Submitted on: 2024, 30th September; Revised on: 2024, 28th November; Accepted on: 2024, 3rd December
On the maxillary dentition of the Western whip snake, Hierophis viridiflavus (Lacépède, 1789): heterodonty is not opisthoglyphy
1 Istituto Zooprofilattico Sperimentale del Piemonte, Liguria e Valle d’Aosta, Via Bologna 148, 10154 Turin, Italy
2 Faculty of Veterinary Medicine, Department of Pathobiology, Pharmacology and Zoological Medicine, Wildlife Health Ghent, Ghent University, 9820 Merelbeke, Belgium
3 Dipartimento di Scienze della Terra, Università degli Studi di Torino, via Valperga Caluso 35, 10125 Turin, Italy
4 Institut Català de Paleontologia Miquel Crusafont (ICP-CERCA), Edifici ICTA-ICP, c/ Columnes s/n, Campus de la UAB, 08193 Cerdanyola del Vallès, Barcelona, Spain
*Corresponding author. Email: matteoriccardo.dinicola@izsplv.it
Abstract. The Western whip snake (Hierophis viridiflavus) is a colubroid snake typically considered aglyphous and non-venomous, although this is currently a subject of debate. Indeed, the maxillary dentition of this species has recently been described as opisthoglyphous due to the presence of enlarged rear maxillary teeth, reportedly characterised by prominent grooves and ridges. Aiming to test the actual presence of these structures and provide a clearer characterisation of the maxillary dentition of the species, we analysed 17 dry-prepared and disarticulated H. viridiflavus specimens using both scanning electron and optical microscopy. The imaging confirmed the presence of posterior, enlarged maxillary teeth displaying a distal carina, highlighted by shallow longitudinal inflections on the disto-labial and disto-lingual tooth surfaces. Nonetheless, the analysis revealed the consistent absence of the meso-labially placed groove typical of opisthoglyphous taxa. Based on the results of our study, the maxillary dentition of the Western whip snake is heterodont, specifically opisthomegadont, and is confirmed to be aglyphous.
Keywords. Aglyphous, Colubrid, non-front-fanged snakes, opisthoglyphous, proteroglyphous, rear-fanged, solenoglyphous, tooth morphology.
Snakes exhibit a wide variety of dental morphologies and adaptations that are crucial for their survival and feeding strategies. Broadly speaking, snakes are carnivorous reptiles, and their teeth are variably long, slender, and slightly curved, making them ideal for piercing and, in most cases, holding onto prey. Snake teeth are attached to the jaws in a modified pleurodont manner, where each tooth is set within a shallow depression (Lillywhite, 2014; Pough et al., 2016).
In members of the clade Alethinophidia Nopcsa, 1923, encompassing all snakes other than blind snakes (Scolecophidia Cope, 1864), teeth are present on the maxillae, palatines, and pterygoids in the upper jaw, and on the dentary in the lower jaw. The premaxilla bears teeth in some basal species (Lee and Scanlon, 2002; Mahler and Kearney, 2006; Pough et al., 2016; Berkovitz and Shellis, 2023).
Teeth replacement in snakes is a continuous process throughout their lives, characterised by a unique mechanism where functional teeth are replaced by new ones developing in successive waves along the jaw. Unlike most other amniotes, snakes lack external resorption pits during this process; instead, tooth replacement involves internal resorption within the pulp cavity. This adaptation allows snakes to maintain a constant supply of functional teeth, essential for their feeding efficiency and survival, as damaged or lost teeth are replaced rapidly. This mechanism supports their ability to ingest large or resistant prey and minimises the risk of collateral damage to neighbouring teeth during replacement (Lillywhite, 2014; LeBlanc et al., 2023).
