Submitted on: 2024, 11th November; revised on: 2024, 3rd December; accepted on: 2025, 3rd March
Estimation of the body condition of European cave salamanders (genus Speleomantes) from digital images
1 Dipartimento di Medicina clinica, Sanità pubblica, Scienze della Vita e dell’Ambiente (MeSVA), Università degli Studi dell’Aquila, via Vetoio, Coppito (AQ), 67100 - L’Aquila, Italy
2 Zoologisches Institut – Technische Universität Braunschweig, Mendelssohnstraße 4, 38106 Braunschweig - Braunschweig, Germany
3 Aquila, via dell’Aringo, Preturo (AQ), 67100 - L’Aquila, Italy
*Corresponding author. Email: eleonora.cialente@student.univaq.it
Abstract. Species monitoring is a key activity for conservation studies. Some of the monitoring methods require individual handling, which may provoke negative effects on animal fitness. We here present a new non-invasive method that allows to estimate the body condition of the European cave salamanders (genus Speleomantes) using the tail area as a proxy, since these species usually accumulate fat tissue in their tail. We selected 915 high-quality images of individuals belonging to the eight Speleomantes species. Using the ImageJ program we calculated the tail measurements (length and area) from which we obtained the Fat Tail Index (FTI). The FTI was then correlated with the Scaled Mass Index (SMI) of individuals. We used GLMM to assess whether SMI is correlated to FTI, individual sex, species identity and the type of inhabited environment. We observed a significant correlation between SMI and FTI (R2 = 0.62). The GLMM analysis showed a significant effect on SMI due to sex, species and the type of environment. Females and individuals from surface environments showed the highest SMI. Among species, we observed a significant variability in their body condition and in the correlation between SMI and FTI. This study provided a reliable and non-invasive method that allows to estimate the body condition for terrestrial salamanders of the genus Speleomantes.
Keyword. Scaled mass index, conservation, Hydromantes, monitoring, morphometry, photography.
Monitoring is a key activity for species conservation (Beebee and Griffiths, 2005). It consists of repeated sampling activities that allow us to collect important information on the status of the species and on the ecological features that characterise their environment (McCravy, 2018; Lunghi et al., 2020a). Species monitoring provides direct information on the population consistency and the individuals’ fitness (Ficetola et al., 2018a; Lunghi et al., 2022). It can be performed by adopting non-invasive methods such as counting the observed individuals (Ficetola et al., 2018a), or methods that require animals’ handling (Dunn and Ralph, 2004; Lunghi and Bruni, 2018). Capture-mark-recapture and the record of biometrics parameters represent powerful tools for collecting important data on species; however, they may provide negative side effects on individuals. For example, invasive marking methods (e.g., toe-clipping) can affect individuals’ behaviour and negatively impact their fitness (Golay and Durrer, 1994; Davis and Ovaska, 2001). Negative effects could also be provoked by low-impact techniques such as handling. Individual handling can be a direct source of pathogen transmission, and it can also represent a source of stress that weakens the individual immune system and increases its exposure to potential pathogens (Bliley and Woodley, 2012; Lunghi et al., 2016).
During the last decades, there has been a tendency to adopt less invasive methods aiming to reduce the negative effects on individuals without compromising the quality of data (Soto-Azat et al., 2009; Perry et al., 2011; Gabor et al., 2013). For example, biocompatible compounds (e.g., Visible Implant Elastomers) or distinctive individual patterns are preferred over invasive toe-clipping to individually recognize wild animals (Speybroeck and Steenhoudt, 2017; Lunghi and Bruni, 2018). The use of digital photography in conservation studies further contributes to reducing the negative effects on individuals. Indeed, digital datasets composed of high-quality images have been demonstrated to be a valuable source of information for species monitoring and conservation (Husain et al., 2017).
