Submitted on: 2024, 20th June; revised on: 2024, 20th June; accepted on: 2024, 28th November
Screening for Batrachochytrium salamandrivorans in a population of Golden Alpine Salamanders at the edge of their distribution range
1 Consiglio Nazionale delle Ricerche - Istituto per la BioEconomia, Via dei Taurini 19, I-00185 Roma, Italy
2 Ambito Biologia della Conservazione, Ufficio Ricerca e Collezioni, MUSE—Museo delle Scienze, Corso del Lavoro e della Scienza 3, I-38122 Trento, Italy
3 Department of Earth, Environment and Life Sciences (DISTAV), University of Genova, Corso Europa 26, I-16132 Genova, Italy
*Corresponding authors. Email: antonio.romano@cnr.it; luca.roner@muse.it
Abstract. Amphibian populations worldwide are experiencing significant declines, highlighting a critical aspect of the broader biodiversity crisis. Approximately 43% of all amphibian species are facing extinction due to factors such as habitat loss, pollution, climate change, and emerging diseases. The chytrid fungus Batrachochytrium salamandrivorans (Bsal) represents one of the major threats, because it is particularly dangerous for European salamanders. Southern Europe is especially vulnerable due to the presence of numerous endemic salamander species. Despite the risks, few studies have screened Italian salamanders for Bsal. We conducted a Bsal screening on 44 Golden Alpine Salamanders (Salamandra atra aurorae) from the Vezzena plateau in the Trentino-Alto Adige region (Northern Italy). Our molecular analysis of skin swabs revealed no presence of Bsal in any of the 44 specimens examined. Additionally, no macroscopic signs of Bsal-related skin damage were observed. The absence of Bsal in our samples is encouraging, suggesting that the investigated sites are currently unaffected by this pathogen. This finding aligns with other studies reporting no evidence of Bsal in Italy. Future research should explore the factors contributing to the absence of Bsal and the effectiveness of current conservation practices. While our findings are reassuring, the threat of Bsal remains a critical concern. Continued vigilance and enhanced conservation efforts are essential to protect salamander diversity and ensure the long-term survival of these important amphibian populations.
Keywords. Amphibians, Chytridiomycosis, conservation, endemic taxon, threats.
Amphibian populations worldwide are experiencing a significant decline, underscoring a critical aspect of the broader biodiversity crisis. Approximately 43% of all amphibian species face extinction due to factors like habitat loss, pollution, climate change, and emerging diseases (Luedtke et al., 2023). Two chytrid fungi, Batrachochytrium dendrobatidis (Bd) and B. salamandrivorans (Bsal), pose major threats to amphibians’ conservation. They are capable of causing mass die-offs likely due to pathogen pollution (caused by global animal trade for food, collecting, etc.) and the susceptibility of species naïve to new chytrid lineages (McKenzie et al., 2012; Martel et al., 2013; Rosa et al., 2013; O’Hanlon et al., 2018). Although our understanding of the immune response is still limited, Bsal is especially dangerous for European salamanders due to their susceptibility and the virulence factors of this fungal pathogen, which can lead to immune system compromise, tissue erosion, and impaired respiratory and rehydration functions (Martel et al., 2014; Stegen et al., 2017; Grogan et al., 2020).
Southern Europe, including the Italian and Iberian peninsulas, is particularly vulnerable due to numerous endemic salamander species. Italy, for instance, hosts 19 species of urodeles, many of which are endemic (Sindaco and Razzetti, 2021). Despite the risks, only in recent years few studies have screened Italian salamanders for Bsal (Grasselli et al., 2019; Grasselli et al., 2021; Bernabo et al., 2024; Böning et al., 2024). Although Bsal has not yet been reported in Italian salamander populations, the entire urodeles fauna remains at risk from potential pathogen pollution. Therefore, proactive monitoring of Bsal in wild amphibian populations is crucial.