Heterodonty, the presence of differently shaped teeth, is evident in many species. For example, venomous snakes (all belonging to the clade Caenophidia Hoffstetter, 1939) tend to have heterodont dentitions, characterised by the presence of differently shaped teeth. Specifically, these snakes typically exhibit enlarged, specialised teeth (i.e., fangs) with remarkable morphological features (e.g., grooves, hollow centres, ridges along the fang channels) that aid in venom delivery (Triep et al., 2013; Broeckhoven and du Plessis, 2017; Avella et al., 2021). In contrast, nonvenomous snakes tend to homodonty (i.e., all teeth present the same shape), although variations in tooth size and morphology are common (Young and Kardong, 1996; Pough et al., 2016; Berkovitz and Shellis, 2023). In any case, the differences in the shape and function of the teeth within each snake species, even when minimal, make it overly simplistic to distinguish solely between heterodonty and homodonty (see Ryerson and Valkenburg, 2021; Segall et al., 2023). Indeed, Westeen et al. (2020) recently highlighted the presence of a wide range of dentition phenotypes in snakes, particularly in rear-fanged ones, consistent with adaptation to different, specialised diets and prey capture modes. Therefore, for many snake species indicated as homodonts, it would be more accurate to describe them as having low degrees of heterodonty rather than true homodonty.
The diversity of maxillary dentition phenotypes in colubroid snakes is traditionally categorised into four main types: i) aglyphous, ii) opisthoglyphous, iii) proteroglyphous, and iv) solenoglyphous (e.g., Boulenger, 1893, 1894, 1896; Pough et al., 2016; Gower and Zaher, 2022; Delfino and Villa, 2024).
These terms are commonly used to reflect the following characteristics on an etymological basis (e.g., Merriam-Webster, 2024; Treccani, 2024): aglyphous (Greek: ‘a-’ for ‘without’ + ‘gluphé’ for ‘carving’ or ‘engraving’) snakes lack any grooved fangs; opisthoglyphous (Greek: ‘ópisthen-’ for ‘behind’ + ‘gluphé’) refers to snakes with grooved fangs positioned at the rear of the maxilla; proteroglyphous (Greek: ‘próteros-’ for ‘first’ or ‘earliest’ + ‘gluphé’) indicates snakes with grooved hollow, generally non-movable fangs at the front of the maxilla; solenoglyphous (Greek: ‘solén-’ for ‘pipe’ or ‘tube’ + ‘gluphé’) denotes snakes with non-grooved hollow, retractable, independently movable fangs at the front of the maxilla (for differences in fang canaliculation between solenoglyphous and proteroglyphous snakes, refer to Broeckhoven and du Plessis, 2017). A further term, endoglyphous (Greek: ‘éndon-’ for ‘within’ or ‘inside’ + ‘gluphé’), is rarely used for lumping together the proteroglyphous and solenoglyphous conditions in order to underline the presence of fangs with an inner cavity (see Golay et al., 1993; Pin, 2009).
This classification clearly has limitations, as not all snake species possess dentition that perfectly and exclusively matches one of these categories. For instance, non-front-fanged snakes (NFFS), exhibit different dental types, also in terms of tooth surface (see Young and Kardong, 1996). According to Weinstein et al. (2022), the term ‘non-front-fanged’ is preferred over ‘opisthoglyphous’ because the broad range of modifications in the maxillary teeth of these snakes resists simple classification under traditional terms.
A number of snake species possess enlarged, but not grooved rear maxillary teeth (e.g., Natricidae Bonaparte, 1838 and some Colubridae Oppel, 1811 snakes; see Edmund, 1969; Berkovitz and Shellis, 2017, 2023; Weesten et al., 2020). For some of them, the term ‘opisthoglyphous’ has sometimes been imprecisely used solely with reference to the larger size of the posterior teeth (as for Natricidae in Westeen et al., 2020: fig. 1).
