Submitted on: 2024, 14th June; revised on: 2025, 27th February; accepted on: 2025, 17th March
Can artificial rearing benefit the natural breeding of European pond turtle (Emys orbicularis) in its northern distribution range?
1 Laboratory of Fish Ecology, State Scientific Research Institute Nature Research Centre, Verkiu str. 98, LT-12201 Vilnius, Lithuania
2 Lithuanian Zoological Gardens, Radvilėnu rd. 21, LT-50299 Kaunas, Lithuania
3 Meteliai Regional Park Directorate, Kampeliu str. 10, Aleknoniu v., LT-64351, Alytus r., Lithuania
4 Laboratory of Evolutionary Ecology of Hydrobionts, Nature Research Centre, Akademijos str. 2, LT-12201 Vilnius, Lithuania
*Corresponding author. Email: vytautas.rakauskas@gamtc.lt
Abstract. The European pond turtle (Emys orbicularis) is a protected and endangered species in Europe experiencing a drastic demographic decrease over most of its range. In Lithuania, at its northern distribution range, E. orbicularis populations are small and fragmented, making them highly vulnerable to decline and deserving of active protection measures. The main reasons for the decrease in E. orbicularis populations are habitat loss due to changes in land use and the destruction of nesting sites. The survival of healthy E. orbicularis populations depends on the survival of eggs and the first overwintering of hatchlings. The aim of this study was to compare the hatching success of naturally vs artificially incubated E. orbicularis eggs and further artificially reared hatchling survival during their first year. We also provided an overview of the main nesting measurements, including clutch size and depth, from the small E. orbicularis populations in Lithuania. Finally, we furnished detailed descriptions of the artificial egg incubation and subsequent hatchling-rearing methodologies that can be used to preserve and increase the size of local small E. orbicularis populations in their northern distribution range.
Keywords. Turtle conservation, nesting ecology, Natura 2000.
The European pond turtle, Emys orbicularis (Linnaeus, 1758), is listed as Near Threatened in the International Union for Conservation of Nature (IUCN) Red List of Threatened Species and is protected in Europe by the Bern Convention and Habitats Directive 92/43/EEC (Luiselli and Vamberger, 2024). Being native to most European countries, E. orbicularis is considered extinct in the Netherlands and Belgium. In some European countries, such as Czech, Denmark and the United Kingdom, the species has been reintroduced (Luiselli and Vamberger, 2024). Lithuania and Latvia mark the northern edge of its distribution in Europe (Meeske et al., 2006). While it was widespread in Lithuania at the beginning of the 20th century, it is now found in only a few populations in the south and in a few single individuals in the north (Bastytė-Cseh, 2021). The current population of E. orbicularis in Latvian waters is even sparser and located mainly in the southern part of the country (Pupins and Pupina, 2008). Nowadays, E. orbicularis is classified as an Endangered species and included in the Red Data Book of Lithuania (Bastytė-Cseh, 2021).
The decline of the E. orbicularis population is predominantly of anthropogenic origin, due to direct destruction of their nesting sites, habitat loss from changes in land use (Schneeweis and Breu, 2013), environmental pollution (Savic, 2010), collecting for trade (Meeske and Pupins, 2009; Mollov et al., 2013), accidental killing by traffic (Isailovic and Mesaroš, 2013), and introduction of alien species and predators (Fritz and Chiari, 2013; Liuzzo et al., 2023; Purger et al., 2023). Finally, climate change is also a factor that has a clear impact on the survival and distribution of E. orbicularis (Joos et al., 2017; Cerasoli et al., 2019; Nekrasova et al., 2021) followed by genetic fragmentation (Vecchioni et al., 2020).
Previous studies have shown that predators attack E. orbicularis at all stages of life: eggs, hatchlings, juvenile turtles and adults (Fritz and Chiari, 2013; Ayaz et al., 2017; Nekrasova et al., 2021; Purger et al., 2023). However, the species is most vulnerable in the early stages of life, such as incubation of eggs and overwintering of hatchlings. Reducing mortality rates in these early stages is crucial to increase the population size of E. orbicularis (Mitrus and Zemanek, 1998). There are several strategies to achieve this. One effective method is to cover turtle nests with wire mesh, which protects eggs and hatchlings from predators in their natural environment (Schindler et al., 2017; Kiss et al., 2021). Additionally, newly hatched juveniles can be collected and raised artificially during their first year. Alternatively, freshly laid eggs can be collected and artificially incubated, followed by rearing the hatchlings for their first year. After a wintering period, carefully screened juvenile turtles, evaluated by veterinarians, can be released back into their natural habitats. This process can contribute to increasing the size of vulnerable E. orbicularis populations.
This study aimed to compare the hatching success of artificially vs naturally incubated E. orbicularis eggs and hatchling survival during their first year. We expected that hatching success would be higher under artificial incubation compared to natural conditions. The study also provides valuable insights into the characteristics of E. orbicularis clutches from Lithuanian populations, including nest depth, herbaceous coverage, mean clutch size, and average egg weight. Furthermore, it presents a comprehensive methodology for the artificial incubation and subsequent rearing of hatchlings.
E. orbicularis eggs and hatchlings were collected from the wild in Southern Lithuania, mostly in Natura 2000 sites: Juodabalė Zoological Reserve (LTLAZ0010), Kučiuliskė village surroundings (LTLAZ0001), Drapalai village surroundings (LTDRU0004), Margiai village surroundings (LTLAZ0035), Petroškiai forest (LTLAZ0020), Paveisėjai village, and Stankūnai village. The study was performed in the 2015−2022 year’s period.
Collection and transportation of eggs
Eggs for the artificial incubation experiment were excavated in early summer (in June) within the first 24 hours after they were laid to minimise embryo lethality at this stage. E. orbicularis clutch measurements, such as the mean clutch size and average egg weight, were recorded for each excavated clutch. After excavation, the eggs were placed in plastic boxes filled with a 100 mm thick layer of moist sand. Two thirds of each egg were immersed in the sand to prevent moisture loss, and the layer of sand protected the eggs from possible smashes during transportation. The eggs were not repositioned in the chamber because the embryos begin to develop at the top, very close to the shell, and repositioning them would cause the embryo to die.