The Alpine salamander, Salamandra atra Laurenti 1758, has been found to be highly susceptible to Bsal in captivity (Fitzpatrick et al., 2018), raising significant conservation concerns. This species, restricted to the European and Dinaric Alps, includes several intraspecific lineages, with some recognized as subspecies (Bonato et al., 2018). The Italian endemic subspecies, S. a. aurorae Trevisan 1982, and S. a. pasubiensis Bonato and Steinfartz 2005, inhabit areas of less than 100 km² each. In a recent study involving 90 individuals of S. atra, including 28 S. a. aurorae, the Golden Alpine Salamander, no presence of Bsal was found (Böning et al., 2024). Here, we report the results of a Bsal screening conducted on a population of the Golden Alpine Salamander from Trentino province. This locality differs from those sampled in Böning et al. (2024) and is located at the edge of the distribution range of this taxon.
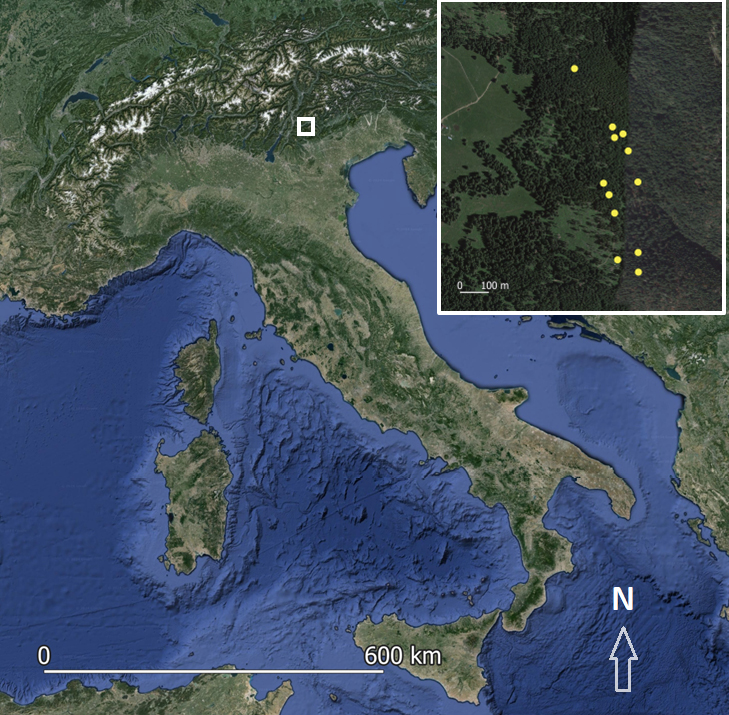
The study area (Fig. 1) encompasses a small segment of the Golden Alpine Salamander’ range, situated on the Vezzena plateau in the Trentino Alto Adige region (45°57′10″N, 11°22′25″E) at an elevation of approximately 1450 meters above sea level. The Vezzena plateau is characteristic of the Alpine Mountain region’s general climatic conditions. Forty-four Golden Alpine salamanders (10 females, 31 males, 3 juveniles) were captured on summer 2020, measured (total length, weight) and sexed. All skin swabs were collected using a standardized protocol (Blooi et al., 2013), specifically following the procedure established in previous Bd and Bsal studies on Italian salamanders (Grasselli et al., 2019; Costa et al., 2021; Grasselli et al., 2021). Sterile cotton swabs were rubbed 30 times on various parts of the salamander’s body and stored in individual sterile plastic tubes at 4 °C until extraction (Dondero et al., 2023). Additionally, all individuals were visually inspected for any physical abnormalities and signs of chytridiomycosis like skin lesions and ulcerations (Martel et al., 2013).