In this study, we aimed to test a new method that allows to predict the body condition (defined as the amount of fat stored; Wilder et al., 2016) in European cave salamanders (genus Speleomantes) from digital images. The genus Speleomantes includes eight strictly protected amphibian species that are endemic (or sub-endemic) to Italy (Lanza et al., 2006; Rondinini et al., 2022). Speleomantes are fully terrestrial troglophile species (see Howarth and Moldovan, 2018) inhabiting both surface and subterranean environments (Costa et al., 2016; Ficetola et al., 2018b). They are generalist predators that prey on a wide range of invertebrate species (Cianferoni and Lunghi, 2023) and use their tails as organs to accumulate fat tissue (Wake and Dresner, 1967; Fitzpatrick, 1973). Accordingly, the area of the tail should provide reliable information on the amount of fat stored by individuals, which can be considered a fitness-related trait (Wilder et al., 2016). Previous studies have shown a positive correlation between tail width and body condition in urodeles (Bendik and Gluesenkamp, 2013); however, this seemed to not apply to Speleomantes (Rosa et al., 2021). In both studies only the width at the tail base was considered, overlooking the potential that the rest of the tail may have in energy storage. We here specifically tested the use of the overall tail area as a reliable method for predicting the body condition of individuals.
We analysed high-quality images of Speleomantes from different datasets (Lunghi et al., 2020c; Lunghi et al., 2021b; Coppari et al., 2024). Pictures were taken directly in the field and showed a dorsal view of individuals in their natural position (Fig. 1) (Lunghi et al., 2021a). We randomly selected photos of 915 individuals (Table 1), for which sex, weight, and total length were known. To avoid introducing bias, we did not include pictures of individuals with tail issues and gravid females with visible eggs in our dataset. Due to the possibility of recognizing individuals of Speleomantes from the dorsal pattern (Lunghi et al., 2019), pictures taken on the same population but during different periods were checked to avoid pseudoreplication. We used the program ImageJ to obtain measurements of the tail, which extends from the end of the cloaca (tail base) to the tip of the tail (Fig. 1) (Lunghi et al., 2020b). We measured the tail length and area. We divided the tail area for its length to produce a standardized Fat Tail Index (FTI), which should be linked to the amount of adipose tissue the individual stored in its tail (Wake and Dresner, 1967; Fitzpatrick, 1973). We used the Scaled Mass Index (SMI) as a reference to evaluate the reliability of the FTI in predicting Speleomantes’ body condition. The SMI is a reliable index used to estimate fat stored in amphibians (MacCracken and Stebbings, 2012; Rosa et al., 2021), and it is based on the relationship between the body mass of individuals and a linear predictor of body size that accounts for allometric growth (Peig and Green, 2009). Considering the size difference between juveniles and adults and between the different Speleomantes species (Lanza et al., 2006; Lunghi, 2022), we estimated the SMI for each age class (adult vs juveniles) and species separately. We used Generalized Linear Mixed Models (GLMM) implemented in R Studio to evaluate how well FTI correlates with SMI. The SMI was the dependent variable, while FTI, individuals’ sex, species, and habitus (surface vs. subterranean) were independent factors. The use of sex and species allowed us to evaluate the potential divergences occurring between sexes or between species (Lanza et al., 2006; Rosa et al., 2021). Including the habitus among independent variables allowed us to assess whether subterranean populations might show higher efficiency in fat storage, a feature observed in other cave vertebrates (Lunghi and Zhao, 2020). Population identity and the sampling period were included as random factors to account for geographical and seasonal variance in individuals’ body condition (Lunghi et al., 2022). We added the interactions between FTI and the other three variables (sex, species, habitus) as additional predictors. The FTI variable was log-transformed and then centred on its mean to mitigate collinearity. Model selection was performed based on the AIC criterion and through the Likelihood Ratio Test. The model with the lowest AIC showed severe collinearity issues (Variance Inflation Factor, VIF > 10), making parameter estimates unreliable. Therefore, we selected a slightly higher AIC model with acceptable collinearity levels (VIF < 7) to ensure stability and interpretability (Table 2).

