Submitted on: 2025, 8th March; revised on: 2025, 16th July; accepted on: 2025, 17th July.
Oxyrhopus in Bahia, Brazil: Pholidosis, maturity, sexual dimorphism, and reproduction
1 Departamento de Ciências Biológicas, Universidade Estadual de Santa Cruz, Rodovia Jorge Amado, km 16, CEP 45662-900- Ilhéus, Brasil
2 Red de Investigadores en Herpetología-Bolivia, La Paz, Bolivia
3 Museum Koenig Bonn (ZFMK), Leibniz Institute for the Analysis of Biodiversity Change, Adenauerallee 160, 53113 Bonn, Germany
*Corresponding author. Email: paola.d.c.1186@gmail.com
Abstract. Research on snake reproduction in the Neotropics is limited by a lack of data, necessitating further studies to better understand their reproductive biology. The genus Oxyrhopus has received increasing interest in studying its natural history, although several aspects remain unknown. This investigation aims to shed light on Oxyrhopus morphometry, maturity, and sexual dimorphism in the state of Bahia, Brazil. An analysis was conducted on 421 snake specimens from the Museum of Zoology at the State University of Santa Cruz to determine their external characteristics and reproductive conditions through gonadal inspection. Species identification was performed via pholidosis, and various measurements such as snout-vent length, tail length, head length, and width were meticulously recorded. Sexual maturity was estimated, and sexual dimorphism was assessed through statistical analysis, including regressions and seasonal comparisons, to study reproductive cycles and testicular variations in these species. The species of the genus Oxyrhopus studied included O. trigeminus, O. petolarius, O. guibei, and O. rhombifer. The investigation delineated the minimum size required for sexual maturity in each species and clarified the disparities in length between males and females. Reproductive males were found throughout the year in O. trigeminus and O. petolarius, with no significant seasonal differences in testicular volume. Females of both species exhibited annual reproductive activity. Similar patterns were observed in O. guibei and O. rhombifer, albeit with fewer data available. A relationship between female body size and fecundity was found, but further research is needed on how climatic factors may influence the reproductive cycle.
Keywords. Reproductive biology, morphometry, snakes, sexual dimorphism and maturity, testicular variations.
Index
Maturity and determination of reproductive status
Morphometry and sexual dimorphism
Analysis of reproductive conditions
Morphometry and sexual dimorphism
Male reproductive cycle and testicular variation
Fecundity and reproductive cycles of females
Given the remarkable diversity of snakes in the Neotropics, comprising over 880 species, the study of their ecology and natural history remains a field with gaps requiring further exploration (Reed and Shine, 2002; Pizzatto, 2005). However, in recent years there has been an increase in research focused on the natural history of the genus Oxyrhopus, commonly referred to as false coral snakes. These studies have addressed various aspects, including diet, distribution, and certain reproductive traits (Andrade and Silvano, 1996; Pizzatto and Marques, 2002; Alencar et al., 2012; Cabral and Scott, 2014; Coelho et al., 2019). This information on natural history and reproductive ecology is important for making appropriate comparisons between species and determining patterns (Pizzatto and Marques, 2002).
Reproductive dynamics constitute a fundamental aspect of an organism’s life history, allowing for reproduction and species continuity (Pizzatto and Marques, 2002; Pizzatto, 2005). However, crucial reproductive parameters such as sexual maturity (both minimum and average size) and sexual dimorphism can exhibit substantial variation among species and populations due to multifaceted influences, including abiotic, ecological, environmental, phylogenetic, and geographical factors (Vitt and Vangilder, 1983; Di Bernardo et al., 1998; Barron and Andraso, 2001). These factors can also affect reproductive cycles, fecundity, reproductive frequency, and reproductive potential (Di Bernardo et al., 1998; Barron and Andraso, 2001; Pizzatto et al., 2006).
In most snake species, females are longer than males (Shine, 1994; Cox et al., 2007). This sexual size dimorphism (SSD) in snakes is related to fecundity selection, favoring longer females to produce more offspring (Ford and Seigel, 1989; Shine, 2000; Cox et al., 2007). Males mature earlier and are smaller than females, likely due to different selective pressures. Enhanced chemosensory abilities are presumed to be favored by sexual selection, as they help males more effectively locate and choose mates (Madsen and Shine, 1993; Rivas and Burghardt, 2001; Shine et al., 2003). Overall, there is still much unknown about the reproductive processes of tropical snakes (Pizzatto and Marques, 2002; Pizzatto et al., 2006). Available information often comes from observations of captive snakes, sporadic encounters in the wild, or community studies (Pizzatto, 2005; Sawaya et al., 2008; Maia and Travaglia-Cardoso, 2017; Coelho et al., 2019). However, studies conducted with snakes preserved in scientific collections are still scarce, yet, these studies could provide valuable insights into the reproductive biology of tropical snakes (Pizzatto and Marques, 2002; Bizerra et al., 2005; De Aguiar and Di Bernardo, 2005). Such research could lead to a better understanding of snake reproductive biology across various regions, as well as the identification of trends and patterns in species reproduction (Pizzatto and Marques, 2002; Pizzatto, 2005).
The genus Oxyrhopus belongs to the tribe Pseudoboini, a monophyletic group composed of species that are closely related to each other. (Zaher et al., 2009; Gaiarsa et al., 2013). With 15 primarily terrestrial species distributed throughout the Neotropical region, they inhabit a wide range of habitats, from tropical forests to oceanic islands, from southern Mexico to northern Argentina (Zaher et al., 2009; Alencar et al., 2012; Gonzales et al., 2020). In the state of Bahia, Brazil, six species of Oxyrhopus have been recorded, some of which coexist in the same distribution area, with up to three or four species occurring together (Lira da Silva, 2011; Nogueira et al., 2019).
A study conducted by Pizzatto and Marques (2002) in the state of São Paulo determined that O. guibei females have longer body sizes, which gives them a considerable selective advantage, as fecundity depends on this trait. Females appear to adopt two possible reproductive strategies: producing many small eggs or only a few large ones. This trade-off aligns with classical r- and K-selection strategies in animals, where r-selected species maximize reproductive output with numerous offspring and minimal parental investment per offspring, while K-selected species invest more in fewer offspring, improving their survival probability (Pianka, 1970). Additionally, O. guibei’s reproductive cycle is continuous, producing a single clutch per year, although this can occur at any time within the annual cycle, while males exhibit spermatogenesis throughout the year. Given this information, it is reasonable to assume that other Oxyrhopus species may exhibit characteristics similar to O. guibei.
This research delves into the reproductive characteristics of Oxyrhopus species in the state of Bahia, Brazil. We examine physical traits, minimum size for sexual maturity, and attributes that potentially contribute differences between males and females. In addition, we provide information on reproductive aspects, such as to estimation of fecundity, reproductive frequency, and reproductive potential in females, and determination of the periods of reproductive activity in males, through gonadal analysis.