Laboratory protocols for Bd and Bsal detection
Nucleic acid extraction from skin swabs for quantitative PCR (qPCR) was performed according to the method described by Boyle et al. (2004). In brief, nucleic acids were extracted using 200 μL of PrepMan Ultra - Sample Preparation Reagent (Thermo-Fisher Scientific Technologies, Monza, Italy) and 0.03-0.04 g of Zirconium/silica beads (Biospec Products). The extraction involved two rounds of incubation in a Bead Beater (MM200 - Retsch GmbH, Hann, Germany) for 1 minute at the highest frequency (25 Hz), followed by centrifugation at 13,000 g for 1 minute. This was followed by incubation at 100 °C for 10 minutes in a Dry Block Thermostat, and a subsequent centrifugation at 13,000 g for 3 minutes. The supernatant was then recovered and stored at -20 °C until Real-Time PCR analysis was performed. Samples were analyzed in at least duplicate for the presence of Bsal DNA using a SYBR Green- based Real-Time PCR assay targeting the 5.8S rRNA gene of Bsal, as described by Blooi et al. (2013), without the employ of the probe, as in the original assay. The Bsal SYBR Green assay was conducted on a CFX96 real-time system (Bio-Rad Laboratories, Hercules, CA) with a reaction mixture comprising 12.5 µL iQ™ SYBR® Green Supermix (Bio-Rad Laboratories, Hercules, CA), 300 nM of each primer (Bsal fwd primer: SterF - 5′-AGCCAAGAGATCCGTTGTCAAA-3′; Bsal rev primer: SterR - 5′-TGAACGCACATTGCACTCTAC-3’), 5 μL of template, and RNase/DNase-free water to a total volume of 25 μL per reaction. To quantify Bsal DNA, ten-fold serial dilutions of Bsal DNA were prepared, achieving concentrations ranging from 1,000 to 0.1 genomic equivalents (GEs) of zoospores per reaction mixture, as per Thomas et al. (2018). Bsal DNA standards for qPCR were kindly provided by Prof. An Martel and Frank Pasmans (University of Ghent, Belgium). The amplification conditions were set at 10 minutes at 95 °C, followed by 40 cycles of 15 seconds at 95 °C and 15 seconds at 62 °C. A melting curve from 60 to 95 °C was obtained, with readings at every 0.5°C increment (Blooi et al., 2013).
It is noteworthy that our results are derived from a modification of the Blooi et al. 2013 method, as we did not include the probe in our assay. The absence of positive samples within the salamander population is accompanied by a positive amplification of the standard curve.
The males were on average 12.6 cm long (range: 10.7-14.9 cm; SD = 0.95), the females 13.0 cm (range: 12.0-13.9 cm; SD = 0.67), the juveniles 10.3 cm (range: 6.6-12.6 cm; SD = 2.07), and weighed 7.9 g (range: 5.3-9.7 g; SD = 1.08), 9.6 g (range: 7.4-13.9 g; SD = 1.82) and 5.0 g (range: 3.9-6.2; SD = 1.13) respectively. Our molecular analysis of skin swabs revealed the absence of Bsal in all 44 specimens examined in this study. Additionally, no Bsal-typic macroscopic skin damage was observed during our surveys.
Bsal infection poses a major threat to salamander diversity in Central Europe and Spain (Martel et al., 2014; Bosch et al., 2021) and, in future projections, particularly to Italian amphibians (Luedtke et al., 2023; but see the critique in Canessa et al., 2024). However, none of the qPCR samples from our study sites tested positive for Bsal infection. The relatively high number of salamanders we sampled from the same population should ensure accurate screening, capable of detecting infections even at low prevalence rates. The absence of Bsal infection in the qPCR samples collected from our investigated sites is an encouraging finding. This result suggests that, at least for the moment, the sites under study remain unaffected by this pathogen. Our finding align with the study by Dondero et al. (2023), Bernabo et al. (2024) and Böning et al. (2024), who also reported no evidence of Bsal presence in Italy.
Several factors may contribute to the absence of Bsal in our sampled areas. Simulation studies, employing Species Distribution Models (SDMs) to anticipate the spread of invasive species, have considered bioclimatic suitability, salamander species richness, and salamander imports (Katz and Zellmer, 2018). These studies suggest that, although most Italian salamanders are highly susceptible to chytrid fungus (Beukema et al., 2018; Dondero et al., 2023), the ecological and climatic conditions in Italy are not optimal for Bsal. Consequently, the risk of pathogen spread in Italy seems to be relatively low. However, the specific area we studied is nationally recognized as having the highest probability (refer to Fig. 3 in Katz and Zellmer, 2018).
One possibility is that the geographic or environmental conditions in these regions are not conducive to the survival or spread of Bsal. Additionally, local salamander populations might possess some level of resistance or immunity to the pathogen, which could prevent its establishment.