| Species | Number of juveniles | Number of males | Number of females | Total |
|---|---|---|---|---|
| S. italicus | 30 | 56 | 55 | 141 |
| S. supramontis | 46 | 31 | 44 | 121 |
| S. sarrabusensis | 36 | 21 | 17 | 74 |
| S. ambrosii | 62 | 29 | 49 | 140 |
| S. strinatii | 47 | 41 | 35 | 123 |
| S. imperialis | 41 | 30 | 15 | 86 |
| S. genei | 41 | 41 | 40 | 122 |
| S. flavus | 29 | 34 | 45 | 108 |
| Total | 332 | 283 | 300 | 915 |
| Model | Df | AIC | BIC | Log-Likelihood | Deviance | Chi-Square | ΔDf | P-value |
|---|---|---|---|---|---|---|---|---|
| FTI*Domain + Species + FTI*Sex | 15 | 566.89 | 639.17 | -268.445 | 536.89 | |||
| Domain + Species + Sex | 15 | 566.89 | 639.17 | -268.445 | 536.89 | 0 | 0 | |
| FTI*Domain + Species + Sex | 16 | 539.94 | 617.41 | -253.971 | 507.94 | 28.949 | 1 | < 0.001 |
| Domain + Species + FTI*Sex | 17 | 567.86 | 649.78 | -266.929 | 533.86 | 0 | 1 | 1 |
| Domain + FTI*Species + Sex | 22 | 237.08 | 343.09 | -96.538 | 193.08 | 340.781 | 5 | < 0.001 |
| FTI*Domain + FTI*Species + Sex | 23 | 239.07 | 349.91 | -96.537 | 193.07 | 0.001 | 1 | 0.970 |
| Domain + FTI*Species + FTI*Sex | 24 | 225.96 | 341.62 | -88.981 | 177.96 | 15.113 | 1 | < 0.001 |
| FTI*Domain + FTI*Species + FTI*Sex | 25 | 227.43 | 347.90 | -88.714 | 177.43 | 0.534 | 1 | 0.465 |
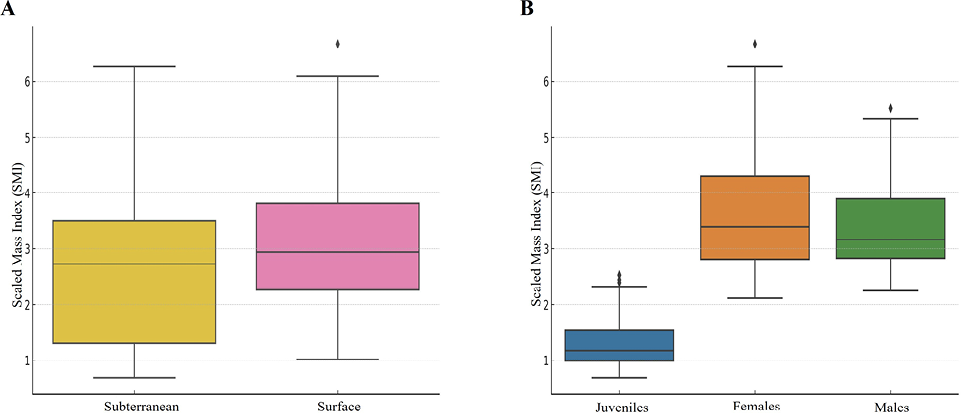
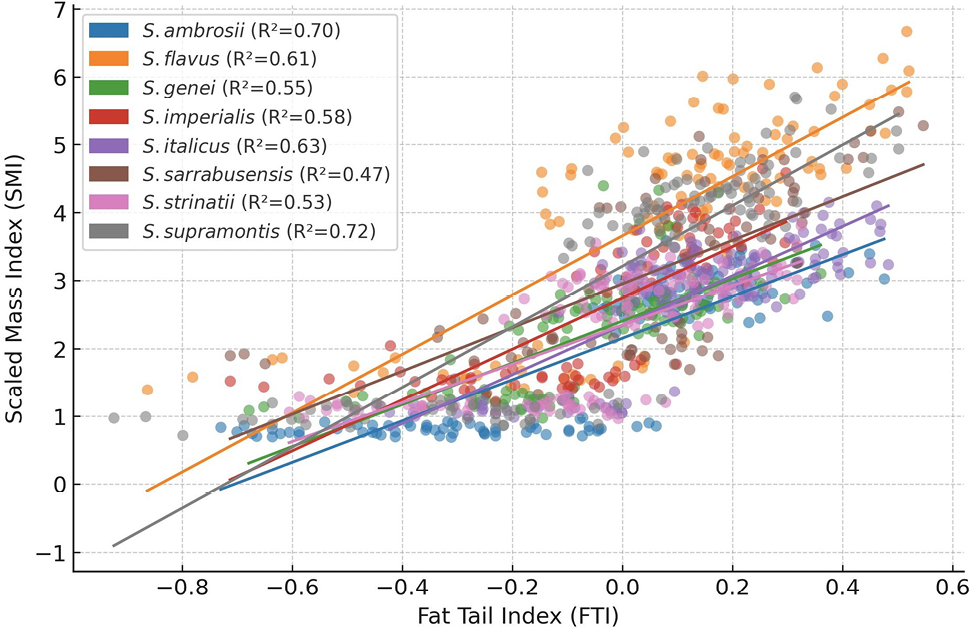
We identified a significant correlation of SMI with FTI (χ2 (1) = 204.07, P < 0.001), habitus (χ2 (1) = 5.73, P = 0.017), species (χ2 (7) = 110.64, P < 0.001), sex (χ2 (2) = 5400.25, P < 0.001) and with the interaction between FTI and species (χ2 (7) = 424.90, P < 0.001). The SMI was higher in surface populations (β =0.159, SE = 0.07, P = 0.017) (Fig. 2A) and in females (males, β = -0.05, SE = 0.01, P < 0.001; juveniles, β = -0.93, SE = 0.01, P < 0.001) (Fig. 2B). The correlation between SMI and FTI significantly diverged between Speleomantes species (Fig. 3, Supplementary Materials Fig. S1). This model showed a very high explanatory power (marginal R2 = 0.95, conditional R2 = 0.96).
Our study identified the Fat Tail Index (FTI) as a reliable method to estimate individual body condition regarding the amount of energy reserves. The correlation between FTI and SMI was strong and relatively high (R2 = 0.62), indicating that this index can be reliably employed to estimate the body condition of Speleomantes from digital images, even in the absence of individual weight. This outcome further highlights the usefulness of digital photos in conservation studies and the potential contribution that citizen science could provide for the monitoring of animal population.