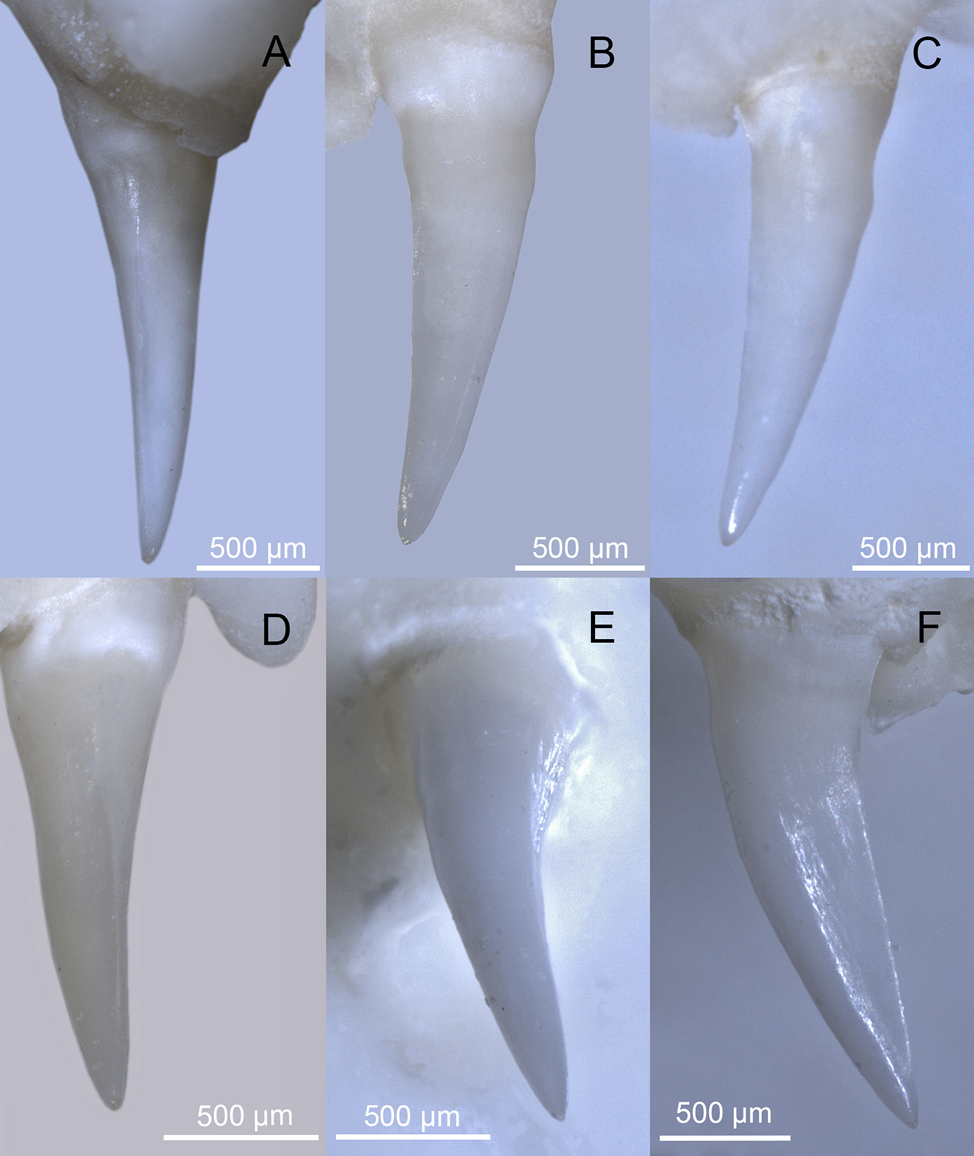
The dentition of species with enlarged rear maxillary teeth, regardless of whether the tooth surface is grooved or not, should be referred to as opisthomegadont (sensu Edmund, 1969). The distinction between grooved and ungrooved teeth may not always be unambiguous (see Young and Kardong, 1996), and thus it is challenging to have categorising terms for each intermediate condition. In agreement with Berkovitz and Shellis (2023), who equate opisthoglyphy with the ‘Type 3’ categorisation by Young and Kardong (1996), it is thus reasonable to use the term ‘opisthoglyphous’ to identify taxa that possess enlarged rear maxillary teeth with a well-defined groove on the meso-labial side, such as Boiga Fitzinger, 1826, Dispholidus Duvernoy, 1832, Erythrolamprus Boie, 1826, Galvarinus Trevine et al., 2022, Malpolon Fitzinger, 1826, and Telescopus Wagler, 1830 (for the morphology of these teeth, see: Fig. 1; Young and Kardong, 1996; Broeckhoven and du Plessis, 2017; Modahl and Mackessy, 2019; Sánchez et al., 2019; Herrera et al., 2022; Weinstein et al., 2022).

Regardless of instances where ‘opisthoglyphous’ is used broadly as a synonym for ‘non-front fanged’ or ‘rear fanged’ (e.g., Westeen et al., 2020; Gower and Zaher, 2022), a recent manuscript has defined the Western whip snake as opisthoglyphous in the strict sense, indicating that its enlarged posterior maxillary teeth are characterised by the presence of ‘prominent grooves and ridges’ (Paterna, 2023: p. 126).