A total of 421 samples preserved at the Museum of Zoology of the State University of Santa Cruz (MZUESC), from various parts of the state of Bahia, were analyzed. The museum’s collection is combined with that of the Gregorio Bondar Zoological Collection, held by the Cacao Research Center (CEPLAC/CZGB). All individuals were preserved in 70% ethanol. The initial identification of species followed dichotomous keys (Peters and Orejas-Miranda, 1970; Gonzales et al., 2020) that involved determining the scale count of each snake (dorsal, ventral, subcaudal, infralabial, and supralabial), followed by recording various morphometric data for each specimen. These included snout–vent length (SVL; from the rostral scale to the cloaca), Total length (TL; from rostral scale to the tip of the tail) and tail length (LC; from the cloaca to the tip of the tail, using only complete tails to avoid underestimation), which were measured using a measuring tape. Head length (HL; from the rostral scale to the quadrate bone), head height (HH; the highest part of the head) and head width (HW; measured at the level of the quadrate bones) were measured using a digital caliper with an accuracy of 0.01 millimeters (Boretto and Ibargüengoytía, 2006). The capture times of the individuals were determined based on museum records from 1990 to 2019, which did not include exact dates, so the data were classified into the wet season (October to May) or the dry season (June to September). The dataset supporting this study is available at Zenodo: https://doi.org/10.5281/zenodo.14861556.
Maturity and determination of reproductive status
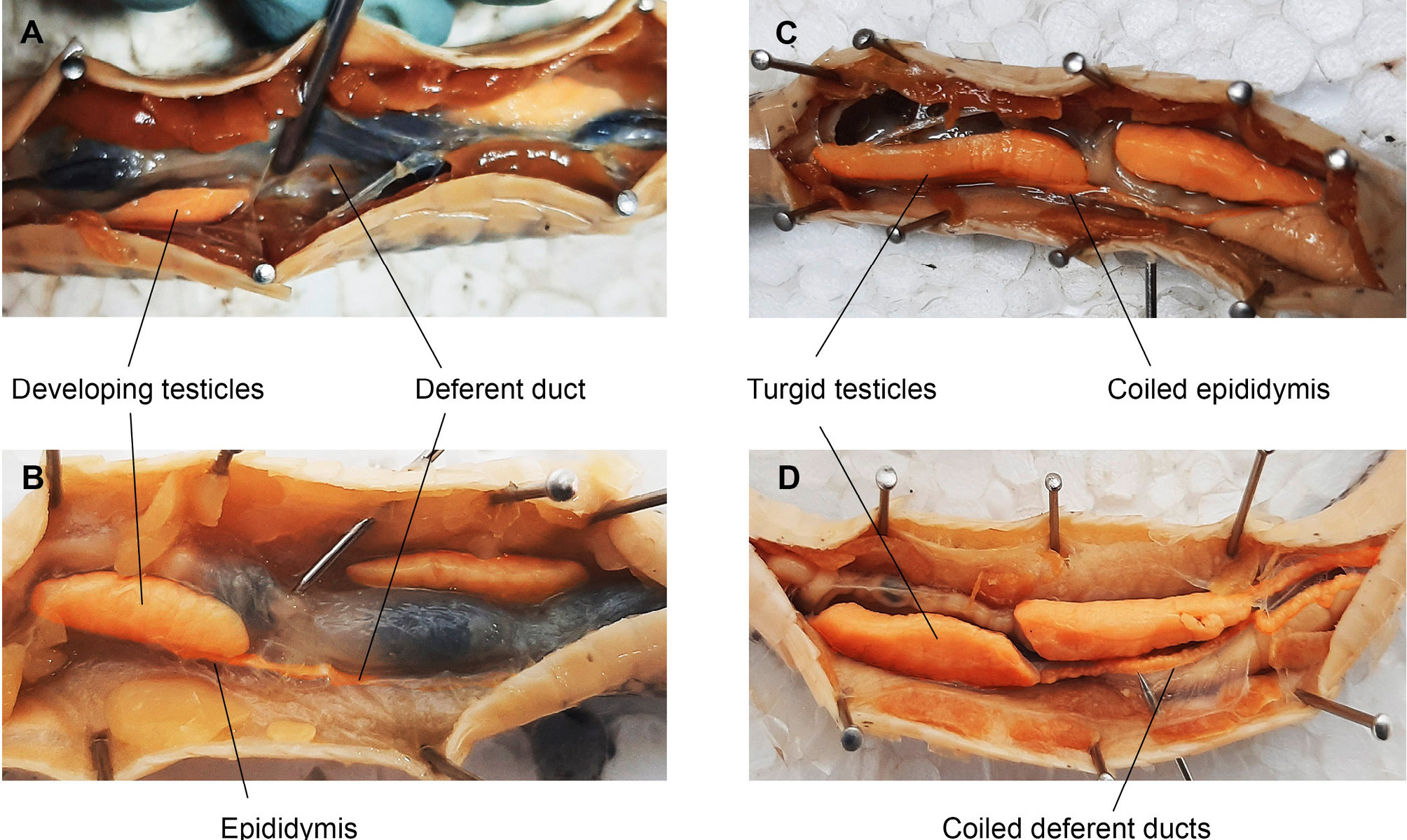
The sex of each individual was determined by examining the gonads, following the methodology described by Alencar et al. (2012), which involved making an incision in the midsection of the ventral scales to determine the presence of testes or ovaries. The reproductive conditions and sexual maturity of each species were determined through gonadal inspection. For females, uterine characterization was categorized into two groups: juvenile uterus, characterized by the absence of folds or folds that do not extend throughout the uterus; and adult uterus, characterized by folds extending throughout the uterus, indicative of distended oviducts, generalized enlargement of the oviducts associated with vitellogenic recrudescence, or markedly stretched oviducts resembling a post-reproductive uterus, indicating recent parturition (Boretto and Ibargüengoytía, 2006). Additionally, females with oviductal eggs or vitellogenic follicles ≥10 mm were also considered adult and reproductive (De Aguiar and Di Bernardo, 2005; Pizzatto, 2005; Boretto and Ibargüengoytía, 2006; Coelho et al., 2019; Fig. 1).

Males were considered adult and reproductively active if they had turgid and opaque testes, and if they presented coiled epididymides and deferent ducts, indicative of the presence of spermatozoa typical of adults (Pizzatto and Marques, 2002; Pinto and Fernandes, 2004; De Aguiar and Di Bernardo, 2005; López et al., 2009). The minimum maturity size was estimated as the size of the smallest reproductive individual for each sex (Alencar et al., 2012), taking the minimum snout-vent length (SVL) at sexual maturity for each species.
Morphometry and sexual dimorphism
Comparisons regarding general characteristics of age and sex among the studied species were conducted. Before performing any statistical analyses, the collected data were subjected to normal distribution and homogeneity of variances assessments using the Shapiro-Wilk test and Levene’s test, respectively. A significance threshold of P < 0.05 was used to determine significant results and all statistical analyses were performed using R software (version 4.3.1; R Core Team, 2024).
To evaluate sexual dimorphism, two methods were used. i) Quantifying the degree of sexual size dimorphism (SSD): this involved calculating the SSD index by dividing the mean snout-vent length (SVL) of the longer sex by that of the smaller sex, then subtracting 1 from the result. This index was considered arbitrarily positive when females were the longer sex and negative when males were longer (Gibbons and Lovich, 1990; Shine, 1994). This index was compared with other studies from Brazil. ii) Two-factor analysis of variance (ANOVA): this method was employed with sex and species as factors, and number of ventral scales, number of subcaudal scales, SVL, and tail length (LC) as dependent variables (Pinto and Fernandes, 2004; Pizzatto, 2005). Juveniles were excluded from the morphometric study based on SVL. Additionally, a covariance analysis (ANCOVA) was performed for tail length (LC), head length (HL), and head width (HW), with SVL as the covariate. Finally, as an exploratory analysis, we conducted a principal component analysis (PCA), separated by sex for each species.