Our findings opposed the results of previous studies where the use of the tail width was not recommended for the estimation of the body condition for this genus. In their study, Rosa et al. (2021) considered the width at the tail base as a proxy of individual fitness in S. imperialis (using the Scale Mass Index, SMI). Instead, we propose using the overall tail area (FTI) because adipose tissue is likely stored throughout the tail length and not just at its base (Fitzpatrick, 1973). Our method was highly supported by the analysis, confirming the hypothesis that fat tissue is accumulated not only in a specific section but throughout the tail organ. In some circumstances, the enlargement at the tail base might be considered a proxy for the overall tail enlargement due to fat storage (Bendik and Gluesenkamp, 2013). Still, it does not always work (Rosa et al., 2021). Indeed, in their study, Rosa et al. (2021) assessed the correlation between SMI and the tail base only in S. imperialis. Considering the variability of the correlation between SMI and FTI observed here (Fig. 3, but see also Fig. S1), it may be possible that the tail base may be more diagnostic for some other Speleomantes species. On the other hand, the assessment of the overall increase in the tail volume seems more reliable and informative than the use of specific tail parts (Rosa et al., 2021). Although the proposed method already represents a reliable alternative for estimating the body condition in Speleomantes, considering the overall tail volume might further increase the accuracy of the estimation.
Rosa et al. (2021) found a significant correlation between tail width and SMI in females but not males. In our analysis, we observed a strong effect of sex on SMI, where females showed the highest correlation and juveniles the lowest (Fig. 2B). Sexually mature females need to store large amounts of energy to carry out highly demanding reproduction and consequently usually have a higher body condition than males of similar size (Lunghi et al., 2018b). On the other hand, the low correlation with SMI observed for juveniles can also be easily explained. Juvenile Speleomantes likely invest more in growth than in fat storage to quickly reach a size that makes them unsuitable for some predators (Lunghi and Corti, 2021). This was also hypothesized in other studies where juveniles often occurred in sub-optimal environmental conditions to find the highest prey availability (Ficetola et al., 2013; Lunghi et al., 2015).
