Submitted on: 2024, 26th November; revised on: 2024, 6th December; accepted on: 2025, 12th May
A comparative analysis of female genitalia of seven Old World snake species using a silicone modeling technique
1 Institute of Biodiversity and Ecosystem Research, Bulgarian Academy of Sciences, 2 Gagarin Street, 1113 Sofia, Bulgaria
2 Independent researcher, Krakra 30 street, Dragichevo, Bulgaria
*Corresponding author. Email: kandonov91@gmail.com
Abstract. Genitalia diversity has long been an object of evolutionary and functional morphology studies, with a primary focus on male copulatory organs. Despite extensive studies on snake genitalia, particularly hemipenes, female copulatory organs remain understudied. This research aims to fill this gap by modifying a recently introduced silicone modeling methodology for preparation and by describing female snake genitalia for seven previously undescribed species. The methodology is based on employing a two-component condensation silicone into snakes’ genitalia to create internal models for intersexual comparative morphology. We conducted a comprehensive examination of seven Old World snake species – Boidae: Eryx jaculus, Colubridae: Dolichophis caspius, Zamenis longissimus, Elaphe quatuorlineata, Coronella austriaca, and Viperidae: Vipera ammodytes, and V. berus. Our results reveal significant variations in female genitalia morphology and we speculate that this is also reflected in copulation adjustment between the different types of species’ genitalia.
Keywords. Copulation, morphology, cloaca, urodaeum, pouches.
Closely related taxa often exhibit morphologically distinctive genitalia (Arnqvist, 1998; Gredler et al., 2014). Understanding genital diversity has long been an object of functional morphology and evolutionary research, with most studies focused mainly on male copulatory organs (i.e., Dufour, 1844; Darwin, 1871; Mayr, 1963; Thornhill, 1983; Eberhard, 1985; Shapiro and Porter, 1989; East et al., 1993; Hosken and Stockley, 2004; Cohn, 2011; Gredler et al., 2014; Brennan and Prum, 2015; Langerhans et al., 2016; Brennan, 2022). Since the pioneering work of Dufour (1844), who proposed the lock-and-key mechanism, researchers have concentrated not only on the morphology of male copulatory organs but their evolution as well. However, in the past several decades, the lock-and-key mechanism has been confronted (Eberhard, 1985, 2010; Shapiro and Porter, 1989). A strong argument against the lock-and-key hypothesis is the lack of the supposed female “locks” in many groups (Eberhard 1985; Shapiro and Porter, 1989), and the expected pattern of character displacement in males within zones of sympatry among closely related species is often absent (i.e., Ware and Opell, 1989; Eberhard, 2010). Nevertheless, most of the studies have focused mainly on male copulatory organs, while female genitalia are still heavily understudied (Ah-King et al., 2014), and thus, more studies on female genitalia with various techniques are required.
Snake genitalia, in particular, have been studied for many years. After the fundamental study of Cope (1895) who describes the hemipenial morphology of more than 200 species, the general morphology of hemipenes and female genitalia are considered species-specific (Dowling and Savage, 1960; Keogh, 1999; Zaher, 1999, Siegel et al., 2012). Male snakes possess two copulatory organs known as hemipenis, which are often ornamented with various structures (Cope, 1895; Dowling and Savage, 1960; Keogh, 1999; Andonov et al., 2017). The hemipenes are inverted inside a hemipenial sheath in the proximal part of the tail, close to the cloacal opening and are everted during copulation or sometimes as a defense behavior (i.e., Cope, 1895; Dowling and Savage, 1960). The female reproductive tract consists of two oviducts (pouch, non-glandular uterus, glandular uterus, posterior infundibulum, anterior infundibulum), and two ovaries, with sperm storage primarily occurring in the non-glandular uterus and posterior infundibulum (Siegel et al., 2012; Jurkfitz et al., 2023). However, there is an ongoing debate whether the pouch is derivative of the urodeaeum (Blackburn 1988) or the oviducts (Siegel et al., 2011). The two paired oviducts are enclosed in a thin visceral pleuroperitoneum and are suspended in the coelomic cavity by a dorsal mesentery. The non-glandular uterus features a thin lamina propria and is lined with an epithelium mainly made up of ciliated cells (Siegel et al., 2011). From a macroscopic perspective, the cloaca can be segmented into three to four distinct regions, varying by species: the proctodaeum, urodaeum, an anterior extension of the urodaeum (which may not exist in all species), and a coprodaeal complex consisting of two regions (Siegel et al., 2011, 2012). Despite the prevalence of studies on male genitalia, over the last century that have been a number of studies on female genitalia as well (i.e., Pope, 1941; Edgren, 1953; Inger and Marx, 1962; Gabe and Saint-Girons, 1965; Pisani, 1976; Siegel et al., 2011, 2012; Showalter et al., 2014, Granados et al., 2022). Still, there are only а few studies analyzing the potential alignment between hemipenes and pouch (Pope, 1941; Inger and Marx, 1962, Granados et al., 2022).