Using both optical microscopy and Scanning Electron Microscopy (SEM), we investigated whether the enlarged rear maxillary teeth of the Western whip snake are indeed characterised by pronounced grooves and ridges, and aimed to clarify the classification of the maxillary dentition of the species.
The Western whip snake, Hierophis viridiflavus (Lacépède, 1789), is a colubroid snake with an average total length between 110 and 150 cm (Vanni and Zuffi, 2011; Di Nicola et al., 2021a). Its range extends across south-central Europe, from northeastern Spain to coastal Croatia, and includes the entire Italian Peninsula, as well as Sardinia, Sicily, and numerous smaller islands and islets (Avella et al., 2017; Di Nicola et al., 2021b). Currently, two subspecies are recognised: Hierophis viridiflavus viridiflavus (Lacépède, 1789), in the western part of the species range, typically characterised by a pattern of black and yellow dots and stripes; and Hierophis viridiflavus carbonarius (Bonaparte, 1833), in the eastern part, often predominantly or completely melanistic (Di Nicola et al., 2021a, b; Senczuk et al., 2021; Sindaco and Razzetti, 2021; Storniolo et al., 2023).
The species is highly defensive and prone to biting when cornered/handled (Bea, 1998; Di Nicola et al., 2021a; Avella et al., 2024). Early research identified the presence of Duvernoy’s glands in H. viridiflavus (Phisalix, 1922; Taub, 1967). Additionally, some bite reports have described the onset of symptoms in humans possibly indicating envenomation (e.g., Bédry et al., 1998; Dutto et al., 2015; Avella et al., 2024), although the clinical interpretations of some of them is questionable (see Weinstein et al., 2022; Avella et al., 2024; Paolino et al., 2024). There is a lack of recent research on the oral glands of the Western whip snake, as well as proteomic studies aimed at identifying the components of its saliva and oral secretions, which could provide valuable insights into the biochemical potential of H. viridiflavus and its classification as ‘non-venomous’ (Avella et al., 2024).
The dentition of the Western whip snake, though sometimes described as homodont (e.g., Dutto et al., 2015), features two enlarged teeth at the rear of each maxilla (Fig. 2), which are less curved compared to other maxillary teeth (Racca et al., 2020; Avella et al., 2024). In light of this, the species dentition is clearly heterodont.
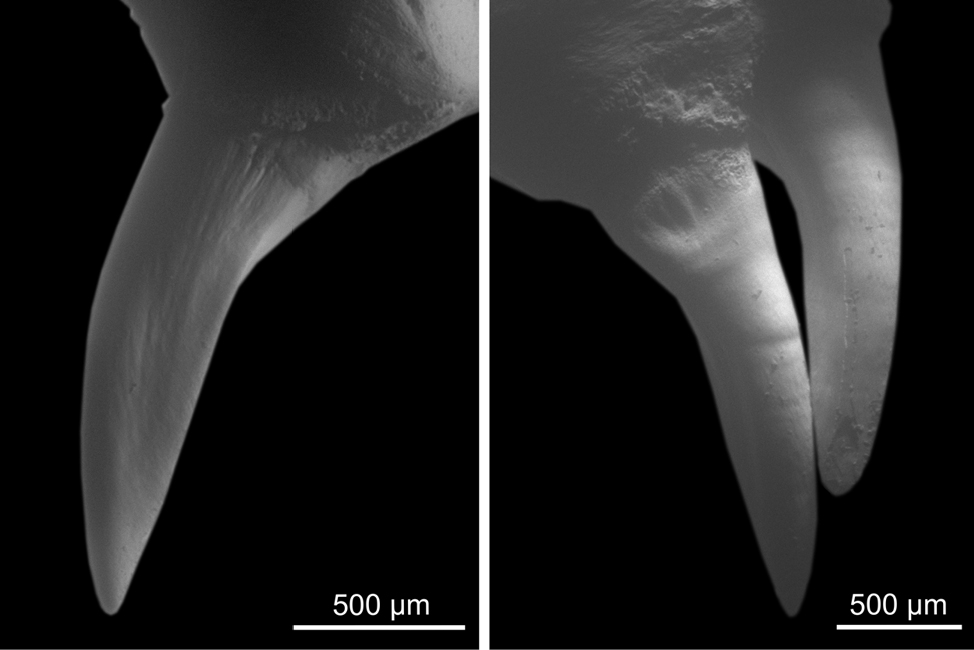
Focusing on the morphology of the enlarged rear teeth, we observed the maxillary dentition of 17 disarticulated, dry prepared skeletons of morphologically mature and immature Western whip snakes from both the subspecies H. v. carbonarius and H. v. viridiflavus (MDHC 9, 34, 69, 74, 80, 118, 198, 199, 219, 265, 298, 306, 328, 442, 458, 460, 502; Collection acronym: MGPUT MDHC - Massimo Delfino Herpetological Collection, Museo di Geologia e Paleontologia, Dipartimento di Scienze della Terra, Università degli Studi di Torino). The sample included both females and males.