Collection and transportation of hatchlings
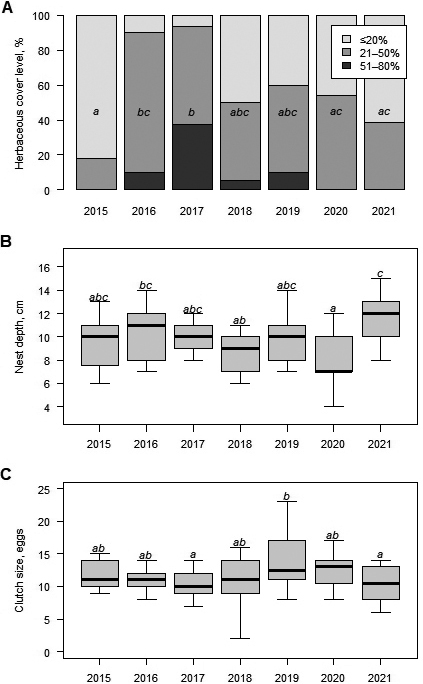
In the spring, we selected nests for the experiments and covered them with wire netting (10 × 25 mm mesh size) as soon as the female laid eggs and walked away from their nests. We attached the wire nets tightly to the ground with 5 mm diameter metal hooks (at least 25 cm long) so that predators could not tear or dig them out from the sides. Hatchlings for the experiments were dug out at the beginning of autumn, when the air temperatures start to decrease but still are > 10 °C. Small metal spades were used for digging, and as the clutches were reached, digging was finished by hand to avoid injuring the hatchlings. All hatchlings were active when taken from the nests and were placed in boxes with moss (Sphagnum sp.) and taken to the Lithuanian Zoological Gardens. During the excavation, the following measurements of E. orbicularis nests were taken: nest depth, herbaceous coverage, mean clutch size, and average egg weight for non-hatched eggs. A wooden frame (50 × 50 cm) was used to assess the herbaceous coverage. It was positioned above the selected nest, and the percentage of herbaceous coverage was visually evaluated.
Morphometric measurements and sex determination
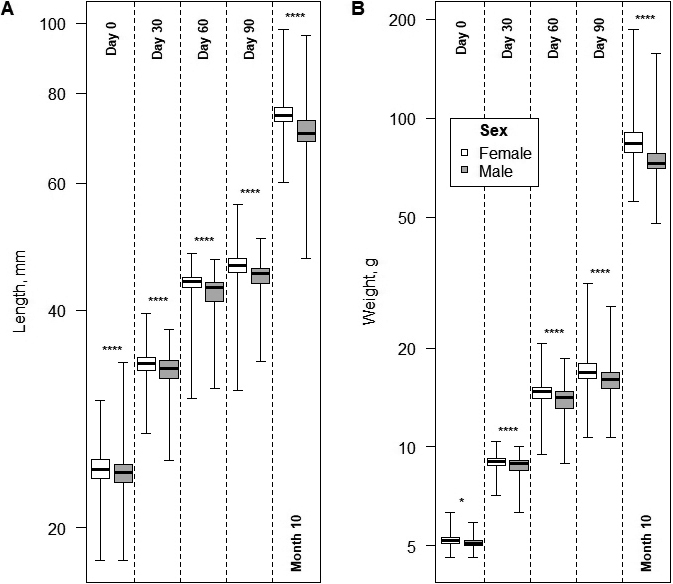
We measured E. orbicularis juveniles’ plastron length (PL) and weight five times for each individual: one day after hatching, at 30, 60 and 90 days of age, and ten months of age, before releasing them back into the wild. We used scales (Romansas, model KB, Lithuania) to measure the weight, with an accuracy of 0.01 g and an error of ± 0.1 g. We used a digital caliper (Carbon Fiber Composites, model CTCF1506, China) with a resolution of 0.1 mm and an accuracy of ± 0.1 mm to measure length. Each juvenile turtle has unique plastron patterns (Salom-Oliver et al., 2022), so photos of each individual pattern were taken to help identify individuals before taking repeated measurements.
The gender of the reared turtle was determined by the concavity of the lower part of the plastron: females have a flat (smooth) section of the lower plastron, whereas males have a slightly concave posterior part of the plastron (Fig. 1). While other methods exist for determining the sex of turtles, such as the colour of the iris or the length of the nails (Avanzi and Millfanti, 2003; Berthomieu and Vermeer, 2021), these methods are unreliable due to the young age of the turtles.

We successfully reared juvenile turtles and released them in the same Natura 2000 sites where we collected them. At the start of the summer, when the sun was shining, the air temperature was >20 °C and the water temperature was >15 °C, we carefully screened E. orbicularis juveniles by veterinarians and handed them over to the responsible specialists from the protected area for release into the wild. Two to three days before release, the turtles were kept a few degrees cooler and not fed. It was difficult to predict the outside air temperature, and excess food in the gastrointestinal tract of the turtles could have spoiled and killed them when the air cooled down suddenly. The turtles from each egg clutch were released into a natural water course that met all the requirements of a suitable E. orbicularis habitat. These water courses are under reserve protection in the same territories of Meteliai and Veisiejai Regional Parks where the turtles were collected.
The entire protocol including all the details about the eggs incubation, rearing conditions of hatchlings, feeding and overwintering procedures, are presented in Supplementary Materials.
All contingency tables (annual variation in nesting herbaceous cover level; incubation treatment-wise hatching success and survival, as well as overall and treatment-wise sex ratio) were tested using Chi-squared tests. The annual variation in nesting depth and clutch size were analysed using one-way ANOVAs.