Female genitalia have been described mainly in situ after dissection of certain individuals with very few exceptions (Granados et al., 2022), while male hemipenes are usually described in their everted and inflated state. Such two-dimensional observation of female genitalia restricts the potential for further investigation of copulatory adjustment of male and female genitalia. We believe that using a better methodology for female snake genitalia preparation and their respective description, with a three-dimensional (3D) view of the organs following Granados et al. (2022), is necessary for a proper comparison to the common hemipenial descriptions, so further analyses for potential copulatory adjustments could be performed. Similar methodology for three-dimensional genitalia models has been used not only for snakes, but also for caimans (Moore et al., 2022), and marine mammals (Orbach et al., 2018; 2021).
In the present study, we use a recently proposed methodology that allows a description of female snake genitalia in a shape that presumably has the closest resemblance to its form during copulation (Granados et al., 2022) with a few minor additions. Such descriptions can give useful information for comparative morphology between male and female genitalia and provide valuable knowledge for testing one of the main hypotheses for genital evolution, i.e., lock-and-key mechanism. Moreover, we provide the first descriptions of female genitalia for seven Old World snake species, based on this methodology.
We investigated the female genitalia morphology of seven Old World snake species from three families. For the purposes of the study and intersexual comparative morphology, we used 16 adult female individuals belonging to the following species: Boidae: Eryx jaculus (Linnaeus, 1758) (n = 1); Colubridae: Dolichophis caspius (Gmelin, 1789) (n = 2), Zamenis longissimus (Laurenti, 1768) (n = 2), Elaphe quatuorlineata (Lacepède, 1789) (n = 2), Coronella austriaca Laurenti, 1768 (n = 1); Viperidae: Vipera ammodytes (Linnaeus, 1758) (n = 5), V. berus (Linnaeus, 1758) (n = 1). Since there are records for ontogenetic differences in the female genitalia shape (Showalter et al., 2014), only adult individuals were used. Individuals were considered adults based on species-specific total length (Ltot) threshold, which is the sum of the snout-vent length (SVL) and the tail-length: E. jaculus: Ltot >40 cm, D. caspius and E. quatuorlineata: Ltot >100 cm; Z. longissimus: Ltot >90 cm; C. austriaca and V. ammodytes: Ltot >46 cm; V. berus: Ltot >35 cm (Biserkov et al., 2007; Stojanov et al., 2011; the authors’ unpublished data). The individuals that we used were either from the collection of the National Museum of Natural History, Bulgarian Academy of Sciences in Sofia, or freshly found dead specimens (i.e., dead on the road individuals) (see Supplementary file 1).
The posterior end (tail with part of the abdomen) of each tested snake was immersed in 2% KOH solution for 3-6 hours, depending on the level of fixation and the size of the individual (Pesantes, 1994). After the tissue softened and became rubber-like, a two-component condensation dental silicone was injected into the individuals’ cloaca.