We observed a divergence in SMI among the eight Speleomantes species (Fig. 3). The group of species that showed the highest correlation with SMI was composed of four Sardinian species (S. flavus, S. imperialis, S. supramontis, S. sarrabusensis) and S. italicus. The four Sardinian species showed the highest divergence (β = 0.30-0.51), while that for S. italicus was moderate (β = 0.15). This group of Sardinian Speleomantes is defined as “giant” due to their larger size compared to the other congeneric species (Lanza et al., 2006). The larger size of these species may allow them to increase the proportion of fat stored in their body (Fitzpatrick, 1973), or they can have access to bigger prey that provide a more significant amount of nutrients (Lunghi et al., 2018a). On the other hand, hypothesizing the reasons for the higher SMI observed in S. italicus compared to the other similar-sized species is more challenging, and, considering the lack of supporting information, we save this for future assessments to avoid falling into mere speculations.
The correlation between FTI and SMI showed significant variability between species, ranging from 0.47 for S. sarrabusensis to 0.72 for S. supramontis (Fig. 2). This means that, although FTI is a good proxy to estimate Speleomantes body condition, for some species this prediction seems to be stronger. Unfortunately, we do not have supporting information to explain this result. Further analyses aiming to evaluate potential physiological and morphological interspecific divergences are needed to shed light on this interesting case.
We observed a higher SMI in surface populations of Speleomantes. This result does not support the hypothesis that subterranean populations of Speleomantes show improved abilities in fat storage (Lunghi and Zhao, 2020) but rather raises alternative hypotheses. Speleomantes from surface environments are mostly active during wet seasons, meaning that their main foraging activity likely occurs only during limited periods (Costa et al., 2016; Salvidio et al., 2017). Contrarily, subterranean populations can buffer hostile seasons (too hot and/or dry) thanks to the underground microclimate (Culver and Pipan, 2019), which allows them, and their prey as well, to be more or less active all year round (Lunghi et al., 2022). Therefore, in this circumstance, the opposite could occur, and the surface populations may show enhanced abilities for fat storage. On the other hand, although being active for less time, surface populations have at disposal higher prey diversity and availability compared to those living in subterranean environments (Culver and Pipan, 2019), a key factor that can compensate for the reduced foraging activity and provide more nutrients that can be translated into a higher body condition of individuals. These hypotheses need to be explored to shed light on the dynamics behind the observed divergence in SMI between surface and subterranean populations.
In conclusion, our study demonstrated the validity of the correlation between the tail area and the individuals’ body condition in Speleomantes, making this a valuable proxy for implementing conservation and ecological studies.
Supplementary material associated with this article can be found at <http://www-9.unipv.it/webshi/appendix/index.html> manuscript number 16853
Beebee, T.J.C., Griffiths, R.A. (2005): The amphibian decline crisis: a watershed for conservation biology? Biol. Conserv. 125: 271-285.
Bendik, N.F., Gluesenkamp, A.G. (2013): Body length shrinkage in an endangered amphibian is associated with drought. J. Zool. 290: 35-41.
Bliley, J.M., Woodley, S.K. (2012): The effects of repeated handling and corticosterone treatment on behavior in an amphibian (Ocoee salamander: Desmognathus ocoee). Physiol. Behav. 105: 1132-1139.
Cianferoni, F., Lunghi, E. (2023): Inferring on Speleomantes foraging behavior from gut contents examination. Animals 13: 2782.
Coppari, L., Di Gregorio, M., Corti, C., Merilli, S., Mulargia, M., Cogoni, R., Manenti, R., Ficetola, G.F., Lunghi, E. (2024): Four years monitoring of the endangered European plethodontid salamanders. Sci. Data 11: 706.
Costa, A., Crovetto, F., Salvidio, S. (2016): European plethodontid salamanders on the forest floor: local abundance is related to fine-scale environmental factors. Herpetol. Conserv. Biol. 11: 344-349.
Culver, D.C., Pipan, T. (2019). The biology of caves and other subterranean habitats. New York, Oxford University Press.