The current absence of Bsal in the study area provides a valuable opportunity to implement proactive preventive measures. As highlighted by Thomas et al. (2019), effective mitigation strategies in Bsal-free areas include establishing early warning systems, continuous surveillance, and stringent biosecurity protocols. The surveyed area has traditionally been subject to forest management, ungulate hunting, and regulated mushroom picking, with more recent and limited recreational activities, such as outdoor walking. Completely restricting access for biosecurity reasons, however, is both impractical and potentially ineffective, particularly within the study area in Trentino. This is because the Trentino populations of the salamanders is territorially contiguous with the area in Veneto occupied by the rest of the population of this taxon. Nevertheless, other targeted measures are both feasible and necessary. These include regular (e.g., biennial) screening for Bsal infection, educational initiatives emphasizing the importance of practices like shoe disinfection, and managing human access if recreational use significantly increases. It is particularly important to emphasize the need for health precautions for herpetologists conducting studies in the area. These researchers must continue adhering to the health protocols established by the Societas Herpetologica Italica (http://www-9.unipv.it/webshi/conserv/monitanf.htm), which they have so far complied with. This is crucial, as the health impact of herpetological research may not be negligible (Razzetti and Bonini, 2001). The relative isolation of the habitat and the currently low intensity of human activity are favourable factors, potentially reducing the risk of pathogen introduction. However, these conditions should not lead to complacency, and ongoing surveillance is essential to ensure the area remains pathogen-free. This aligns with broader efforts in Italy, where continuous surveillance for herpetofaunal diseases, as outlined by Marini et al. (2023), has proven effective. Regular monitoring of salamander populations and their habitats can help detect early signs of Bsal infection, enabling swift responses to prevent the pathogen’s establishment and spread.
Captured animals were handled with the authorisation of the Italian Ministry of Ecological Transition (PNM-2018-0006709). Thanks to Giulia Bombieri, Claudia Pellegrini and Aurora Colangelo for the support in the field. The municipality of Levico Terme (TN) provided logistical facilities. The Autonomous Province of Trento supported the research activity.
Bernabò, I., Cittadino, V., Iannella, M., Corapi, A., Tiberti, R., Dondero, L., Grasselli, E., Romano, A. (2024): A taxon under siege from multiple threats: results of analysis of chytrid fungi on the Calabrian alpine newt. Amphib.-Reptil. 46: 1-12.
Beukema, W., Martel, A., Thien Nguyen, T.T, Goka, K, Schmeller, D.S., Yuan, Z., Laking, A.E., Quang Nguyen, T., Lin, C-F., Shelton, J., Loyau, A., Pasmans, F. (2018): Environmental context and differences between native and invasive observed niches of Batrachochytrium salamandrivorans affect invasion risk assessments in the Western Palaearctic. Divers. Distrib. 24: 1788-1801.
Blooi, M., Pasmans, F., Longcore, J.E., Spitzen-van der Sluijs, A., Vercammen, F., Martel, A. (2013): Duplex Real-Time PCR for Rapid Simultaneous Detection of Batrachochytrium dendrobatidis and Batrachochytrium salamandrivorans in Amphibian Samples. J. Clin. Microbiol. 54: 246-246.
Bonato, L., Corbetta, A., Giovine, G., Romanazzi, E., Sunje, E., Vernesi, C., Crestanello, B. (2018): Diversity among peripheral populations: genetic and evolutionary differentiation of Salamandra atra at the southern edge of the Alps. J. Zool. Syst. Evol. Res. 56: 533-548.
Böning, P., Lötters, S., Barzaghi, B., Bock, M., Bok, B., Bonato, L., Ficetola, G.F., Glaser, F., Griese, J., Grabher, M., Leroux, C., Munimanda, G., Manenti, R., Ludwig, G., Preininger, D., Rödel, M.O., Seibold, S., Smith, S., Tiemann, L., Thein, J., Veith, M., Plewnia, A. (2024): Alpine salamanders at risk? The current status of an emerging fungal pathogen. PLOS One 19: e0298591.
Bosch, J., Martel, A., Sopniewski, J., Thumsova, B., Ayres, C., Scheele, B.C., Guillermo Velo-Anton, G., Pasmans, F. (2021): Batrachochytrium salamandrivorans threat to the Iberian urodele hotspot. J. Fungi 7: 644.