For optical microscopy, a Leica M205C stereomicroscope equipped with a Leica DMC 2900 camera was used. SEM observations were performed with a Tescan Essence microscope. The samples were not coated with a conductive material to preserve their original surface characteristics; for this reason, we operated in Single Vac conditions using a Back Scattered Electron Detector. The system was configured with an accelerating voltage of 15 keV and a beam current of 30 nA.
In all analysed specimens, the last two maxillary teeth are different in size and morphology from those preceding the diastema. These rear teeth are characterised by a distal carina, which is bordered on both sides by a shallow depression extending from the distal region to the disto-labial and disto-lingual surfaces. These are inflections of the surface and do not constitute an actual groove (see Figs 2-3). It can be excluded the presence of any meso-labial (antero-lateral) grooves comparable to those of snake traditionally considered opisthoglyphous (e.g., genera Malpolon and Telescopus; compare Figs 2-3 with Fig. 1).
Given the considerable diversity in snake dentition, the term ‘non-front-fanged snakes’ is currently to be preferred over more common yet overgeneralising terms like ‘rear-fanged’ and ‘aglyphous’ when referring to snakes lacking anterior fangs. For instance, NFFS commonly exhibit more or less pronounced heterodonty. In this case, if the posterior maxillary teeth are larger, it is appropriate to describe the heterodont dentition as opisthomegadont. This condition may involve enlarged maxillary teeth that are grooved to varying extents, or not at all, potentially in combination with other superficial morphological features.
Based on our results, the dentition of the Western whip snake is heterodont, opisthomegadont. While the rear maxillary teeth display a complex morphology including sulci and carinae, these features are weakly expressed and can be variably perceived, for instance depending on the light orientation under microscopy. From our perspective, these characteristics do not correspond to the deep and very-well-marked anterolateral groove characteristic of teeth from snakes traditionally classified as opisthoglyphous in the strict etymological sense. Nevertheless, we agree with Weinstein et al. (2022) that the dichotomy between aglyphous and opisthoglyphous dentition fails to adequately capture the extensive morphological diversity observed in NFFS, and that each case should be analysed individually.
It is worth noting that a recent paper, published after the submission of our manuscript, suggests that even Dolichophis caspius (Gmelin, 1789), along with several other taxa, is opisthoglyphous (Paterna and Grano, 2024). However, our direct inspection of D. caspius specimens MDHC 518 and 519 confirmed that the dentition of this species is clearly aglyphous, with the morphology of the posterior maxillary teeth closely resembling that described here for H. viridiflavus. Through the application of optical and scanning electron microscopy we were able to determine the absence of prominent grooves in the posterior, enlarged maxillary teeth of H. viridiflavus. In light of this, we do not support classifying this species as opisthoglyphous in the morphological sense. Further investigations, employing tools such as micro-CT scanners, will enable a more detailed analysis of the tooth morphology of this and other NFFS species.
The authors wish to thank Simona Cavagna and Marco Pavia for their assistance with the scanning electron microscopy and optical imaging at the Department of Earth Sciences, University of Turin, and Ignazio Avella and Marco Sassoè for their comments, which have improved our manuscript.
Massimo Delfino is supported by Fondi di Ricerca Locale UNITO (2021-2023). This is publication number 383 of the Museo di Geologia e Paleontologia collections at the Università degli Studi di Torino.
Avella, I., Barajas-Ledesma, E., Casewell, N.R., Harrison, R.A., Rowley, P.D., Crittenden, E., Wüster, W., Castiglia, R., Holland, C., Van Der Meijden, A. (2021): Unexpected lack of specialisation in the flow properties of spitting cobra venom. J. Exp. Biol. 224: jeb229229.