For the preparation of the genitalia models, we followed a methodology proposed by Granados et al. (2022) with a few adjustments and changes, such as not tying the oviducts or the intestine immediately after the cloaca, but further, in order to receive additional information about key structures, and not removing the cloaca from the individual. Each specimen was securely tied approximately 5-6 cm anterior to the cloacal opening to ensure the silicone enters the vaginal pouch first and does not spread to the abdomen and intestines. We used Stomaflex™ Light as a solid silicone component and Stomaflex™ Gel Catalyst to harden the solid component. Each filling hardened for 3-5 minutes, after which a small incision was made at the base of the cloacal opening, followed by removal of the silicone model (Fig. 1). The models were washed with water and kept in plastic tubes. The snake specimens were also washed with water and re-preserved in 90% ethanol.

For the description of the female genitalia, we follow (with some additions) the terminology adopted by Siegel et al. (2011, 2012) where the bifurcated cloacal area, that receives the distal part of the hemipenis, is named “vaginal pouch” (or only “pouch”). Considering that Seigel et al. (2011, 2012) describe the states of bifurcation of the pouch histologically, while we observe them only morphologically, we do not follow the four-state categorization of the pouches (bifurcated pouch, separated pouch arms with no urodaeal divide, separated pouch arms with urodaeal divide, and simple pouch), but classify them as follows: simple – no bifurcation, slightly separated – when the silicone model arms are less than 25% of the total model length, separated – when the model arms are between 25%-50% of the total model length, and bifurcated – when the model arms are more than 50% of the total model length. This classification has a close resemblance to the respective term for the general shape of hemipenes, allowing the comparisons to be as accurate as possible. For the description of hemipenes, we followed Andonov et al. (2017), where the terminology is mainly based on Dowling and Savage (1960) and Zaher (1999) with a few additions and clarifications. Following the characteristic for male genitalia introduced in Andonov et al. (2017) – hemipenial proportion index (HPI), we suggest a similar index for the female genitalia description – female genital proportion index (FGPI) where the maximal width of the silicone model is divided by its total length. Female genitalia models with FGPI>0.5 were considered “stubby”; those with FGPI between 0.5 and 0.25— “medium formed”; those with FGPI< 0.25— “elongated”.
Considering that we have tested five female V. ammodytes individuals, we conducted descriptive statistics for the species.
All photos of the models were made with Sony DSC-HX400V Ver. 2.10 on a black background, and the hemipenes described by Andonov et al. (2017) have been photographed using a high-resolution digital camera (Nikon COOLPIX P510) by placing the object on a glass slide positioned about 20 cm above a black background to reduce unwanted shadows.
The research was carried out in accordance with permit № 861/13.01.2021 provided by the Ministry of Environment and Waters.
The models showed that female cloaca and vaginal pouches were prominent and some silicon has entered the intestines as well (Fig. 2).