Generalized linear models (GLMs) with binomial (or beta binomial in case of significant overdispersion) error distribution were fitted to test the annual variation in hatching success and sex ratio (as female percentage) using clutch-wise data. Mixed general linear models (GLMMs) with a binomial (or beta binomial under overdispersion) error distribution and a random effect of the year were fitted to explore the hatching success (per fertilised clutch) and sex ratio (as female percentage per viable clutch) as functions of the additive effects of nesting depth and herbaceous cover level (the interaction term was dropped according to an insignificant partial test). Similar GLMMs were also fitted to test the differences in the same two endpoints among incubation treatments.
The weight of eggs was compared between females and males (as identified later) by fitting a linear mixed model (LMM) with a random effect of clutch (initially nested in year factor, which was later removed, see Results). The growth of naturally and artificially incubated turtles was analysed by log-transformed body length and weight. The two growth LMMs were built in a forward-stepwise extension procedure. The null model only included a random effect of an individual turtle nested within the random effect of the clutch. The pool of potential fixed effects included the measurement date, sex, and incubation treatment factors with all possible interactions. Within each step, the most informative fixed term, associated with the largest decrease in model Akaike Information Criterion (AIC) value, would be added, respecting the hierarchy of interactions. The minimum threshold of AIC decrease to significantly improve the model was considered to be 2.
The analyses were performed using R v. 4.3.1 software. Mixed models were fitted using the package glmmTMB v.1.1.10. Appropriate post hoc analyses were aided by the packages rcompanion v. 2.4.36, emmeans v. 1.8.8, and multcomp v. 1.4-25. Compliance with the assumptions of all linear models was inspected using the functions from package performance v. 0.12.4. The significance level of P < 0.05 was specified for all statistical analyses a priori.
We collected 126 clutches of Emys orbicularis between 2015 and 2021 (Table 1). In the autumn, 96 clutches were excavated and naturally incubated, while 30 clutches were excavated at the beginning of summer for artificial egg incubation. The herbaceous cover of the studied nests was generally low, with 47.9% shaded by 21–50%, 42.7% by less than 20%, and 9.4% by 51–80% (none under more than 80% cover). However, the shading of studied nests varied significantly among years (Chi-squared test: χ212 = 35.7, P = 0.0004; Fig. 2A). The mean nest depth was 9.7 ± 2.3 (range 4–15) cm and varied annually (one-way ANOVA: F6,89 = 5.7, P < 0.0001; Fig. 2B). On average, there were 11.4 ± 3.0 (range 2–23) eggs per clutch, but this number also varied significantly among years (one-way ANOVA: F6,119 = 2.8, P = 0.015; Fig. 2C). According to the available data, excavated eggs weighed 9.2 ± 0.5 (range 7.1–10.9).
| Collection year | Egg incubation type (natural, artificial) | N° excavated clutches | N° collected eggs/ hatchlings | Alive hatchlings | Released number |
|---|---|---|---|---|---|
| 2015 | natural | 11 | 133 | 103 | 101 |
| artificial | 2 | 23 | 18 | 18 | |
| 2016 | natural | 10 | 109 | 57 | 58 |
| artificial | 4 | 43 | 28 | 28 | |
| 2017 | natural | 16 | 170 | 20 | 17 |
| artificial | 5 | 50 | 34 | 36 | |
| 2018 | natural | 18 | 191 | 124 | 152 |
| artificial | 15 | 174 | 145 | 145 | |
| 2019 | natural | 10 | 142 | 92 | 90 |
| artificial | 2 | 23 | 18 | 18 | |
| 2020 | natural | 13 | 162 | 128 | 114 |
| artificial | 2 | 25 | 10 | 10 | |
| 2021 | natural | 18 | 185 | 159 | 157 |
| Total | 126 | 1430 | 970 | 943 |
Hatching success and sex ratio
In the autumn, a total of 1092 eggs/hatchlings from 96 clutches were excavated. Of these, 712 (65.2%) were viable juveniles, 239 (21.9%) were non-viable hatchings, 133 (12.2%) were non-fertilised, and 8 (0.7%) eggs were physically damaged (most likely by the activity of predators). When considering only the 845 fertilised cases, 74.9% were viable. The hatching success (per fertilised clutch) varied among years (beta binomial GLM: P < 0.0001; Fig. 3A). After considering the random year effect, it was negatively related to both nest depth and herbaceous cover level (beta binomial GLMM: P < 0.046).
Viable juveniles were brought to the laboratory for artificial rearing. Of these hatchlings, 394 (55.3%) were identified as females, while 318 (44.7%) were recognised as males, giving an overall sex ratio of 1.2:1, which was significantly female-skewed (Chi-squared test: χ21 = 8.1, p = 0.004). There was no significant variation in female percentage (per viable clutch) among years (binomial GLM: P = 0.30; Fig. 3B). Nest depth and herbaceous cover had no effect on female percentage (binomial GLMM: P ≥ 0.37).
In summer, 338 eggs from 30 clutches were excavated for artificial incubation. Of these, 253 (74.8%) successfully hatched into viable juveniles, with only 8 (2.4%) being non-viable, 72 (21.3%) being non-fertilised, and 5 (1.5%) being physically damaged. This means that out of 261 fertilised eggs, 96.9% successfully hatched. The hatching success (per fertilised clutch) under artificial incubation was consistent across years (binomial GLM: P = 0.38; Fig. 3C). Of those successfully artificially hatched, 177 (70.0%) were females, while 76 (30.0%) were identified as males, showing a 2.3/1 sex ratio, which was even more female-skewed (Chi-squared test: χ21 = 40.3, P < 0.0001). There was no significant annual variation in female percentage per viable clutch (binomial GLM: P = 0.32; Fig. 3D).