Analysis of reproductive conditions
Measurements of the gonads were taken with a digital caliper with an accuracy of 0.01 millimeters, recording the length and width of the testes, as well as the length of vitellogenic follicles and eggs (Pizzatto, 2005; Bellini, 2013). To calculate the testicular volume for both the right and left testes, reflecting reproductive activity, the ellipsoid formula (4/3π abc) was used, where a = length/2, b = width/2, and c = height/2 (Pizzatto and Marques, 2002; Pizzatto, 2005). A simple regression was performed between testicle size and the snout-vent length (SVL) of all males. Male reproductive cycles were studied by comparing testicular variation and deferent duct condition between seasons, only in reproductively active adult males, using SVL as a covariate through an ANCOVA (Bellini, 2013).
To estimate clutch size or female fecundity, the number of oviductal eggs was used. A simple regression was performed between the SVL of adult females and clutch size, as well as another simple regression between clutch size and the size of the largest oviductal egg (Pizzatto, 2005; López et al., 2009). In cases where fecundity could not be determined with oviductal eggs, data from follicles larger than 10 mm were used (Almeida-Santos et al., 2014). Reproductive frequency was estimated by the percentage of reproductively active mature females in the sample (with vitellogenic follicles >10 mm or eggs), where a result below 50% indicates a biannual or multiannual reproductive cycle, and more than 50% indicates an annual cycle, this analysis was conducted per year, and the final frequency was averaged across years with sufficient data (Pizzatto, 2005; Bellini et al., 2013 2018). Reproductive potential was calculated, showing the potential number of neonates per female per year, following the formula: (mean clutch size) × (reproductive frequency) (Bellini, 2013).
Morphometric data of 217 Oxyrhopus trigeminus individuals (148 Adults, 69 Juvenils), 142 O. petolarius (86 A, 56 J), 35 O. guibei individuals (20 A, 15 J), 18 O. rhombifer individuals (13 A, 5 J), seven O. formosus (4 A, 3 J), and two juvenils of O. clathratus were obtained. The latter two species were excluded from the statistical analyses due to the limited amount of data, and their information is presented for comparison purposes (Table 1). We determined the minimum size (SVL) at which each species reaches sexual maturity. For O. trigeminus, females mature at 43.4 cm and males at 31.8 cm; for O. petolarius, females mature at 65.6 cm and males at 42.1 cm; for O. guibei, females mature at 44 cm and males at 44.3 cm; for O. rhombifer, females mature at 49.8 cm and males at 37.7 cm; and for O. formosus, females mature at 70.8 cm and males at 63.3 cm.
| Specie | Sex | Age | n | V | SC | TL | SVL | LC | % LC | HL | % HL | HW |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Oxyrhopus clathratus | ♀ | J | 1 | 189 | 60 | 29.6 | 24.8 | 4.8 | 16.21 | 11.1 | 4.5 | 4.44 |
| ♂ | J | 1 | 184 | 74 | 32.4 | 26 | 6.4 | 19.75 | - | - | - | |
| Oxyrhopus formosus | ♀ | A | 2 |
199 (197-203) |
69 (66-75) |
(70.8-90.5) | (56.5-74) | (56.5-74) | (18.23-20.20) | 21.35 | 2.89 | 12.56 |
| J | 2 | (33.4-48.8) | (27-40.1) | (6.4-8.7) | (17.83-19.16) | (10.61-14.7) | (3.67-3.93) | (6.55-7.79) | ||||
| ♂ | A | 2 |
190 (189-191) |
86 (83-87) |
(63.3-64.7) | (48.6-51) | (13.7-14.7) | (21.17-23.22) | (17.49-17.56) | (3.44-3.6) | (8.47-8.95) | |
| J | 1 | 53.3 | 41.2 | 12.1 | 22.7 | 14.24 | 3.46 | 8.23 | ||||
| Oxyhropus guibei | ♀ | A | 9 |
196 (192-212) |
75 (70-82) |
85.64 (54.9-109.6) |
69.27 (44-93.6) |
16.42 (10.9-20.2) |
19.89 (19.13-21.45) |
22.84 (19.98-26.67) |
3.17 (2.85-3.47) |
12.61 (8.92-18.73) |
| SD 15.954 | SD 14.082 | SD 2.719 | SD 0.762 | SD 2.501 | SD 0.209 | SD 3.198 | ||||||
| J | 8 |
46.96 (20.3-64) |
38.1 (16.4-51.8) |
9.52 (3.9-12.3) |
20.13 (19.06-21.59) |
14.61 (9.92-18.24) |
4.16 (3.51-6.05) |
7.32 (5.29-9.66) |
||||
| SD 14.374 | SD 11.517 | SD 3.226 | SD 0.943 | SD 2.834 | SD 0.893 | SD 1.387 | ||||||
| ♂ | A | 11 |
181 (172-185) |
82 (80-85) |
67.01 (57.5-75.3) |
51.62 (44.3-58.6) |
15.38 (12.9-18) |
22.94 (20.98-24.41) |
17.56 (9.36-20.91) |
3.42 (1.65-3.78) |
9.36 (8.31-10.39) |
|
| SD 7.071 | SD 5.399 | SD 1.838 | SD 1.034 | SD 3.108 | SD 0.603 | SD 0.668 | ||||||
| J | 7 |
35.6 (23.9-41.1) |
27.84 (18.9-31.8) |
7.76 (5-9.3) |
21.74 (20.92-22.63) |
12.12 (9.74-13.91) |
4.51 (4.16-5.17) |
6.15 (4.75-6.92) |
||||
| SD 6.396 | SD 4.916 | SD 1.494 | SD 0.595 | SD 1.464 | SD 0.437 | SD 0.766 | ||||||
| Oxyrhopus petolarius | ♀ | A | 42 |
204 (199-215) |
90 (81-97) |
102.08 (85.2-127.5) |
80.93 (65.6-100.8) |
21.92 (18.6-27.5) |
21.46 (20.2-23.51) |
25.83 (20.97-35-49) |
3.17 (2.66-4.22) |
12.4 (9.66-16.23) |
| SD 11.461 | SD 9.556 | SD 2.53 | SD 0.877 | SD 3.081 | SD 0.237 | SD 1.449 | ||||||
| J | 33 |
48.9 (24.5-77.5) |
38.27 (18.7-61.1) |
10.64 (5.6-16.5) |
21.78 (20-23.98) |
14.94 (11.3-21.95) |
4.19 (3.57-6.15) |
7.63 (6-10.46) |
||||
| SD 14.075 | SD 11.026 | SD 3.081 | SD 0.913 | SD 2.343 | SD 0.697 | SD 1.352 | ||||||
| ♂ | A | 44 |
196 (191-200) |
100 (95-107) |
82.72 (55.2-107.3) |
