Ahl, 1930 in Vietnam: Microhabitat characterization and habitat selection. Herpetol. Conserv. Biol. 10: 948-958.
Wells, K.D. (2007): The Ecology and Behavior of Amphibians. Chicago, Illinois, USA, University of Chicago Press.
Yang, D.T., Su, C.Y. (1980): A new species of the family Microhylidae frog from Yunnan. Zool. Res. 1: 257-260.