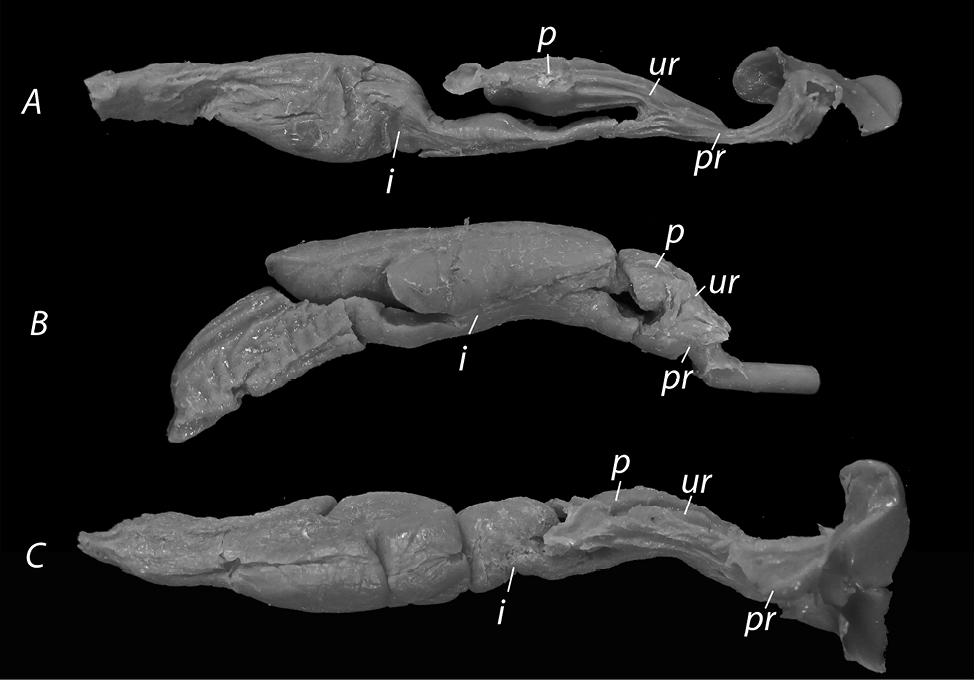
In this section, we provide a detailed morphological description of the female cloaca. All figures present the silicone models provided during the current research, as well as pictures of the species’ hemipenes as described in Andonov et al. (2017).
Eryx jaculus. The silicone model (n = 1) is short and the pouch arms are almost absent. However, there is a very slight bifurcation present; the genitalia model is not bifurcated to slightly bifurcated and stubby – FGPI > 0.5 (Fig. 3). It differs from all other models significantly, being the only stubby model. On the contrary, the hemipenis is medium formed according to Andonov et al., 2017, with an undivided sulcus spermaticus and with not much ornamentation, lacking any calcified structures or calyces, with only a few flounces with scalloped edges present.
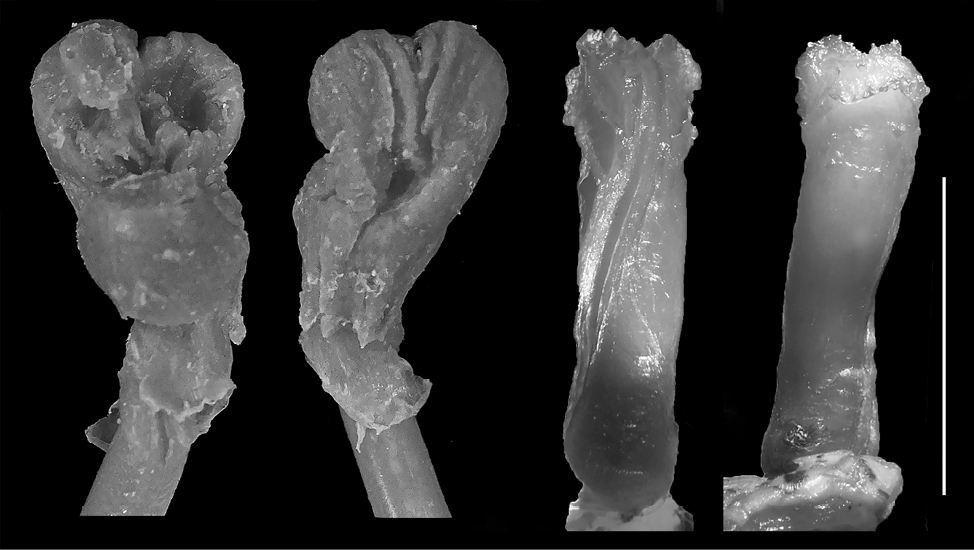
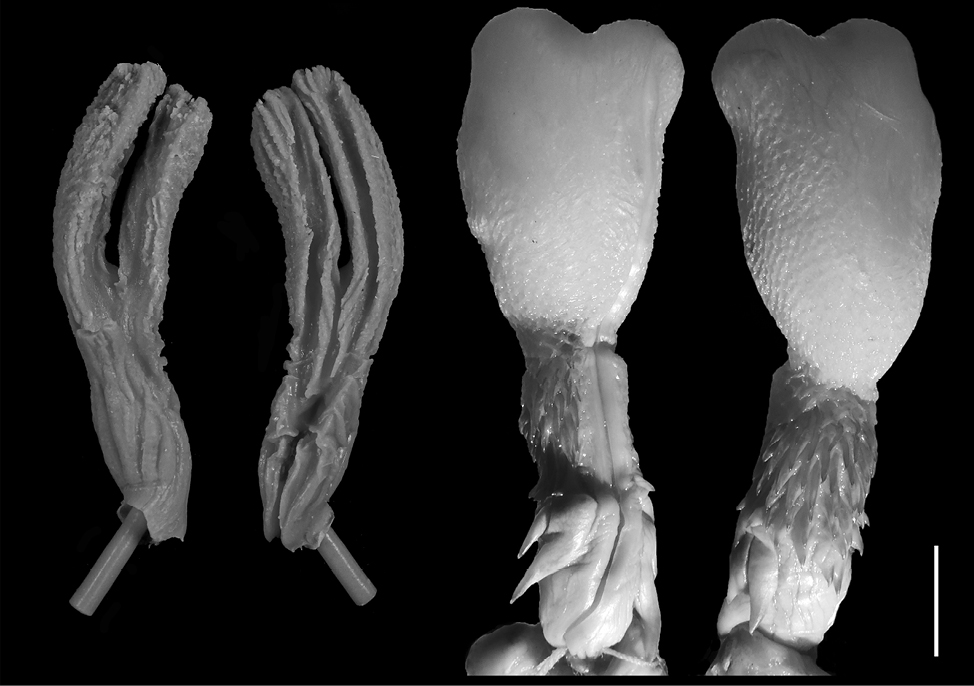