Boyle, D.G., Boyle, D.B., Olsen, V., Morgan, J.A.T., Hyatt, A.D. (2004): Rapid quantitative detection of chytridiomycosis (Batrachochytrium dendrobatidis) in amphibian samples using real-time Taqman PCR assay. Dis. Aquat. Organ. 60: 141-148.
Canessa, S., Biancolini, D., Iannella, M., Bernabò, I., Salvi, D., Vignoli, L., Lunghi, E., Costa,E.,Razzetti, E., Ficetola, G.F., Romano,A. (2024): Uncertain future and uncertain projections: assessing extinction risks in European salamanders from projected chytrid fungus invasion using IUCN Criterion E. Acta Herpetol. 19: 155-159.
Costa, A., Dondero, L., Allaria, G., Morales Sanchez, B.N., Rosa, G., Salvidio, S., Grasselli, E. (2021): Modelling the amphibian chytrid fungus spread by connectivity analysis: towards a national monitoring network in Italy. Biodivers. Conserv. 30: 2807-2825.
Dondero, L., Allaria, G., Rosa, G., Ficetola, G. F., Cogoni, R., Grasselli, E., Salvidio, S. (2023): Threats of the emerging pathogen Batrachochytrium salamandrivorans (Bsal) to Italian wild salamander populations. Acta Herpetol. 18: 3-9.
Fitzpatrick, L.D., Pasmans, F., Martel, A., Cunningham, A.A. (2018): Epidemiological tracing of Batrachochytrium salamandrivorans identifies widespread infection and associated mortalities in private amphibian collections. Sci. Rep. 8: 13845.
Grasselli, E., Bianchi, G., Dondero, L., Marchiano, V., Carafa, M., Perrone, M., Salvidio, S. (2019): First screening of Batracochytrium salamandrivorans (Bsal) in wild and captive salamanders from Italy. Salamandra 55: 124-126.
Grasselli, E., Bianchi, G., Salvidio, S. (2021): Batrachochytrium salamandrivorans in Europe: the Italian contribution to the international mitigation project. In: Atti del XII Congresso Nazionale della Societas Herpetologica Italica (Rende, Cosenza), pp. 214-217. Tripepi, S., Ed, Ventura Edizioni, Senigallia.
Grogan, L.F., Humphries, J.E., Robert, J., Lanctot, C.M., Nock, C.J., Newell, D.A., McCallum, H.I. (2020): Immunological aspects of chytridiomycosis. J. Fungi 6: 234.
Katz, T.S., Zellmer, A.J. (2018): Comparison of model selection technique performance in predicting the spread of newly invasive species: a case study with Batrachochytrium salamandrivorans. Biol. Invasions 20: 2107-2119.
Luedtke, J. A., Chanson, J., Neam, K., Hobin, L., Maciel, A. O., Catenazzi, A., Borzée, A., Hamidy, A., Aowphol, A., Jean, A., Sosa-Bartuano, Á., Fong G, A., de Silva, A., Fouquet, A., Angulo, A., Kidov, A. A., Muñoz Saravia, A., Diesmos, A. C., Tominaga, A., Shrestha, B., … Stuart, S. N. (2023): Ongoing declines for the world’s amphibians in the face of emerging threats. Nature 622: 308-314.
Marini, D., Filippi, E., Montinaro, G., Origgi, F.C., (2023): Screening of Ophidiomyces ophidiicola in the free-ranging snake community annually harvested for the popular ritual of San Domenico e dei Serpari (Cocullo, AQ, Italy). Acta Herpetol. 18: 45-52.
Martel, A., Blooi, M., Adriaensen, C., Van Rooij, P., Beukema, W., Fisher, M.C., Farrer, R.A., Schmidt, B.R., Tobler, U., Goka, K., Lips, K.R., Muletz, C., Zamudio, K., Bosch, J., Lotters, S., Wombwell, E., Garner, T.W.J., Cunningham, A.A., Spitzen-van der Sluijs, A., Salvidio, S., Ducatelle, R., Nishikawa, K., Nguyen, T.T., Kolby, J.E., Van Bocxlaer, I., Bossuyt, F., Pasmans, F. (2014): Recent introduction of a chytrid fungus endangers Western Palearctic salamanders. Science 346: 630-631.