62.73 (42.1-82.2) |
20.1 (13.1-26.1) |
24.72 (23.14-26.96) |
21.08 (12.27-26.4) |
3.35 (1.98-4.18) |
10.46 (7.49-13.11) |
|
| SD 13.066 | SD 9.872 | SD 3.610 | SD 0.983 | SD 2.892 | SD 0.325 | SD 1.192 | ||||||
| J | 23 |
40.26 (25.3-56.4) |
30.77 (19.3-42.6) |
9.67 (6-13.8) |
23.88 (22.67-25.66) |
13.17 (10.73-16.89) |
4.42 (3.82-5.56) |
6.92 (5.28-8.37) |
||||
| SD 7.863 | SD 5.965 | SD 1.989 | SD 0.758 | SD 1.489 | SD 0.531 | SD 0.792 | ||||||
| Oxyhropus rhombifer | ♀ | A | 10 |
210 (204-217) |
77 (70-85) |
75.53 (59.8-87.8) |
60.39 (49.8-70.1) |
16 (12.4-17.7) |
20.71 (19.5-22.24) |
19.54 (18.1-21.91) |
3.21 (3-3.63) |
9.9 (8.82-12.38) |
| SD 8.837 | SD6.229 | SD 1.572 | SD 0.891 | SD 1.32 | SD 0.194 | SD 1.507 | ||||||
| J | 3 |
65 (51.3-75.2) |
50.93 (40.1-59.5) |
14.1 (11.2-15.7) |
21.68 (20.88-22.34) |
14.97 (12.38-17.56) |
3.19 (3.09-3.3) |
8.42 (8.35-8.49) |
||||
| SD 12.328 | SD 9.897 | SD 2.491 | SD 0.687 | SD 3.663 | SD 0.148 | SD 0.099 | ||||||
| ♂ | A | 3 |
198 (194-200) |
86 (82-90) |
58.35 (48.9-67.8) |
44.55 (37.7-51.4) |
13.8 (11.2-16.4) |
23.55 (22.9-24.19) |
17.11 (14.16-20.06) |
3.83 (3.76-3.9) |
9.2 (8.81-9.59) |
|
| SD 13.36 | SD 6.22 | SD 3.677 | SD 0.908 | SD 4.172 | SD 0.099 | SD 1.25 | ||||||
| J | 2 |
34.4 (32.7-36.1) |
27 (25.7-28.3) |
7.4 (7-7.8) |
21.51 (21.41-21.61) |
12.53 (12.04-13.02) |
4.6 (4.6-4.68) |
6.85 (6.74-6.96) |
||||
| SD 20.404 | SD 1.838 | SD 0.566 | SD 0.141 | SD 0.693 | SD 0.057 | SD 0.156 | ||||||
| Oxyrhopus trigeminus | ♀ | A | 85 |
199 (191-211) |
61 (54-70) |
78.07 (53.4-103.7) |
65.26 (43.4-86.9) |
12.88 (9.1-17.4) |
16.5 (14-19.7) |
22.4 (16.47-27.71) |
3.46 (2.85-3.95) |
11.09 (7.9-13.77) |
| SD 11.58 | SD 9.97 | SD 1.77 | SD0.96 | SD 2.51 | SD 0.23 | SD 1.55 | ||||||
| J | 38 |
39.12 (19.8-68.5) |
32.59 (18-59.4) |
6.61 (3.3-9.8) |
17.07 (14.04-20.42) |
13.9 (10.31-20.36) |
4.5 (3.43-5.95) |
7.27 (4.75-10.82) |
||||
| SD 11.62 | SD 9.82 | SD 1.74 | SD 1.36 | SD 2.62 | SD 0.67 | SD 1.45 | ||||||
| ♂ | A | 63 |
187 (173-197) |
69 (61-81) |
59.31 (39.2-77.7) |
47.28 (31.8-63.2) |
12.18 (6.9-15.9) |
20.14 (17.2-24.6) |
18.28 (13.78-23.16) |
3.98 (3.26-5.63) |
9.34 (6.98-12.71) |
|
| SD 8.24 | SD 6.53 | SD 1.87 | SD 1.33 | SD 1.99 | SD 0.37 | SD 1.3 | ||||||
| J | 31 |
33.46 (18.9-50.3) |
27.16 (15.7-39.7) |
6.85 (4-10.6) |
19.45 (17.62-21.58) |
12.49 (9.63-17.18) |
4.8 (3.85-6.38) |
6.46 (4.25-8.2) |
||||
| SD 8.34 | SD 6.53 | SD 1.69 | SD 1.11 | SD 1.84 | SD 0.62 | SD 1.09 |
Morphometry and sexual dimorphism
The SSD was calculated for five species, including O. formosus, despite the limited data. Across all species, females consistently displayed longer sizes than males, with the indices closely to each other (Table 2), a trend that is statistically supported for the species in our study. We observed that adult females of all species were significantly longer (SVL) than adult males (Fsex = 37.002; P < 0.001), and there was a significant difference in body size among species (Fspecies = 46.885; P < 0.001), with O. petolarius exhibiting the largest body size. However, the interaction between sex and species did not show a significant difference (Finteraction = 0.035; P = 0.998), indicating that males of one species could be as large as females of another species.
| Species | n | SVL mean (cm) | SSD | Location | Reference | ||
|---|---|---|---|---|---|---|---|
| Female | Male | Female | Male | ||||
| O. formosus | 2 | 2 | 65.25 | 49.8 | 0.31 | Bahia/Br | This study |
| O. guibei | 9 | 11 | 69.27 | 51.62 | 0.34 | Bahia/Br | This study |
| “ | 15 | 15 | 73.28 | 49.79 | 0.47 | São Paulo/Br | Rocha-Barbosa et al., 2000 |
| “ | 9 | 9 | 65.61 | 53.94 | 0.22 | Minas Gerais/Br | Vaz, 1999 |
| “ | 211 | 160 | 81.9 | 59.9 | 0.37 | São Paulo/Br | Pizzatto and Marques, 2002 |
| O. petolarius | 42 | 44 | 80.93 | 62.73 | 0.29 | Bahia/Br | This study |
| “ | 6 | 12 | 97.0 | 70.3 | 0.41 | Minas Gerais/Br | Mendonça et al., 2024 |
| O. rhombifer | 10 | 2 | 60.39 | 44.55 | 0.36 | Bahia/Br | This study |
| O. trigeminus | 85 | 63 | 65.26 | 47.28 | 0.38 | Bahia/Br | This study |
| “ | 3 | 4 | 67.17 | 54.25 | 0.24 | Minas Gerais/Br | Vaz 1999 |
| “ | 16 | 13 | 71.77 | 55.34 | 0.30 | Minas Gerais/Br | Alencar et al., 2012 |
| “ | 42 | 25 | 46.71 | 38.94 | 0.20 | Pernambuco/Br | Coelho et al., 2019 |
Through comparisons of the number of ventral scales, significant differences were evident between species (Fspecies = 63.326; P < 0.001), notably with O. rhombifer exhibiting the highest number of ventrals among the species examined. Furthermore, in examining sexes, a significant distinction emerged (Fsexes = 54.077; P < 0.001), with females possessing more ventral scales than males. The interaction between species and sexes also yielded a significant difference (Finteraction = 2.454; P = 0.033).
Similarly, the analysis of subcaudal scales revealed a comparable trend, with O. petolarius exhibiting the highest count of these scales compared to other species (Fspecies = 959.592; P < 0.001). Additionally, males across all studied species displayed a greater number of subcaudal scales than females (Fsexes = 82.045; P < 0.001; Finteraction = 3.617; P = 0.003).