Coronella austriaca. The model (n = 1) is relatively long with prominent bifurcation and long pouch arms, being elongated (FGPI = 0.22) and slightly separated (Fig. 4). It has a similarity with the hemipenis general shape of the species where one of the hemipenial lobes is slightly shorter than the other, although this similarity might be due to a preparation imperfection or an artifact in the intestine (see Discussion). The hemipenis has an undivided s. spermaticus.
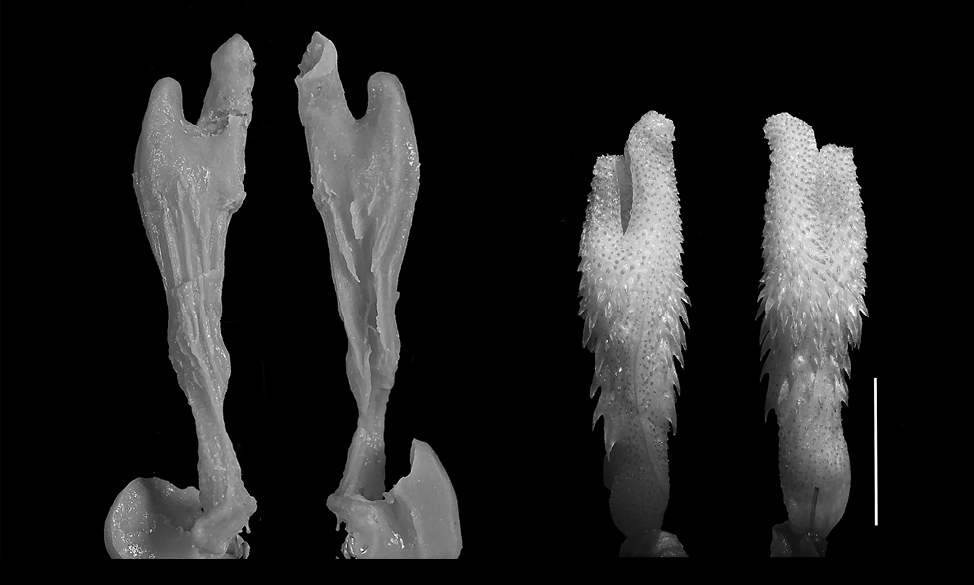
Dolichophis caspius. The models (n = 2) are elongated (FGPI = 0.33-0.36) with prominent bifurcation and long, separated pouch arms (Fig. 5). There is a distinctive crease in the base of the vaginal pouch arms, which shows a conspicuous similarity with the species general hemipenial shape. The hemipenis, as described in Andonov et al. (2017) is simple and bulbous and descriptively fits to one of the vaginal pouch arms.