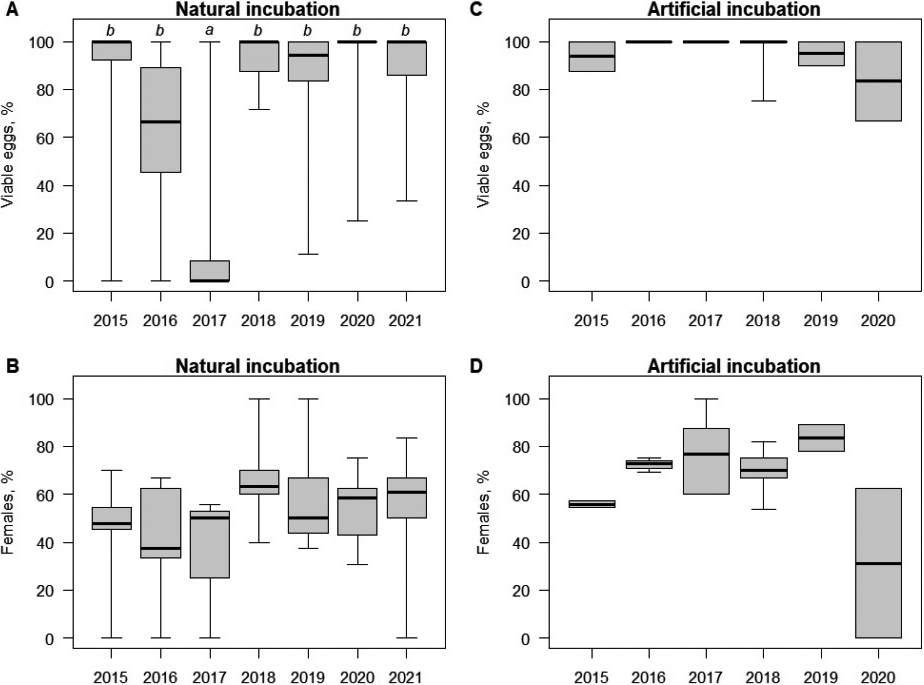
Artificially incubated eggs generally hatch more successfully (Chi-squared test: χ21 = 60.1, p < 0.001) and exhibit a significant female-skewed sex ratio (Chi-squared test: χ21 = 15.9, p < 0.001). After accounting for the random year effect, both differences remained significant (beta binomial GLMM for hatching success per fertilised clutch and binomial GLMM for female percentage per viable clutch: P ≤ 0.0018).
A total of 957 hatchlings were taken for further artificial rearing: 704 were of natural origin, while 253 were of artificial incubation. Twelve deaths occurred: 10 of natural origin (one upon hatching and nine before the 300-day measurement) and 2 of artificial incubation (both before the 300-day measurement). The mortality during rearing was low (1.2%) and did not differ between the two treatments (Chi-squared test: χ21 = 0.54, P = 0.46). Further growth analyses were conducted using only data from surviving turtles. It is noteworthy that even at the egg stage, “female” eggs were significantly heavier than “male” eggs (LMM: F1, 224 = 5.6, P = 0.019), after accounting for the significant random effect of clutch (P < 0.001). Both random effects of turtle and clutch were significant in terms of turtle length and weight (p < 0.001) (the nesting random year factor was insignificant (p = 0.8) and thus removed).
In the null LMMs of turtle growth, both random effects of turtle and clutch were significant in terms of turtle length and weight (P < 0.00001). The forward stepwise extension of models indicated that changes in length and weight significantly varied only between sexes (LMM, Data: Sex effect: F4,3772 ≥ 33,6, P ≤ 0.00001) but not by incubation. Females were generally larger, and these increasing differences were consistent across all measurement dates (Fig. 4 and Table 2).
| Date | Length (mm) | Weight (g) | ||
|---|---|---|---|---|
| Females | Males | Females | Males | |
| Natural incubation | ||||
| Day 0 | 24.1 ± 0.9 (23.5–24.6) | 23.8 ± 1.1 (23.2–24.4) | 5.2 ± 0.2 (5.1–5.3) | 5.1 ± 0.2 (5.0–5.2) |
| Day 30 | 33.7 ± 1.1 (33.1–34.2) | 33.0 ± 1.5 (32.4–34.0) | 8.9 ± 0.5 (8.6–9.2) | 8.7 ± 0.7 (8.2–9.1) |
| Day 60 | 43.3 ± 2.0 (43.0–44.2) | 42.1 ± 2.7 (41.1–43.8) | 14.9 ± 1.2 (14.2–15.5) | 14.0 ± 1.5 (13.2–14.8) |
| Day 90 | 46.0 ± 2.1 (45.1–47.0) | 44.5 ± 2.1 (43.9–45.8) | 17.9 ± 2.7 (16.4–18.1) | 16.4 ± 2.3 (15.2–16.8) |
| Month 10 | 75.8 ± 4.9 (73.2–76.8) | 71.0 ± 5.0 (68.5–73.3) | 88.2 ± 17.5 (77.9–91.5) | 75.0 ± 11.1 (70.2–76.5) |
| Artificial incubation | ||||
| Day 0 | 24.3 ± 1.9 (23.0–25.3) | 23.8 ± 1.8 (22.8–25.0) | 5.2 ± 0.3 (5.1–5.4) | 5.1 ± 0.2 (5.0–5.3) |
| Day 30 | 33.5 ± 2.1 (32.5–35.0) | 33.0 ± 2.1 (31.6–34.3) | 9.1 ± 0.5 (9.0–9.3) | 9.0 ± 0.5 (8.9–9.3) |
| Day 60 | 43.8 ± 1.5 (43.0–44.8) | 42.7 ± 2.1 (42.2–44.2) | 14.2 ± 1.1 (13.6–14.8) | 13.5 ± 1.1 (12.8–14.2) |
| Day 90 | 46.9 ± 1.7 (45.7–47.9) | 44.8 ± 2.3 (43.5–46.0) | 16.8 ± 1.2 (16.0–17.6) | 15.4 ± 1.3 (14.5–16.4) |
| Month 10 | 74.2 ± 2.9 (72.5–75.8) | 70.6 ± 4.0 (68.5–72.8) | 85.1 ± 7.7 (80.2–88.7) | 75.7 ± 6.0 (70.3–80.1) |
Many scientific studies are dedicated to Emys orbicularis nesting ecology (Drobenkov, 2000; Ayaz et al., 2017; Mitrus et al., 2018), however, there is a lack of such information from known Lithuanian populations (Meeske, 1997). This study revealed that most (91%) of E. orbicularis females lay eggs in rather open areas with little (up to 50%) herbaceous vegetation in Lithuania. Similar results were obtained in other countries, including Poland (Meeske, 1997), Slovenia (Novotny et al., 2004), Italy (Zuffi and Rovina, 2006) and Spain (Diaz-Paniagua et al., 2014). The shores of small water bodies are usually covered by dense vegetation, so E. orbicularis females sometimes have to migrate hundreds of metres or even kilometres to find such an area. As suitable nesting places are rare, females may lay eggs in unsuitable areas more distant from water bodies, such as forest and gravel roads, cultivated fields or any other open soil (Mitrus and Zemanek, 2004). Eggs left in such nests do not survive until spring, especially if there is a lack of snow in winter and thus low nest temperatures (Najbar and Szuszkiewicz, 2005). Furthermore, females from northern populations usually lay a single clutch of eggs only once a year (Mitrus and Zemanek, 2004), so if these clutches are placed in areas where individuals have little chance of survival, small E. orbicularis populations can decrease significantly in a relatively short period. Furthermore, juveniles are exposed to higher risks of predation due to the lack of a suitable nesting habitat near water bodies (Tetzlaff et al., 2020). Turtles use human-altered environments for egg-laying (Joyal et al., 2001; Purger et al., 2023), making them vulnerable to generalist predators. The presence of potential predators, such as mammals or bird species, is generally prevalent within natural and semi-natural environments (Chelazzi et al., 2000; Rössler, 2000; Zuffi, 2000). It was hypothesised that these predators may impact approximately 75-95% of undisturbed nesting sites (Rovero and Chelazzi, 1996; Zuffi and Odetti, 1998; Rössler, 2000). Therefore, it was emphasised (Mitrus and Zemanek, 1998), that to increase small E. orbicularis population size, the mortality of the individuals must be reduced in the first year of their life.