Avella, I., Castiglia, R., Senczuk, G. (2017): Who are you? The genetic identity of some insular populations of Hierophis viridiflavus s.l. from the Tyrrhenian Sea. Acta Herpetol. 12: 209-214.
Avella, I., Savini, F., Di Nicola, M.R. (2024): A prolonged bite by a Western Whip Snake, Hierophis viridiflavus carbonarius (Bonaparte 1833) (Serpentes, Colubridae), resulting in pronounced local oedema. RandA 31: e21448.
Bea, A. (1998): Coluber viridiflavus Lacépède, 1789. In: Reptiles, Fauna Ibérica, Vol. 10, pp. 358-363. Salvador, A., Ramos, M.A., Eds, Madrid, Museo Nacional de Ciencias Naturales.
Bédry, R., Hilbert, G., Goyffon, M., Laffort, P., Parrens, E. (1998): Is the saliva of the European whip snake (Coluber viridiflavus) neurotoxic? Toxicon 12: 1729-1730.
Berkovitz, B.K.B., Shellis, R.P. (2017): The teeth of non-mammalian vertebrates - 1st ed. London, Academic press.
Berkovitz, B.K.B., Shellis, R.P. (2023): The teeth of non-mammalian vertebrates: form, function, development and growth. Amsterdam, Academic Press.
Boie, H. (1826): Merkmale einiger japanischer Lurche. Isis von Oken 2: 203-216.
Bonaparte, C.L.J.L. (1833): Iconographia della fauna Italica per le quattro classi degli animali vertebrati. Roma, Salviucci.
Bonaparte, C.L.J.L. (1838): Amphibiorum tabula analytica. Nuovi Ann. Sci. Nat. 1: 391-393.
Boulenger, G.A. (1893): Catalogue of the snakes in the British Museum (Natural History). Vol. I. London, British Museum (Natural History).
Boulenger, G.A. (1894): Catalogue of the snakes in the British Museum (Natural History). Vol. II. London, British Museum (Natural History).
Boulenger, G.A. (1896): Catalogue of the snakes in the British Museum (Natural History). Vol. III. London, British Museum (Natural History).
Broeckhoven, C., Du Plessis, A. (2017): Has snake fang evolution lost its bite? New insights from a structural mechanics viewpoint. Biol. Lett. 13: 20170293.
Cope, E.D. (1864): On the characters of the higher groups of Reptilia Squamata: And especially of the Diploglossa. Proc. Acad. Nat. Sci. Philadelphia 16: 224-231.
Delfino M., Villa A. (2024). An overview of the skeleton of reptiles. In: Handbook of Zoology, Reptilia, Volume 1: General Biology, Archosauria, Chelonia, pp. 39-56. Joger, U., Ed., Berlin/Boston, Walter de Gruyter GmbH.
Di Nicola, M.R., Cavigioli, L., Luiselli, L., Andreone, F. (2021a): Anfibi & Rettili d’Italia. Edizione aggiornata. Latina, Edizioni Belvedere.
Di Nicola, M.R., Di Rosa, D., Mariacher, A., Zabbia, T. (2021b): Hierophis viridiflavus carbonarius (Western whip snake). Coloration. Herpetol. Rev. 52: 873-874.
Dutto, M., Ineich, I., Serre-Collet, F., Goyffon, M., Bédry, R. (2015): Trois cas de morsures du Colubridé Hierophis viridiflavus (Lacépède, 1789). Bull. Soc. Herp. Fr. 156: 55-62.
Duvernoy, D.M. (1832): Memoire sur le caracteres tires de l’anatomie pour distinquer les serpens venimeux des serpens non venimeux. Ann. Sci. Nat., 26: 113-160.
Edmund, A.G. (1969): Dentition. In: Biology of the Reptilia, pp. 117-200. Vol. 1. Gans, C., Parsons, T.S., Bellairs, A.d’A., Eds, London, Academic Press.
Fitzinger, L.J.F.J. (1826): Neue Classification der Reptilien nach ihren natürlichen Verwandtschaften: nebst einer Verwandtschafts-Tafel und einem Verzeichnisse der Reptilien-Sammlung des K. K. Zoologischen Museum zu Wien. Wien, J.G. Heubner.
Fleischmann, F.L. (1831): Dalmatiae nova serpentum genera. Erlangae, C. Heyderi.