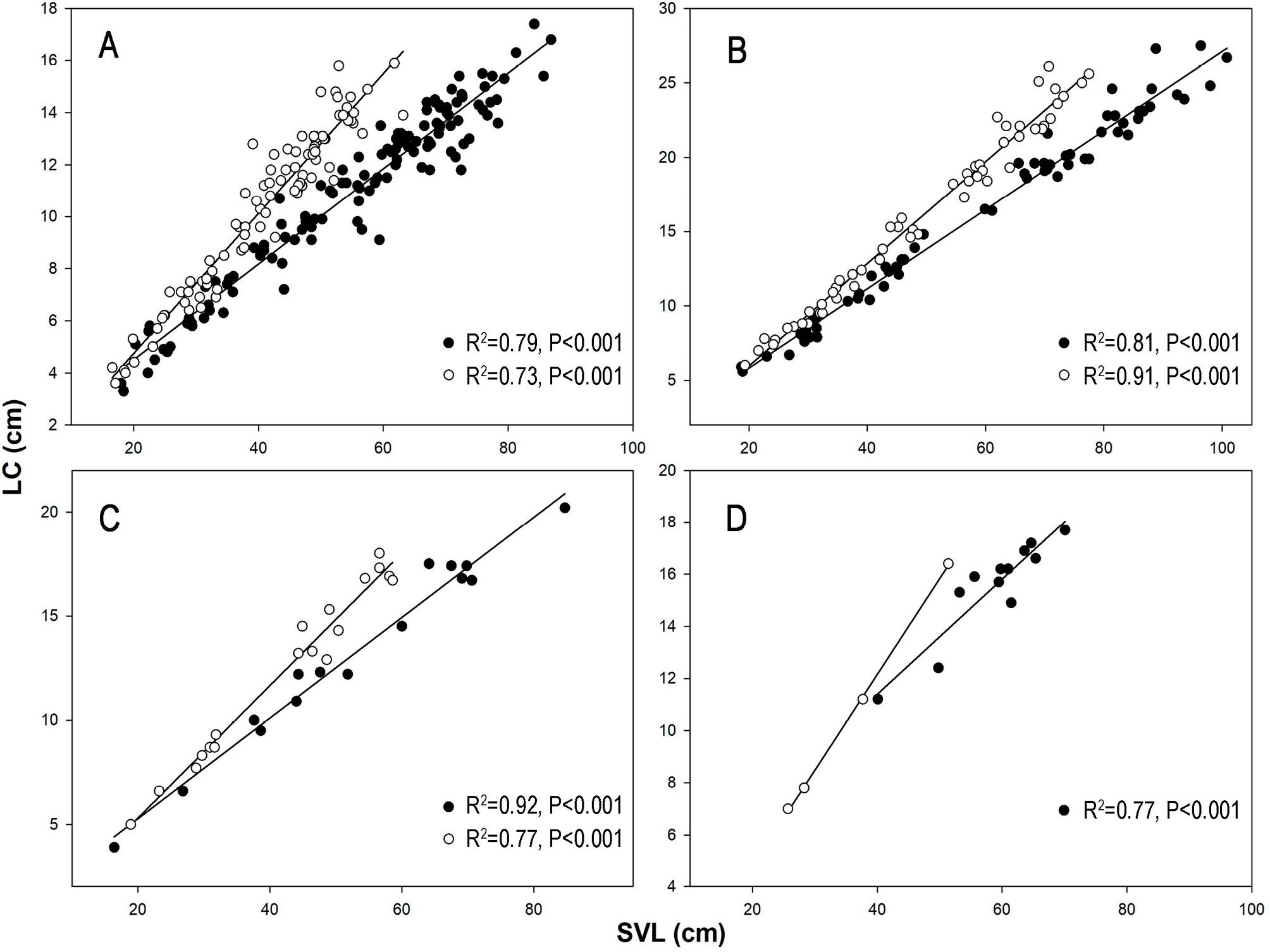
The relative tail length (LC) in males across all species surpassed that of females (Fsex = 7.558; P = 0.006; Fspecies = 212.570; P < 0.001), with O. petolarius exhibiting the largest tail size. However, the interaction between sex and species did not yield a significant difference (Finteraction = 0.962; P = 0.411) indicating that tail length variation is influenced more by interspecific body size differences than by sex alone. As a result, females of larger species may have tails as long as, males of smaller species. Notably, significant differences were observed in O. trigeminus, where adult males’ tails averaged 20.14% of the total body length compared to 16.5% in females. Linear regression analysis between tail length and SVL depicted positive relationships, indicating males tended to have longer tails than females (Fig. 3A).
Similarly, O. petolarius displayed differences in tail size between sexes, with males’ tails accounting for 24.72% and females’ tails for 21.46% of total length. The regressions depicted positive relationships, clearly illustrating the difference between the sexes (Fig. 3B). Likewise, O. guibei exhibited distinct percentages of tail length relative to total body size, with males at 22.94% and females at 19.89%. The regressions for both males and females showed positive relationships, highlighting their differences (Fig. 3C). Although limited by sample size, regression analysis for O. rhombifer showed differences between sexes in the regressions (Fig. 3D), with males’ tails occupying 23.55% of their total length compared to 20.71% in females. Conversely, head length (HL) and head width (HW) did not show significant differences among species (F(HL) = 2.274; P = 0.132; F(WH) = 1.495; P = 0.222).
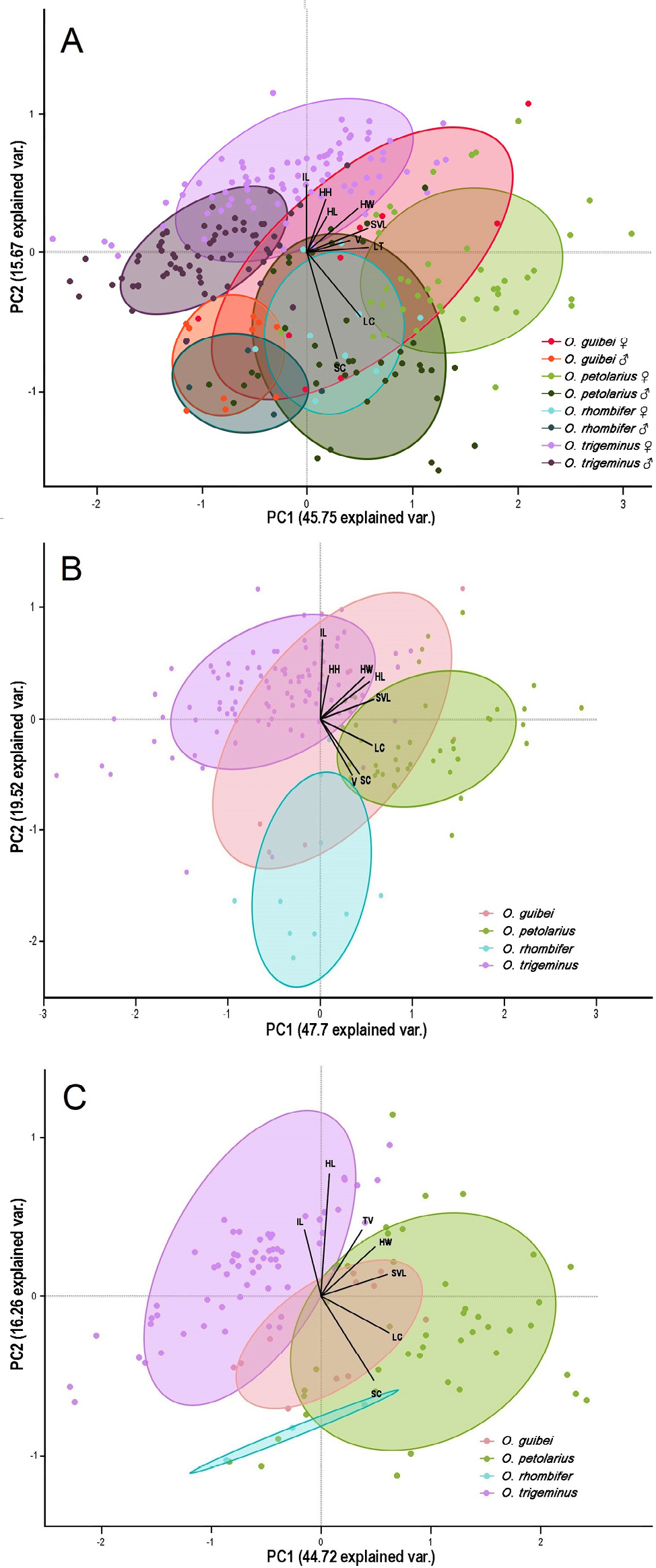
The PCA analysis revealed partial morphological differentiation among the four Oxyrhopus species (Fig. 4). All species showed considerable overlap, particularly along the first principal component. When males and females were plotted together (Fig. 4A), partial separation between sexes was observed within some species, most notably in O. petolarius and O. trigeminus, suggesting the presence of sexual dimorphism. These trends were further explored in separate PCAs for females and males (Fig. 4B and 4C), where similar variable loadings were observed, though slight differences in group distribution and vector orientation reinforce sex-based morphological patterns. Overall, the patterns suggest moderate interspecific differentiation and variable degrees of sexual dimorphism depending on the species.