Davis, T.M., Ovaska, K. (2001): Individual recognition of amphibians: effects of toe clipping and fluorescent tagging on the salamander Plethodon vehiculum. J. Herpetol. 35: 217-225.
Dunn, E.H., Ralph, C.J. (2004): Use of mist nets as a tool for bird population monitoring. Stud. Avian Biol. 29: 1-6.
Ficetola, G.F., Barzaghi, B., Melotto, A., Muraro, M., Lunghi, E., Canedoli, C., Lo Parrino, E., Nanni, V., Silva-Rocha, I., Urso, A., Carretero, M.A., Salvi, D., Scali, S., Scarì, G., Pennati, R., Andreone, F., Manenti, R. (2018a): N-mixture models reliably estimate the abundance of small vertebrates. Sci. Rep. 8: 10357.
Ficetola, G.F., Lunghi, E., Canedoli, C., Padoa-Schioppa, E., Pennati, R., Manenti, R. (2018b): Differences between microhabitat and broad-scale patterns of niche evolution in terrestrial salamanders. Sci. Rep. 8: 10575.
Ficetola, G.F., Pennati, R., Manenti, R. (2013): Spatial segregation among age classes in cave salamanders: habitat selection or social interactions? Popul. Ecol. 55: 217-226.
Fitzpatrick, L.C. (1973): Energy allocation in the Allegheny mountain salamander, Desmognathus ochrophaeu. Ecol. Monogr. 43: 43-58.
Gabor, C.R., Bosch, J., Fries, J.N., Davis, D.R. (2013): A non-invasive water-borne hormone assay for amphibians. Amphib.-Reptil. 34: 151-162.
Golay, N., Durrer, H. (1994): Inflammation due to toe-clipping in natterjack toads (Bufo calamita). Amphib.-Reptil. 15: 81-96.
Howarth, F.G., Moldovan, O.T. (2018). The ecological classification of cave animals and their adaptations. In: Cave Ecology, pp. 41-67. Moldovan, O.T., Kováč, L., Halse, S., Eds., Berlin, Springer.
Husain, N., Roy, P., Hussain Trak, T. (2017): Photography as a conservation tool in science. Trends Biosci. 10: 9317-9321.
Lanza, B., Pastorelli, C., Laghi, P., Cimmaruta, R. (2006): A review of systematics, taxonomy, genetics, biogeography and natural history of the genus Speleomantes Dubois, 1984 (Amphibia Caudata Plethodontidae). Atti Mus. civ. stor. nat. Trieste 52: 5-135.
Lunghi, E. (2022): Doubling the lifespan of European plethodontid salamanders. Ecology 103: 1-4.
Lunghi, E., Bacci, F., Zhao, Y. (2021a): How can we record reliable information on animal colouration in the wild? Diversity 13: 356.
Lunghi, E., Bruni, G. (2018): Long-term reliability of Visual Implant Elastomers in the Italian cave salamander (Hydromantes italicus). Salamandra 54: 283-286.
Lunghi, E., Cianferoni, F., Ceccolini, F., Veith, M., Manenti, R., Mancinelli, G., Corti, C., Ficetola, G.F. (2018a): What shapes the trophic niche of European plethodontid salamanders? PLOS ONE 13: e0205672.
Lunghi, E., Cianferoni, F., Corti, C., Zhao, Y., Manenti, R., Ficetola, G.F., Mancinelli, G. (2022): The trophic niche of subterranean populations of Speleomantes italicus: a multi-temporal analysis. Sci. Rep. 12: 18257.
Lunghi, E., Cianferoni, F., Giachello, S., Zhao, Y., Manenti, R., Corti, C., Ficetola, G.F. (2021b): Updating salamander datasets with phenotypic and stomach content information for two mainland Speleomantes. Sci. Data 8: 150.
Lunghi, E., Corti, C. (2021): Predation of European cave salamanders (Speleomantes) by the spider Meta bourneti. Spixiana 44: 54.
Lunghi, E., Corti, C., Manenti, R., Barzaghi, B., Buschettu, S., Canedoli, C., Cogoni, R., De Falco, G., Fais, F., Manca, A., Mirimin, V., Mulargia, M., Mulas, C., Muraro, M., Murgia, R., Veith, M., Ficetola, G.F. (2018b): Comparative reproductive biology of European cave salamanders (genus Hydromantes): nesting selection and multiple annual breeding. Salamandra 54: 101-108.