Geoffroy Saint-Hilaire, I. (1827): Description des reptiles qui se trouvent en Égypte. In: Description de l’Egypte ou recueil des observations et des recherches qui ont été faites en Égypte pendant l’expedition de l’Armée française, publié par les ordres de sa Majesté-L’Empereur Napoléon le Grand. Histoire Naturelle. Tome premier. Première partie, pp. 121-160. Savigny, M.J.C.L., Ed., Paris, Imprimerie Impériale.
Gmelin, J.F. (1789): Caroli a Linné, Systema Naturae per Regna Tria Naturae, Secundum Classes, Ordines, Genera, Species; cum Characteribus, Differentiis, Synonymis, Locis. Editio Decima Tertia, Aucta, Reformata. Vol. I, part 3. Leipzig, G.E. Beer.
Golay, P., Smith, H. M., Broadley, D. G., Dixon, J. R., McCarthy, C., Rage, J. C., Schätti B., Toriba, M. (1993). Endoglyphs and other major venomous snakes of the world. A checklist. Geneva, Azemiops herpetological data center.
Gower, D.J., Zaher, H. (2022): The origin and early evolutionary history of snakes. Cambridge, Cambridge University Press.
Herrera, Y., Fuentes-Retamal, S., Kemmerling, U., Peichoto, M.E., Ortiz, J.C., Urra, F.A. (2022): Shedding light on the dentition and venom delivery system of the rear-fanged snake, Galvarinus chilensis chilensis (Serpentes: Dipsadidae: Tachymenini) from Chile. Biology 11: 1788.
Hoffstetter, R. (1939): Contribution a l’étude des Elapidae actuels et fossiles et de l’ostéologie des ophidians. Arch. Mus. Hist. Nat. Lyon 15: 1-78.
Lacepède, B.G.É.d.L. (1789): Histoire naturelle des quadrupèdes ovipares et des serpens, vol. 2. Paris, Hôtel de Thou.
LeBlanc, A.R.H., Palci, A., Anthwal, N., Tucker A.S., Araujo R., Pereira M.F.C., Caldwell M.W. (2023): A conserved tooth resorption mechanism in modern and fossil snakes. Nat. Commun. 14: 742
Lee, M.S., Scanlon, J.D. (2002): Snake phylogeny based on osteology, soft anatomy and ecology. Biol. Rev. 77: 333-401.
Lillywhite, H.B. (2014): How snakes work: structure, function and behavior of the world’s snakes. Oxford, Oxford University Press.
Mahler, D.L., Kearney, M.R. (2006): The palatal dentition in squamate reptiles: Morphology, development, attachment, and replacement. Fieldiana Zool. 2006: 1-61.
Merriam-Webster (2024): Merriam-Webster.com dictionary. Retrieved September 1, 2024, from https://www.merriam-webster.com/.
Modahl, C.M., Mackessy, S.P. (2019): Venoms of rear-fanged snakes: New proteins and novel activities. Front. Ecol. Evol. 7: 279.
Nopcsa, F. (1923): Die Familien der Reptilien. Fortschritte der Geologie und Paläontologie, vol. 2. Berlin, Verlag Gebrüder Borntraeger.
Oppel, M. (1811): Die Ordnungen, Familien und Gattungen der Reptilien als Prodrom einer Naturgeschichte derselben. Munich, J. Lindauer.
Paolino, G., Di Nicola, M. R., Ballouard, J. M., Bonnet, X., Damm, M., Le Roux, G., Lüddecke, T., Marini, D., Weinstein, S. A., Avella, I. (2024). A review of bites by non-front-fanged snakes (NFFS) of Europe. Toxicon 250: 108116.
Paterna, A. (2023): The role of modified teeth in the function of prolonged bites in Hierophis viridiflavus (Serpentes: Colubridae). Phyllomedusa 22: 121-130.
Paterna, A., Grano, M. (2024). Morphology of the maxillary bones in the Caspian whip-snake Dolichophis caspius (Serpentes Colubridae) supports the opisthoglyphous model within western palearctic whipsnakes. Biodivers. J. 15: 693-700.
Phisalix, M. (1922): Animaux venimeux et venins: la fonction venimeuse chez tous les animaux; les appareils venimeux; les venins et leurs propriétés; les fonctions et usages des venins; l’envenimation et son traitement. Paris, Masson & cie.