| NFR | VF (mm) | Eggs (mm) | Fecundity | RF | RP | |
|---|---|---|---|---|---|---|
| O. trigeminus | 44 |
14.43 (10.2-19.4; n=43) |
26.99 (22.01-37.88; n=11) |
9.61 (5-13 E) |
0.55 | 5.28 |
| O. petolarius | 29 |
13.85 (10.16-16.55; n=7) |
25.9 (18.21-31.89; n=22) |
8.58 (4-15 E) |
0.66 | 5.72 |
| O. guibei | 6 |
15.02 (12.6 -17.36; n=4) |
22.46-23.07; n=2 |
6.83 (3-11 FV) |
0.66 | 4.55 |
| O. rhombifer | 3 | 12.9 -16.05; n=2 | 17.3; n=1 |
5.33 (3-8 FV) |
0.3 | 1.59 |
For the females of O. trigeminus, a positive correlation was observed between clutch size and SVL (Fig. 6A), as well as a negative relationship between clutch size and egg size (Fig. 6B). For O. petolarius, statistically significant positive correlations were observed between clutch size and snout-vent length (Fig. 6C), and between clutch size and oviductal egg size (Fig. 6D). Similarly, in O. guibei, positive relationships were determined between clutch size with snout-vent length and vitellogenic follicles (Fig. 6E and F), Although none of the relationships were statistically significant, it is important to highlight that fecundity in O. guibei was estimated based on the number of vitellogenic follicles, unlike the other species, for which oviductal eggs were used. This methodological difference should be considered when comparing reproductive parameters across species. For O. rhombifer, regression analyses were not performed due to the limited amount of data.

Our results align with previous studies on Oxyrhopus species and other snake groups, highlighting consistent patterns of sexual dimorphism (SVL, LC, ventrals, and subcaudals) (Shine, 1994; Pizzatto and Marques, 2002; Cox et al., 2007; Alencar et al., 2012; Coelho et al., 2019). This pattern has been observed across various regions of Brazil, including O. trigeminus (Alencar et al., 2012; Coelho et al., 2019), O. guibei (Pizzatto and Marques, 2002; Pires de Toledo, 2020) and O. melanogenys (Pires de Toledo, 2020). Notably, males of all studied Oxyrhopus species exhibited longer tails than females, potentially to accommodate the hemipenis and associated muscles (Cox et al., 2007). Conversely, the longer SVL of females likely relates to reproductive aspects, providing space for larger or more numerous eggs (Rivas and Burghardt, 2001; Cox et al., 2007; Alencar et al., 2012).
Snakes can exhibit territorial behavior and male combat (Rivas and Burghardt, 2001, Senter et al., 2014). However, this behavior has been recorded in only a few Dipsadidae species and none from the Pseudoboini tribe. Following Pizzato et al. (2002) and considering the reported SSD values in Oxyrhopus, it is unlikely that false coral snakes exhibit this behavior, reducing selective pressure for longer males. Our findings support this, showing that males reach sexual maturity at smaller sizes than females, and could be allocating more energy towards reproduction than growth. This contrasts with females, which attain maturity at longer sizes, allowing for extended growth before reproduction.
In several snake species, it has been observed that males mature at a smaller size than females (Shine, 2000). This pattern was also confirmed in the genus Oxyrhopus by the study of Pizzatto and Marques (2002) with O. guibei, where they detected that the minimum size for adult males was 38.8 cm SVL, while for females, it was 63.2 cm, using data from the state of Sao Paulo. When comparing these findings with the results of our study, we observed a similar pattern in O. petolarius and O. trigeminus. Even with the limited amount of data for O. rhombifer, we also found this difference in maturation sizes. However, only O. guibei seems not to present such a marked difference, as both males and females had a minimum adult size with an SVL of approximately 44 cm. This apparent discrepancy could be due to the limited amount of data analyzed for this species (only 20 records), and it is possible that males maturing at even smaller sizes, as observed in the study of Pizzatto and Marques (2002) or even lower, may be found. Nevertheless, in all studied species, females exhibited longer maximum sizes than males.
In adult males, spermatogenesis appears to occur year-round in all the species we studied, which is consistent with previous studies conducted on O. guibei (Pizzatto and Marques, 2002). However, defining the male reproductive cycle is more complex than in females, as it cannot be directly assessed through gonad observation but rather through the analysis of testicular volume (Bellini, 2013) in addition histological analysis of seminiferous tubules and spermatozoa presence, which allow more precise determination of reproductive activity (Mendonça et al., 2024). In the present study, we focused on macroscopic indicators, such as testicular enlargement and the presence of coiled deferent ducts, both of which have been associated with sexual maturity and active spermatogenesis (Pizzatto and Marques, 2002; López et al., 2009; Almeida-Santos et al., 2014). It is also worth noting that the use of preserved specimens may introduce certain biases, as dehydration or compression can alter gonad size, potentially affecting assessments of reproductive seasonality (Almeida-Santos et al., 2014). Therefore, evaluating the condition of the deferent ducts and/or epididymis, particularly whether they are coiled, serves as an important complementary indicator of sperm presence. While the absence of histological data may limit more detailed interpretations, the external and internal morphological traits used here offer reliable evidence of reproductive condition in males.
Given that the production of male gametes is less energetically costly than the production of female gametes, it is expected that males in tropical areas have continuous reproductive cycles and remain active throughout the year (Pizzatto, 2005). Additionally, for the Oxyrhopus species studied here, it makes sense that males are active year-round, given that females exhibit a continuous reproductive cycle, which could stimulate males to produce gametes constantly.
The longer body size of females compared to males provides a selective advantage, as fecundity is correlated with body size. In other words, longer females have the capacity to produce a greater number of eggs or longer offspring (Shine, 2000). In this study, although we did not measure egg volume or width, and therefore cannot infer offspring size or shape, we observed that larger females tended to have either more eggs or eggs with greater length. According to previous studies, O. guibei exhibits two possible reproductive strategies (similar to r and K strategies): the production of many small eggs or few large eggs (Pizzatto and Marques, 2002). Although these strategies were not clearly distinguishable in our data, the variation observed in egg length and clutch size suggests that female body size may influence reproductive investment. Nonetheless, future studies incorporating egg volume and hatchling data would allow for a more detailed understanding of reproductive strategies in these species.