Elaphe quatuorlineata. The models (n = 2) are elongated with prominent bifurcation and very long pouch arms, being elongated (FGPI = 0.16-0.28) and bifurcated (Fig. 6). This model, along with the one of Z. longissimus has the longest pouches in relation to the total length. The hemipenis is slightly bilobed, bulbous, medium-formed, and does not possess many calcified or non-calcified structures.
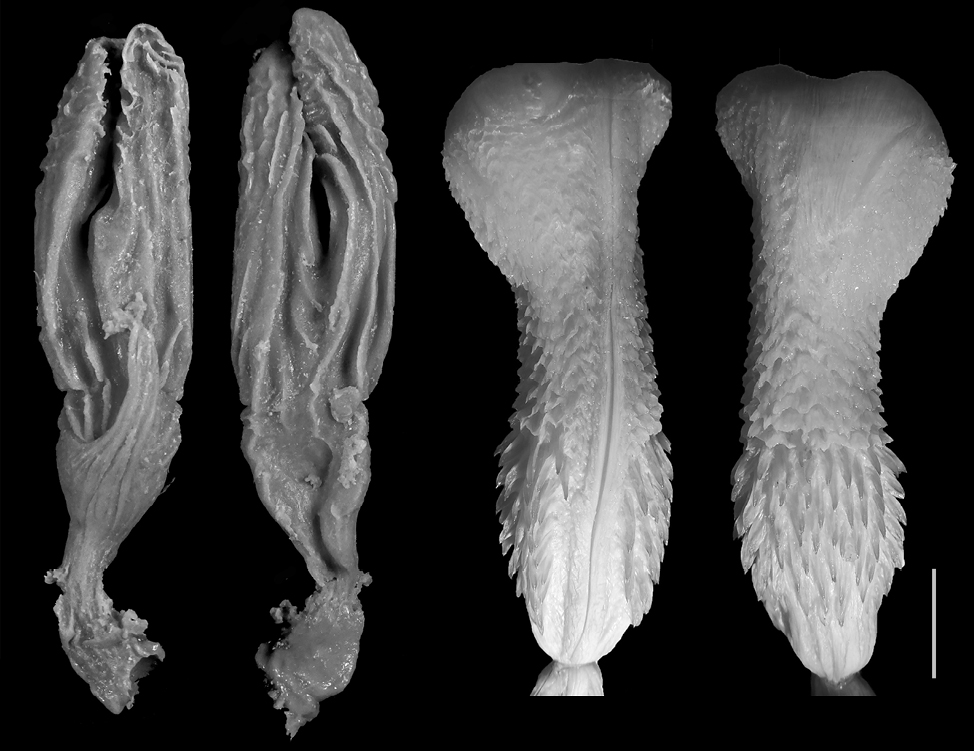
Zamenis longissimus. The models (n = 2) are elongated to medium (FGPI = 0.25-0.35), separated, with prominent bifurcation and very long pouch arms (Fig. 7). This is the model with the longest pouches in relation to the total length. The hemipenis is slightly bilobed, bulbous, medium-formed, and does not possess many calcified or non-calcified structures.