Pin, T.C. (2009): The study of the mediation of Ohanin, a king cobra (Ophiophagus hannah) toxin through the central nervous system. Unpublished MSc thesis. National University of Singapore.
Pough, F.H., Andrews, R.M., Crump, L.C., Savitzky, A.H., Wells, K.D., Brandley, M.C. (2016): Herpetology. Sunderland, Sinauer associates Inc.
Racca, L., Villa, A., Wencker, L.C.M., Camaiti, M., Blain, H., Delfino, M. (2020): Skull osteology and osteological phylogeny of the Western whip snake Hierophis viridiflavus (Squamata, Colubridae). J. Morphol. 281: 808-833.
Ryerson, W.G., Van Valkenburg, T. (2021): Linking tooth shape to strike mechanics in the Boa constrictor. Integr. Comp. Biol. 61: 759-771.
Sánchez, M.N., Teibler, G.P., Sciani, J.M., Casafús, M.G., Maruñak, S.L., Mackessy, S.P., Peichoto, M.E. (2019): Unveiling toxicological aspects of venom from the Aesculapian false coral snake Erythrolamprus aesculapii. Toxicon 164: 71-81.
Segall, M., Houssin, C., Delapré, A., Cornette, R., Herrel, A., Milgram, J., Shahar, R., Dumont, M. (2023): Armed to the teeth: The underestimated diversity in tooth shape in snakes and its relation to feeding behavior and diet. Ecol. Evol. 13: e10011.
Senczuk, G., Gramolini, L., Avella, I., Mori, E., Menchetti, M., Aloise, G., Castiglia, R. (2021): No association between candidate genes for color determination and color phenotype in Hierophis viridiflavus, and characterization of a contact zone. J. Zool. Syst. Evol. Res. 59: 748-759.
Sindaco, R., Razzetti, E. (2021): An updated check-list of Italian amphibians and reptiles. Nat. Hist. Sci. 8: 35-46.
Storniolo, F., Mangiacotti, M., Zuffi, M.A.L., Scali, S., Sacchi, R. (2023): Large scale phenotypic characterisation of Hierophis viridiflavus (Squamata: Serpentes): climatic and environmental drivers suggest the role of evolutionary processes in a polymorphic species. Evol. Ecol. 37: 419-434.
Taub, A.M. (1967): Comparative histological studies on Duvernoy’s gland of colubrid snakes. Bull. Am. Mus. Nat. Hist. 138: 1-50.
Treccani (2024): Enciclopedia Italiana. Accessed August 28, 2024. https://www.treccani.it/.
Trevine, V.C., Grazziotin, F.G., Giraudo, A., Sallaberry‐Pincheira, N., Vianna, J.A., Zaher, H. (2022): The systematics of Tachymenini (Serpentes, Dipsadidae): An updated classification based on molecular and morphological evidence. Zool. Scr. 51: 643-663.
Triep, M., Hess, D., Chaves, H., Brücker, C., Balmert, A., Westhoff, G., Bleckmann, H. (2013): 3D flow in the venom channel of a spitting cobra: Do the ridges in the fangs act as fluid guide vanes? PLOS One 8: e61548.
Vanni, S., Zuffi, M.A.L. (2011): Hierophis viridiflavus (Lacépède, 1789). In: Fauna d’Italia, vol. 45, Reptilia, pp. 509-516. Corti, C., Capula, M., Luiselli, L., Razzetti, E., Sindaco, R., Eds, Bologna, Calderini.
Wagler, J.G. (1830): Natürliches System der Amphibien: mit vorangehender Classification der Säugethiere und Vögel: ein Beitrag zur vergleichenden Zoologie. Munich, J.G. Cotta.
Weinstein, S.A., Warrell, D.A., Keyler, D. (2022): ‘Venomous’ bites from non-venomous snakes. Second edition. Amsterdam, Elsevier.
Westeen, E.P., Durso, A.M., Grundler, M.C., Rabosky, D.L., Davis Rabosky, A.R. (2020): What makes a fang? Phylogenetic and ecological controls on tooth evolution in rear-fanged snakes. BMC Evol. Biol. 20: 80.
Young, B.A., Kardong, K.V. (1996): Dentitional surface features in snakes (Reptilia: Serpentes). Amphib.-Reptil. 17: 261-276.