In the case of O. trigeminus, O. petolarius, and O. guibei, positive relationships were found between the size of the females and the number of eggs they could produce. However, only O. trigeminus showed a negative relationship between the number of eggs and their size, suggesting a reduction in egg size as their number increases, as one reproductive strategy proposes. On the other hand, both O. petolarius and O. guibei did not show this negative relationship; instead, they exhibited a positive relationship, suggesting that in addition to having a greater number of eggs due to their longer size, these eggs also tend to be longer, which could result in longer offspring. However, it is important to note that this has its limits, as the eggs cannot occupy more space than is available within the body of the snakes. Therefore, we might think that these two species are balancing the two strategies proposed by Pizzatto and Marques (2002), having the largest possible number of eggs without compromising their size, thus ensuring a higher number of offspring with better chances of survival by being longer.
Fecundity is a crucial aspect in the study of snake reproduction (Almeida-Santos et al., 2014), commonly assessed by counting eggs in the oviducts of females. However, some studies also consider the number of vitellogenic follicles (Bizerra et al., 2005; Pizzatto, 2005, López et al., 2009; Bellini, 2013; Braz et al., 2014; Coelho et al., 2019), which can lead to overestimation, and should be specified clearly specified the method used to avoid it (Almeida-Santos et al., 2014; Braz et al., 2014). Our data on the fecundity of O. trigeminus and O. petolarius were based on egg counts, showing similar fecundities. However, for O. guibei, fecundity was estimated based on vitellogenic follicles rather than oviductal eggs due to the limited number of females with oviductal eggs available. It was observed that the fecundity of O. guibei was slightly lower than that of the other species. This suggests that considering only follicles larger than 10 mm could provide an accurate estimate of the actual fecundity for this species.
The study of fecundity in Oxyrhopus has been conducted in different regions of Brazil, with variable results. For example, a fecundity of 7.8 eggs has been determined for O. trigeminus in the state of Pernambuco (Coelho et al., 2019), and 7.5 in the state of Minas Gerais (Alencar et al., 2012). In the case of O. guibei, a fecundity of 10.9 was estimated in the state of Sao Paulo (Pizzatto and Marques, 2002). Comparing our data with these studies, we observed that O. trigeminus showed higher fecundity, while O. guibei exhibited a lower fecundity than reported. However, fecundity can vary within the same species depending on its distribution area and be related to climate, food availability, or even behavioral characteristics (Pizzatto et al., 2006). Some authors suggest that pregnant snakes may refrain from feeding (Pizzatto and Marques, 2002; Shine, 2003; Bellini, 2013); however, we observed pregnant snakes with stomach contents, which could affect both the available body space to develop the eggs and the level of energy that snakes can invest in reproduction, leading to variations in species fecundity.
The reproductive cycle of many Neotropical snakes is continuous, implying that they have the capacity to reproduce at any time of the year, although this does not necessarily mean they have multiple clutches (Shine, 2000; Pizzatto and Marques, 2002). It has been observed that oviparous species generally present annual cycles, or at least shorter ones than viviparous species, and longer females tend to reproduce more frequently. However, if females reach excessively large sizes, they may cease to reproduce as frequently due to senescence (Pizzatto et al., 2006). This pattern has been documented in species such as Boiruna maculata, Clelia pumblea, and O. guibei (Pizzatto, 2005), all belonging to the monophyletic tribe Pseudoboini. Therefore, it is reasonable to expect that other members of this group, including species of the genus Oxyrhopus, exhibit a similar reproductive system. Our data confirm that O. trigeminus, O. petolarius, and O. guibei display this type of reproduction, as we found eggs during both the wet and dry seasons. Furthermore, the reproductive cycle of these species appears to be annual, as expected. In contrast, O. rhombifer might exhibit a biannial reproductive cycle; however, due to the limited data available, this cannot be confirmed with certainty. It would be necessary to investigate whether females that reproduce during the dry season in one year also do so in the following year’s dry season, or if they might instead reproduce during the subsequent wet season. This could indicate if their reproductive cycle is shorter than 12 months. Since reproductive cycles can be affected by climate, even if the same species exhibits a continuous cycle in warm areas, it may show seasonal patterns in colder regions (Pinto and Fernandes, 2004). It would be interesting to study the effect of climatic parameters on the reproduction of this genus, considering its wide distribution in the American continent.
We express our gratitude to FAPESB (Fundação de Amparo à Pesquisa do Estado da Bahia) for providing the scholarship to PD. Our gratitude to Antônio Argolo from the Museum of Zoology at the State University of Santa Cruz for generously providing access to the Oxyrhopus specimens under their care.
Alencar, L.R. V., Galdino, C.A.B., Nascimento, L.B. (2012): Life history aspects of Oxyrhopus trigeminus (Serpentes: Dipsadidae) from two sites in Southeastern Brazil. J. Herpetol. 46: 9-13.
Almeida-Santos, S.M., Braz, H.B., Santos, L.C., Sueiro, L.R., Barros, V.A., Rojas, C.A., Kasperoviczus, K.N. (2014): Biologia reprodutiva de serpentes: recomendações para a coleta e análise de dados. Herpetologia Brasileira 3: 14-24.
Andrade, R. de O., Silvano, R.A.M. (1996): Comportamento alimentar e dieta da “falsa-coral” Oxyrhopus guibei Hoge & Romano (Serpentes, Colubridae). Rev. Bras. Zool. 13: 143-150.
Barron, J.N., Andraso, G.M. (2001): The influence of fall foraging success on follicle number in the northern water snake, Nerodia sipedon. J. Herpetol. 35: 504-507.
Bellini, G.P. (2013): Ecología comparada de serpientes ovíparas y vivíparas en el litoral fluvial de la Argentina. Doctoral dissertation. Universidad Nacional del Litoral.
Bellini, G.P., Arzamendia, V., Giraudo, A.R. (2013): Ecology of Thamnodynastes hypoconia in subtropical-temperate South America. Herpetologica. 69: 67-79.
Bellini, G.P., Arzamendia, V., Giraudo, A.R. (2018): Reproductive life history of snakes in temperate regions: What are the differences between oviparous and viviparous species? Amphib.-Reptil. 40: 291-303.
Bizerra, A., Marques, O.A.V., Sazima, I. (2005): Reproduction and feeding of the colubrid snake Tomodon dorsatus from south-eastern Brazil. Amphib.-Reptil. 26: 33-38.
Boretto, J.M., Ibargüengoytía, N.R. (2006): Asynchronous spermatogenesis and biennial female cycle of the viviparous lizard Phymaturus antofagastensis (Liolaemidae): Reproductive responses to high altitudes and temperate climate of Catamarca, Argentina. Amphib.-Reptil. 27: 25-36.
Braz, H.B., Kasperoviczus, K.N., Almeida-Santos, S.M. (2014): Reproductive ecology and diet of the fossorial snake Phalotris lativittatus in the Brazilian Cerrado. Herpetol. J. 24: 49-57.
Cabral, H., Scott, N.J. (2014): Oxyrhopus petolarius (Linnaeus, 1758) (Serpentes, Dipsadidae): Distribution extension and new departmental record for Paraguay. Check List 10: 1207-1209.