Martel, A., Spitzen-van der Sluijs, A., Blooi, M., Bert, W., Ducatelle, R., Fisher, M.C., Woeltjes, A., Bosman, W., Chiers, K., Bossuyt, F. (2013): Batrachochytrium salamandrivorans sp nov causes lethal chytridiomycosis in amphibians. Proc. Natl. Acad. Sci. USA 110: 15325-15329.
McKenzie, J.V., Peterson A.C. (2012): Pathogen pollution and the emergence of deadly amphibian pathogen. Mol. Ecol. 21: 5151-5154.
O’Hanlon, S.J., Rieux, A., Farrer, R.A., Rosa, G.M., Waldman, B., Bataille, A., Kosch, T.A., Murray, K.A., Brankovics, B., Fumagalli, M., Martin, M.D., Wales, N., Alvarado-Rybak, M., Bates, K.A., Berger, L., Boll, S., Brookes, L., Clare, F., Courtois, E.A., Cunningham, A.A., Doherty-Bone, T.M., Ghosh, P., Gower, D.J., Hintz, W.E., Hoglund, J., Jenkinson, T.S., Lin, C-F., Laurila, A., Loyau, A., Martel, A., Meurling, S., Miaud, C., Minting, P., Pasmans, F., Schmeller, D.S., Schmidt, B.R., Shelton, J.M.G., Skerratt, L.F., Smith, F., Soto-Azat, C., Spagnoletti, M., Tessa, G., Toledo, L.P., Valenzuela-Sanchez, A., Verster, R., Voros, J., Webb, R.J., Wierzbicki, C., Wombwell, E., Zamudio, K.E., Aanensen, D.M., James, T.Y., Thomas, M., Gilbert, P., Weldon, C., Bosch, J., Balloux, F., Garner, T.M.J., Fisher, M.C. (2018): Recent Asian origin of chytrid fungi causing global amphibian declines. Science 360: 621-627.
Razzetti, E., Bonini, L. (2001): Infezioni e parassitosi negli Anfibi: il possibile impatto delle ricerche erpetologiche. Atti Soc. It. Sc. Nat. 14: 97-102.
Rosa, G. M., Anza, I., Moreira, P. L., Conde, J., Martins, F., Fisher, M. C., Bosch, J. (2013): Evidence of chytrid‐mediated population declines in common midwife toad in Serra da Estrela, Portugal. Anim. Conserv. 16: 306-315.
Sindaco, R., Razzetti, E. (2021): An updated check-list of Italian amphibians and reptiles. Nat. Hist. Sci. 8: 35-46.
Stegen, G., Pasmans, F., Schmidt, B.R., Rouffaer, L.O., Van Praet. S., Schlaub, M., Canessa, S., Laudelout, A., Kinet, T., Adriaensen, C., Haesebrouck, F., Bert, W., Bossuyt, F., Martel, A. (2017): Drivers of salamander extirpation mediated by Batrachochytrium salamandrivorans. Nature 544: 353-356.
Thomas, V., Blooi, M., Van Rooij, P., Van Praet, S., Verbrugghe, E., Grasselli, E., Lukac, M., Smith, S., Pasmans, F., Martel, A. (2018): Recommendations on diagnostic tools for Batrachochytrium salamandrivorans. Transbound. Emerg. Dis. 65: e478-e488.
Thomas, V., Wang, Y., Van Rooij, P., Verbrugghe, E., Baláž, V., Bosch, J., Cunningham, A.A., Fisher, M.C., Garner, T.W.J.,Gilbert, M.J., Grasselli, E., Kinet, T., Laudelout, A., Lötters, S., Loyau, A., Miaud, C., Salvidio, S., Schmeller, D.K., Schmidt, B.R., Spitzen-van der Sluijs, A., Steinfartz, S., Veith, M., Vences, M., Wagner, N., Canessa, S., Martel, A., Pasmans, F. (2019): Mitigating Batrachochytrium salamandrivorans in Europe. Amphib.-Reptil. 40: 265-290.