Lunghi, E., Corti, C., Mulargia, M., Zhao, Y., Manenti, R., Ficetola, G.F., Veith, M. (2020a): Cave morphology, microclimate and abundance of five cave predators from the Monte Albo (Sardinia, Italy). Biodivers. Data J. 8: 1-16.
Lunghi, E., Giachello, S., Manenti, R., Zhao, Y., Corti, C., Ficetola, G.F., Bradley, J.G. (2020b): The post hoc measurement as a safe and reliable method to age and size plethodontid salamanders. Ecol. Evol. 10: 11111-11116.
Lunghi, E., Giachello, S., Zhao, Y., Corti, C., Ficetola, G.F., Manenti, R. (2020c): Photographic database of the European cave salamanders, genus Hydromantes. Sci. Data 7: 171.
Lunghi, E., Manenti, R., Canciani, G., Scarì, G., Pennati, R., Ficetola, G.F. (2016): Thermal equilibrium and temperature differences among body regions in European plethodontid salamanders. J. Therm. Biol. 60: 79-85.
Lunghi, E., Manenti, R., Ficetola, G.F. (2015): Seasonal variation in microhabitat of salamanders: environmental variation or shift of habitat selection? PeerJ 3: e1122.
Lunghi, E., Romeo, D., Mulargia, M., Cogoni, R., Manenti, R., Corti, C., Ficetola, G.F., Veith, M. (2019): On the stability of the dorsal pattern of European cave salamanders (genus Hydromantes). Herpetozoa 32: 249-253.
Lunghi, E., Zhao, Y. (2020): Do Chinese cavefish show intraspecific variability in morphological traits? Ecol. Evol. 10: 7723-7730.
MacCracken, J.G., Stebbings, J.L. (2012): Test of a body condition index with amphibians. J. Herpetol. 46: 346-350.
McCravy, K.W. (2018): A review of sampling and monitoring methods for beneficial arthropods in agroecosystems. Insects 9: 170.
Peig, J., Green, A.J. (2009): New perspectives for estimating body condition from mass/length data: the scaled mass index as an alternative method. Oikos 118: 1883-1891.
Perry, G., Wallace, M.C., Perry, D., Curzer, H., Muhlberger, P. (2011): Toe clipping of amphibians and reptiles: science, ethics, and the law. J. Herpetol. 45: 547-555.
Rondinini, C., Battistoni, A., Teofili, C. (2022). Lista Rossa IUCN dei vertebrati italiani 2022. Roma, Comitato Italiano IUCN e Ministero dell’Ambiente e della Sicurezza Energetica.
Rosa, G., Costa, A., Renet, J., Romano, A., Roner, L., Salvidio, S. (2021): Energy storage in salamanders’ tails: the role of sex and ecology. Sci. Nat. 108: 27.
Salvidio, S., Pasmans, F., Bogaerts, S., Martel, A., van de Loo, M., Romano, A. (2017): Consistency in trophic strategies between populations of the Sardinian endemic salamander Speleomantes imperialis. Anim. Biol. 67: 1-16.
Soto-Azat, C., Clarke, B.T., Fisher, M.C., Walker, S.F., Cunningham, A.A. (2009): Non-invasive sampling methods for the detection of Batrachochytrium dendrobatidis in archived amphibians. Dis. Aquat. Org. 84: 163-166.
Speybroeck, J., Steenhoudt, K. (2017): A pattern-based tool for long-term, large-sample capture-mark-recapture studies of fire salamanders Salamandra species (Amphibia: Urodela: Salamandridae). Acta Herpetol. 12: 55-63.
Wake, D.B., Dresner, I.G. (1967): Functional morphology and evolution of tail autotomy in salamanders. J. Morphol. 122: 265-306.
Wilder, S.M., Raubenheimer, D., Simpson, S.J. (2016): Moving beyond body condition indices as an estimate of fitness in ecological and evolutionary studies. Funct. Ecol. 30: 108-115.