Coelho, R.D., Sales, R.F., Ribeiro, L.B. (2019): Sexual dimorphism, diet, and notes on reproduction in Oxyrhopus trigeminus (Serpentes: Colubridae) in the semiarid Caatinga of northeastern Brazil. Phyllomedusa 18: 89-96.
Cox, R.M., Butler, M.A., John-Alder, H.B. (2007): The evolution of sexual size dimorphism in reptiles. In: Sex, size and gender roles: evolutionary studies of sexual size dimorphism, pp. 38-49. Fairbairn, D., Blanckenhorn, W., Székely, T., Eds, Oxford, Oxford Academic Press.
De Aguiar, L.F.S., Di Bernardo, M. (2005): Reproduction of the water snake Helicops infrataeniatus (Colubridae) in southern Brazil. Amphib.-Reptil. 26: 527-533.
Di Bernardo, M., Martins, M.B., Pontes, G.M., Hilleshein, R. (1998): Crescimento e maturidade sexual em Liophis jaegeri, Philodryas patagoniensis e Xenodon neuwiedii (Serpentes, Colubridae) no nordeste elevado do RS, Brasil. Available from: https://lume.ufrgs.br/handle/10183/100494
Ford, N.B., Seigel, R.A. (1989): Relationships among body size, clutch size, and egg size in three species of oviparous snakes. Herpetologica 45: 75-83.
Gaiarsa, M.P., de Alencar, L.R.V., Martins, M. (2013): Natural history of pseudoboine snakes. Pap. Avulsos Zool. 53: 261-283.
Gibbons, J.W., Lovich, J.E. (1990): Sexual dimorphism in turtles with emphasis on the slider turtle (Trachemys scripta). Herpetol. Monogr. 4: 1-29.
Gonzales, L., Reichle, S., Entiauspe-Neto, O.M. (2020): A new species of Oxyrhopus Wagler, 1830 (Serpentes: Dipsadidae) from the Bolivian Andes. An. Acad. Bras. Ciênc. 92: e20191428.
Lira da Silva, R.M. (2011): Reptiles of Bahia, Brazil - List of species. Núcleo Regional de Ofiologia de Animais Peçonhentos, Departamento of Zoology, Biology Institute, Federal University of Bahia. Available from: http://www.noap.ufba.br/biotabahia.
López, S.M., Giraudo, A.R., Arzamendia, V., Chiaraviglio, M. (2009): Biología reproductiva de la serpiente semiacuática Liophis semiaureus (Serpentes, Colubridae) en el nordeste de Argentina. Rev. Chil. Hist. Nat. 82: 233-244.
Madsen, T., Shine, R. (1993): Costs of reproduction in a population of European adders. Oecologia 94: 488-495.
Maia, D., Travaglia-Cardoso, S.R. (2017): Oxyrhopus clathratus (Serpentes: Dipsadidae): Reproduction and cannibalism. Oecol. Aust. 21: 197-200.
Mendonça, L.R., Coelho Dias, M.A.P., Lucas, D.F., Da Paz e Figueiredo, Caio. V. Pinto da Matta, S.L., Neves Feio, R. (2024): Insights on the reproductive biology of Oxyrhopus petolarius (Linnaeus, 1758) (Dipsadidae: Pseudoboini) from Southeastern Brazil. Cuad. Herpetol. 38: 51-59.
Nogueira, C.C., Argôlo, A.J.S., Arzamendia, V., Azevedo, J.A., Barbo, F.E., Bérnils, R.S., Bolochio, B.E., Borges-Martins, M., Brasil-Godinho, M., Braz, H., Buononato, M.A., Cisneros-Heredia, D.F., Colli, G.R., Costa, H.C., Franco, F.L., Giraudo, A., Gonzalez, R.C., Guedes, T., Hoogmoed, M.S., Marques, O.A.V., Montingelli, G.G., Passos, P., Prudente, A.L.C., Rivas, G.A., Sanchez, P.M., Serrano, F.C., Silva, N.J., Strüssmann, C., Vieira-Alencar, J.P.S., Zaher, H., Sawaya, R.J., Martins, M. (2019): Atlas of Brazilian snakes: Verified point-locality maps to mitigate the Wallacean shortfall in a megadiverse snake fauna. S. Am. J. Herpet. 14: S1-S274.
Peters, J.A., Orejas-Miranda, B. (1970): Catalogue of the Neotropical Squamata. Part I. Snakes. Bulletin 297: 1-347.
Pianka, E.R. (1970): On r- and K-Selection. Am. Nat. 104: 592-597.
Pinto, R.R., Fernandes, R. (2004): Reproductive biology and diet of Liophis poecilogyrus poecilogyrus (Serpentes, Colubridae) from southeastern Brazil. Phyllomedusa 3: 9-14.
Pires de Toledo, D.G. (2020): Revisão taxonômica do grupo de tríades pertencente ao gênero Oxyrhopus (Serpentes: Pseudoboini). Master’s dissertation. Universidade de São Paulo.
Pizzatto, L. (2005): Body size, reproductive biology and abundance of the rare pseudoboini snakes genera Clelia and Boiruna (Serpentes, Colubridae) in Brazil. Phyllomedusa 4: 111-122.
Pizzatto, L., Marques, O.A. V. (2002): Reproductive biology of the false coral snake Oxyrhopus guibei (Colubridae) from southeastern Brazil. Amphib.-Reptil. 23: 495-504.
Pizzatto, L., Almeida-santos, S.M. De, Marques, O.A.V. (2006): Biologia reprodutiva de serpentes brasileiras. In: Herpetologia no Brasil II, pp. 201-221. Nascimento, L.B., Oliveira, M.E., Eds, Sociedade Brasileira de Herpetologia.
Reed, R.N., Shine, R. (2002): Lying in wait for extinction: Ecological correlates of conservation status among Australian elapid snakes. Conserv. Biol. 16: 451-461.
Rivas, J.A., Burghardt, G.M. (2001): Understanding sexual size dimorphism in snakes: Wearing the snake’s shoes. Anim. Behav. 62: F1-F6.
Sawaya, R.J., Marques, O.A.V., Martins, M. (2008): Composição e história natural das serpentes de Cerrado de Itirapina, São Paulo, sudeste do Brasil. Biota Neotrop. 8: 127-149.
Senter, P., Harris, S.M., Kent, D.L. (2014): Phylogeny of courtship and male-male combat behavior in snakes. PLOS ONE 9: e107528.
Shine, R. (1994): Sexual size dimorphism in snakes revisited. Copeia 1994: 326-346.
Shine, R. (2000): Vertebral numbers in male and female snakes: The roles of natural, sexual and fecundity selection. J. Evol. Biol. 13: 455-465.
Shine, R. (2003): Reproductive strategies in snakes. P. Roy. Soc. B-Biol. Sci. 270: 995-1004.
Vitt, L.J., Vangilder, L.D. (1983): Ecology of a snake community in norteastern Brazil. Amphib.-Reptil. 4: 273-296.
Shine, R., Phillips, B., Waye, H., LeMaster, M., Mason, R.T. (2003): Chemosensory cues allow courting male garter snakes to assess body length and body condition of potential mates. Behav. Ecol. Sociobiol. 54: 162-166.
Zaher, H., Grazziotin, F.G., Cadle, J.E., Murphy, R.W., de Moura-leite, J.C., Bonatto, S.L. (2009): Molecular phylogeny of advanced snakes (Serpentes, Caenophidia) with an emphasis on South American Xenodontines: A revised classification and descriptions of new taxa. Pap. Avulsos Zool. 49: 115-153.