In this study, we took two active measures to reduce mortality of the most vulnerable E. orbicularis life stages. Firstly, E. orbicularis nests were covered with wire mesh to protect eggs and hatchlings from predators in the natural environment. Secondly, eggs were artificial incubated, and hatchlings reared for their first year before being released back into the wild.
Covering nests with wire mesh is one of the simplest tools for protecting eggs and hatchlings from predators and it has been effectively used in many European countries (Schindler et al., 2017; Kiss et al., 2021). In this study, nest protection resulted in only two clutches affected by predators, and eight eggs physically damaged from the overall 1092 eggs laid within protected clutches. In a similar study in Hungary, Kiss et al. (2021) observed that egg-hatching success in wire mesh-protected E. orbicularis nests varied from 67.7 to 84.3% under natural conditions. Previous studies have shown that the abundance of E. orbicularis juveniles in areas with wire mesh protection increased by 50% (Schmidt, 2017).
We excavated 30 clutches of E. orbicularis immediately after the eggs were laid for artificial incubation in the laboratory. Previous attempts to apply the clutch relocation method to protect eggs have not been successful (Marchand and Litvaitis, 2004; Mitrus, 2008; Bona et al., 2012). It was concluded that this method is too risky because the initial stages of egg development are sensitive to environmental changes and relocation can have a negative impact. However, this study proved that this protective measure is highly effective, as only 2.4% of the collected and artificially incubated eggs failed to hatch.
It is vital to understand the natural clutch parameters of small E. orbicularis populations at the northern distribution edge when applying active protection measures. We observed 126 natural clutches of E. orbicularis with a mean of 11 eggs per clutch and some significant difference among years. The number of eggs in a single clutch is similar to the average number of eggs per clutch reported from neighbouring countries: 11-15 eggs in Poland (Jablonski and Jablonska, 1998; Najbar and Szuszkiewicz, 2005), 9–13 eggs in Latvia (Pupins et al., 2019). Our observations confirmed, the average egg number of E. orbicularis clutch tends to differ across different latitudes (Zuffi et al., 2017). In southern regions, clutches of E. orbicularis are generally smaller than in northern populations. On average, the clutch size in Hungary is 9 eggs (Kiss et al., 2021), 7 eggs in Turkey (Ayaz et al., 2017), and 6 eggs in Italy (Liuzzo et al., 2024). This is explained by the fact that females in the Southern European populations lay two clutches per year, while those from Central and East European populations usually lay only one clutch per year (Fritz, 2003). Also, our study found that the average weight of E. orbicularis eggs is 9.2 g. Similarly, 8.1 g was reported as the average egg weight from the neighbouring country of Belarus (Drobenkov, 2000), whereas, in Ukraine, the average weight of deposited E. orbicularis eggs was lower at 7.4 g (Zinenko, 2004).
The mean depth of our studied nests was 9.7 cm (ranging from 4 to 15 cm), which is consistent with the nest depths reported by other authors: 10.1 cm in Italy (Liuzzo et al., 2024) and 8 cm in Turkey (Ayaz et al., 2017), whereas in Spain Diaz-Paniagua and colleagues (2014) found nest depth ranging from 4 to 6.8 cm. There was a significant year effect on the studied nest depth in this study, probably related to the different temperature profiles each year. The depth of the nests is crucial for the development of embryos and the survival of hatchlings in the northern E. orbicularis populations. Our research clearly shows that the nesting depth and herbaceous cover negatively impact hatching success. If eggs are placed in a nest that is too deep or overgrown by plants, there is a lower chance of them hatching. Schneeweiß (2004) explains that survival of northern populations depends on suitable summer temperatures for incubation and the duration of solar radiation at the nest site. Higher or lower nest temperatures can cause higher mortality of embryos. E. orbicularis embryos develop most successfully at temperatures ranging from 18 to 33 °C (Pieau and Dorizzi, 1981). However, if the clutch is placed in a nest that is too shallow, there is a higher risk of the hatchlings freezing to death during their first winter in the northern E. orbicularis populations, where the newly born hatchlings overwinter in their nests.
Our study revealed that the mean body mass of newly hatched turtles was 5.2 g, similar to hatchling weights reported from neighbouring countries: 5.2 g found in the Latvian population (Pupins et al., 2019), 6.1 g in the Belarusian population (Drobenkov, 2000), and 6.9 g in the Ukrainian populations (Pupins et al., 2019). However, lower hatchling weights were reported from the southern E. orbicularis populations: 4.9 g in Hungary (Kiss et al., 2021), 4.8 g in Spain (Diaz-Paniagua, 2014), and 3.6 g in Turkey (Ayaz et al., 2017). Our studied hatchlings had a bigger mean plastron length (24.0 mm) compared to hatchlings of Turkish (19.6 mm; Ayaz et al., 2017) or Spanish populations (22.3 mm; Diaz-Paniagua et al., 2014). Our results clearly confirm the tendency described by Pupins et al. (2019), who compared E. orbicularis hatchlings from different geographic regions and found that hatchlings in the northern parts of the range are larger than in the southern parts. Joss et al. (2017) also found a correlation between latitude and E. orbicularis body size. This and the aforementioned studies support Bergmann’s rule (Bergmann, 1848), which states that animals in colder climates have larger body sizes than those in warmer climates.
The size and weight of E. orbicularis hatchlings vary due to many factors, including the age of the nesting female, the size of the clutch, and the incubation temperature of eggs (year factor) (Pupins et al., 2019). It is vital to understand the size and sex ratio of hatchlings, especially when comparing artificial vs natural incubation and further hatchlings rearing. This study found that hatchlings attain larger weight if incubated naturally vs. artificially. This is because the incubation time of artificial eggs is much shorter (57-80 days) than natural eggs (Pupins et al., 2019) with 85-113 days reported in Poland (Mitrus and Zemanek, 2000), 90-117 days in Austria (Rössler, 2000), and 83 days in Spain (Diaz-Paniagua et al., 2014). However, the initially observed hatchling weight differences disappeared by the end of the 10-month rearing period.
Moreover, newly hatched females were bigger in both weight and length compared to juvenile males. It is important to note that even at egg stage, “female” eggs were significantly larger than “male” eggs. The observed initial gender differences remained unchanged until the end of rearing. Other studies have also reported larger E. orbicularis females (Zuffi et al., 1999; Fediras et al., 2017; Liuzzo et al., 2021).
The study established a clear female-dominated bias in hatchlings from both artificial and natural incubation methods, with a pronounced female-skewed ratio observed in the artificially incubated eggs. E. orbicularis, like other thermophilous species, exhibits a discernible sensitivity to temperature fluctuations (Sommer et al., 2007; 2009; Joos et al., 2017; Cerasoli et al., 2019; Nekrasova et al., 2021). The incubation temperature has a huge impact on hatchlings, determining their sex and influencing survival. At higher temperatures (more than 29.5 °C), hatches occur exclusively of female turtles, while at lower temperatures (below 27.5 °C), hatches occur exclusively of male turtles. At an intermediate temperature (28.5 °C), the hatchling ratio of females and males is equal (Zaborski et al., 1988). In our research, E. orbicularis eggs were incubated at 25-27 °C during the night and at 28-29.5 °C during the day. We observed that on these temperature conditions, the sex ratio of hatchlings was 2.3/1 (female/male). Individuals excavated from nests that experienced lower temperatures during the incubation period under natural conditions had an almost equal female/male ratio (1.2/1).
We experimented with raising E. orbicularis hatchlings in an artificial environment for their first year to reduce the high winter mortality rate in the wild. The hatchlings were raised in either an artificial or natural incubation process, and their survival rates were the same. The survival rate of the reared hatchlings in this experiment was an impressive 98.8%, which is significantly higher than the 77.8% survival rate of artificially reared hatchlings in Slovakia (Bona et al., 2012) and much higher than the 7.1% survival rate of E. orbicularis overwintering hatchlings in nests under natural conditions (Bona et al., 2012).
The decrease in E. orbicularis populations in Europe, especially in its northern distribution edge, requires special protection measures, primarily for the critical early life stages. The methodologies for artificial E. orbicularis egg incubation and further hatchling rearing we fine-tuned resulted highly effective and significantly increased E. orbicularis hatching success and survival. Artificially reared E. orbicularis individuals could be released to the wild in equal numbers of males and females, which would help balance the skewed sex ratio in their small, threatened populations as well as help to protect the overall genetic diversity of E. orbicularis in its northern distribution range.
The research was funded by the project “Implementation of Nature Conservation and Management Measures by Preserving and Increasing the Populations of European Pond Turtles and European Fire-bellied Toad” (Nr. 05.4.1-APVA-V-018-01-0004).
State laws on handling wild animals were followed, with six permits (Nos. 50, 14, 15, 22, 15, 100) from the state Environmental Protection Agency being issued for collecting, handling, rearing and releasing reared individuals of pond turtles back into the wild during the whole study period.
Supplementary material associated with this article can be found at <http://www.unipv.it/webshi/appendix> Manuscript number 16266.
Avanzi, M., Millfanti, M. (2003): Le grand livre des tortues terrestres et aquatiques. De Vecchi Edition.
Ayaz, D., Çiçek, K., Bayrakci, Y., Tok, C.V. (2017): Reproductive ecology of the European pond turtle, Emys orbicularis (Linnaeus, 1758), from Mediterranean Turkey. Acta Zool Bulg. 10: 23-29.
Bastytė-Cseh, D. (2021): Balinis vėžlys Emys orbicularis (Linnaeus, 1758). In: Red Data Book of Lithuania. Animals, plants, fungi, p. 212. Rašomavičius, V. Ed., Vilnius, Lututė.
Bergmann, C. (1848): Über die Verhältnisse der Wärmeökonomie der Thiere zu ihrer Grösse. Vandenhoeck und Ruprecht.
Bona, M., Novotny, M., Danko, S., Buresova, A. (2012): Headstarting in a small population of European pond turtles (Emys orbicularis) in Central European conditions: first results. Herpetol. Notes 5: 547-550.
Berthomieu, L., Vermeer, J. (2021): EAZA Best Practice Guidelines for the European pond turtle (Emys orbicularis), European Association of Zoos and Aquariums, Amsterdam, The Netherlands.
Cerasoli, F., Iannella, M., Biondi, M. (2019): Between the hammer and the anvil: how the combined effect of global warming and the non-native common slider could threaten the European pond turtle. Manag. Biol. Invasions 10: 428-448.
Chelazzi, G., Lebboroni, M., Tripepi, S., Utzeri, C., Zuffi, M.A.L. (2000): A primer on the conservation biology of the European pond turtle, Emys orbicularis, of Italy. In: Proc. II Int. Symp. on Emys orbicularis, Blanc, Brenne. Chelonii 2: 101-104
Diaz-Paniagua, C., Andreu, A.C., Marco, A., Nuez, M., Hidalgo-Vila, J., Perez-Santigosa, N. (2014): Data on nesting, incubation, and hatchling emergence in the two native aquatic turtle species (Emys orbicularis and Mauremys leprosa) from Doñana National Park. Basic Appl. Herpetol. 28: 147-151.
Drobenkov, S.M. (2000): Reproductive ecology of the pond turtle (Emys orbicularis L.) in the northeastern part of the species range. Russ. J. Ecol. 31: 49-54.
Fediras, S., Rouag, R., Ziane, N., Olivier, A., Béchet, A., Benyacoub, S. (2017): Population structure and morphometrics of the European pond turtle (Emys orbicularis (Linnaeus, 1758)) at Lake Tonga, Algeria. Russ. J. Herpetol. 25: 88-96
Frıtz, U. (2003): Die Europäische Sumpfschildkröte. Supplement der Zeitschrift für Feldherptologie No. 1. Bielefeld: Laurenti-Verlag. 224 p.
Fritz, U., Chiari, Y. (2013): Conservation actions for European pond turtles–a summary of current efforts in distinct European countries. Herpetol. Notes 6: 105.
Isailovic, J.C., Mesaroš G. (2013): Conservation activities for the European pond turtle (Emys orbicularis) in Serbia. Herpetol. Notes 6: 119-121.
Jablonski, A., Jablonska, S. (1998): Egg-laying in the European pond turtle, Emys orbicularis (L.). Leczynsko–Wlodawskie Lake District (east Poland). Mertensiella 10: 141-146.
Joyal, L.A., McCollough, M., Hunter Jr., M.L. (2001): Landscape ecology approaches to wetland species conservation: a case study of two turtle species in southern Maine. Con. Biol. 15: 1755-1762.
Joos, J., Kirchner, M., Vamberger, M., Kaviani, M., Rahimibashar, M. R., Fritz, U., Müller, J. (2017): Climate and patterns of body size variation in the European pond turtle, Emys orbicularis. Biol. J. Linn. Soc. 122: 351-365.
Kiss, I., Erdelyi, G., Szabo, B. (2021): Nesting activity and reproductive success of Emys orbicularis in Babat Valley (Gödöllő, Hungary). Herpetol. Conserv. Biol. 16: 624-638.
Liuzzo, M., Borella, S., Ottonello, D., Arizza, V., Malavasi, S. (2021): Population abundance, structure and movements of the European pond turtle, Emys orbicularis (Linnaeus 1758) based on capture-recapture data in a Venice Lagoon wetland area, Italy. Ethol. Ecol. Evol. 33: 561–575.
Liuzzo, M., Fantinato E., Malavasi S. (2023): Successful reproduction of feral Trachemys scripta (Schoepff, 1792) in an inland wetland of the Veneto region, Le Basse del Brenta, Italy. Herpotol. Notes 16: 465-469.
Liuzzo, M., Spada, A., Facca, C., Borella, S., Malavasi, S. (2024): Nesting habitat characteristics and predation patterns in the European pond turtle Emys orbicularis (L., 1758): implications for management and conservation measures. Glob. Ecol. and Conserv. 52: e02975
Luiselli, L., Vamberger, M. (2024): Emys orbicularis (Europe assessment). The IUCN Red List of Threatened Species 2024: e.T7717A207667247.
Marchand, M.N., Litvaitis, J.A. (2004): Effects of landscape composition, habitat features, and nest distribution on predation rates of simulated turtle nests. Biol. Conserv. 117: 243-251.
Meeske, A.C.M., Pupins, M. (2009): The European pond turtle in Latvia. Pp. 214-216. In: European Pond Turtles. The genus Emys. Rogner, M., Ed, Frankfurt, Chimaira.
Meeske, M. (1997): Nesting ecology of European pond turtle (Emys orbicularis) in South Lithuania. Acta Zool. Lit. 7: 138-142.
Meeske, M., Pupins, M., Rybczynski, K. (2006): First results on the distribution and condition of the European pond turtle (Emys orbicularis) at the northern edge of its distribution in Lithuania and Latvia (in German). Z. Feldherpetol. 13: 71-99.
Mitrus, S., Zemanek, M. (2004): Body size and survivorship of the European pond turtle Emys orbicularis in Central Poland. Biol. Brat. 59: 103-107.
Mitrus, S. (2008): Relocation of the European pond turtle Emys orbicularis clutches did not influence the hatching rate. Acta Biol. Univ. Daugavp. 8: 63-66.
Mitrus, S., Najbar, B., Kotowicz, A., Najbar, A. (2018): The body size of headstarted and wild juvenile European pond turtles (Emys orbicularis). Russ. J. Herpetol. 25: 161-164.
Mitrus, S., Zemanek, M. (1998): Reproduction of Emys orbicularis (L.) in central Poland. Mertensiella 10: 187-191.
Mitrus, S., Zemanek, M. (2000): Distribution and biology of Emys orbicularis (L.) in Poland. Stapfia 69: 107-118.
Mollov, I., Naumov, B., Dobrev, D., Popgeorgiev, G. (2013): Conservation activities for European pond turtles (Emys orbicularis) in Bulgaria. Herpetol. Notes 6: 135-138.
Najbar, B., Szuszkiewicz, E. (2005): Reproductive ecology of the European pond turtle Emys orbicularis (LINNAEUS, 1758) (Testudines: Emydidae) in Western Poland. Acta Zool. Cracov. 48: 11-19.
Nekrasova, O., Marushchak, O., Pupins, M., Skute, A., Tytar, V., Čeirāns, A. (2021): Distribution and potential limiting factors of the European pond turtle (Emys orbicularis) in Eastern Europe. Diversity 13: 280.
Novotny, M., Danko, S., Havaš, P. (2004): Activity cycle and reproductive characteristics of the European pond turtle (Emys orbicularis) in the Tajba National Nature Reserve, Slovakia. Biol. Brat. 59: 113-121.
Pieau, C., Dorizzi, M. (1981): Determination of temperature sensitive stages for sexual differentiation of the gonads in embryos of the turtle, Emys orbicularis. J. Morphol. 170: 373-382.
Pupins, M., Nekrasova, O., Marushchak, O., Dubyna, A., Neizhko, I. (2019): Morphological features of European pond turtles’ Emys orbicularis (Linnaeus, 1758) hatchlings at the northern edge (Latvia) and central part (Ukraine) of its distribution range. Pp. 11-13. In: Proceedings of the II Congresso Nazionale Testuggini e Tartarughe, Italy, Albenga.
Pupins, M., Pupina, A. (2008): Distribution of European pond turtle Emys orbicularis (Linnaeus, 1758) on the northern edge of its area in Latvia. Rev. Esp. Herp. 22: 149-157.
Purger, J.J., Molnár, T.G., Lanszki, Z., Lanszki, J. (2023): European pond turtle (Emys orbicularis) nest predation: A study with artificial nests. Biology 12: 342.
Rovero, F., Chelazzi, G. (1996): Nesting migrations in a population of the European pond turtle Emys orbicularis (L.) (Chelonia Emydidae) from central Italy. Ethol. Ecol. Evol. 8: 297-304.
Rössler, M. (2000): Der Lebensraum der Europaischen Sumpfschildkröte Emys orbicularis (L.) in den niederösterreichischen Donau-Auen. Stapfia 69: 145-156.
Salom-Oliver, M., Amengual, A.R.H., Aguiló-Zuzama, A., Ribas-Serra, A., Vallespir, J., Tejada-Gavela, S., Pinya, S. (2022): Plastron color patterns allow for individual photo-identification in two different chelonian species. Can. J. Zool. 100: 789-798.
Savic, A. (2010): Distribution of and potential factors of threat for Emys orbicularis (Linne 1758) within city of Niš and surrounding area. B.Sc. thesis, Faculty of Sciences and Mathematics, University of Niš (In Serbian, English summary).
Schindler, M., Frötscher, H., Hille, A., Bruck, M.R., Schmidt, M., Kornilev, Y.V. (2017): Nest protection during a long-term conservation project as a tool to increase the autochthonous population of Emys orbicularis (L., 1758) in Austria. Acta Zool. Bulg. 10: 147-154.
Schmidt S. D. (2017): Evaluation of habitat features influencing turtle abundance in the Donau-Auen. Wissenschaftliche Reihe Nationalpark Donau-Auen 70: 1–211. Orth an der Donau: Nationalpark Donau-Auen.
Schneeweiß, N. (2004): Climatic impact on reproductive success of Emys orbicularis at the northwestern border of the species range. Biologia 59: 131-137.
Schneeweiß, N., Breu, H. (2013): Conservation activities for the European pond turtle (Emys orbicularis) in Germany. Herpetol. Notes 6: 113-115.
Sommer, R.S., Lindqvist, C., Persson, A., Bringsøe, H., Rhodin, A.G., Schneeweiss, N., Široky, P., Bachmann, L., Fritz, U. (2009): Unexpected early extinction of the European pond turtle (Emys orbicularis) in Sweden and climatic impact on its Holocene range. Mol. Ecol. 18: 1252-1262.
Sommer, R.S., Persson, A., Wieseke, N., Fritz, U. (2007): Holocene recolonisation and extinction of the pond turtle, Emys orbicularis (L., 1758), in Europe. Quat. Sci. Rev. 26: 3099-3107.
Tetzlaff, S.J., Estrada, A., DeGregorio, B.A., Sperry, J.H. (2020): Identification of factors affecting predation risk for juvenile turtles using 3D printed models. Animals 10: 275.
Vecchioni, L., Marrone, F., Arculeo, M., Fritz, U., Vamberger, M., 2020. Stand out from the crowd: small-scale genetic structuring in the endemic sicilian pond turtle. Diversity 12: 343.
Zaborski, P., Dorizzi, M., Pieau, C. (1988): Temperature-dependent gonadal differentiation in the turtle Emys orbicularis: concordance between sexual phenotype and serological HY antigen expression at threshold temperature. Differentiation 38: 17-20.
Zinenko, O. (2004): Notes on egg-laying, clutch size and hatchling feeding of Emys orbicularis in the Kharkiv region, Ukraine. Biol. Brat. 59: 149-151.
Zuffi, M.A.L., Odetti, F. (1998): Double egg deposition in the European pond turtle, Emys orbicularis, from central Italy. Ital. J. Zool. 65: 187-189.
Zuffi, M. A. L., Odetti, F., Meozzi, P. (1999): Body size and clutch size in the European pond turtle (Emys orbicularis) from central Italy. J. Zool., 247: 139-143.
Zuffi, M.A.L. (2000): Biology of the conservation of the European pond turtle, Emys orbicularis, of Italy. Stapfia 69: 219-228.
Zuffi, M.A.L., Mangiacotti, M., Masucci, G. D., Sacchi, R., Scali, S., Sannolo, M. (2017): Stable or plastic body shape? Emys orbicularis hatchlings-juveniles growth patterns under different ecological conditions. North-West. J. Zool. 13: 262-270.
Zuffi, M.A.L., Rovina, L. (2006): Habitat characteristics of nesting areas and of predated nests in a Mediterranean population of the European pond turtle, Emys orbicularis. Acta Herpetol. 1: 37-50.