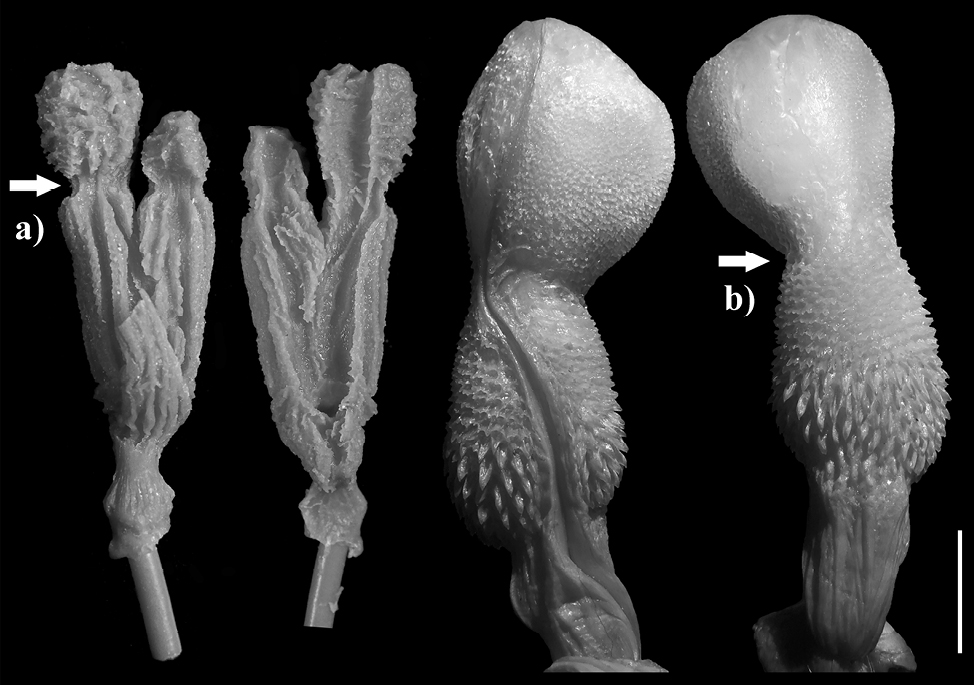
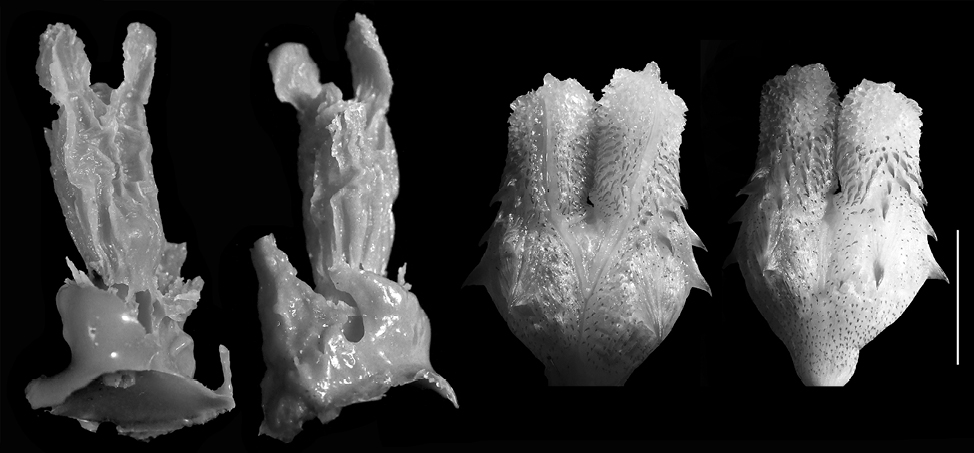
Vipera ammodytes. The models (n = 5) are medium formed (FGPI = 0.36-0.62, M = 0.459, SD = 0.104) and slightly separated, with very short vaginal pouch arms (Fig. 8). The species hemipenis on the other side is divided and subcylindrical in shape. The ornamentation of the male copulatory organ is pronounced with multiple calcified structures present (i.e., spines, calyces).
Vipera berus. The model (n = 1) is medium formed (FGPI = 0.32) and slightly separated, with very short pouch arms (Fig. 9). The characteristics are similar to those of V. ammodytes, and the species hemipenis is also similarly divided, with subcylindrical shape resembling the male organ of V. ammodytes.
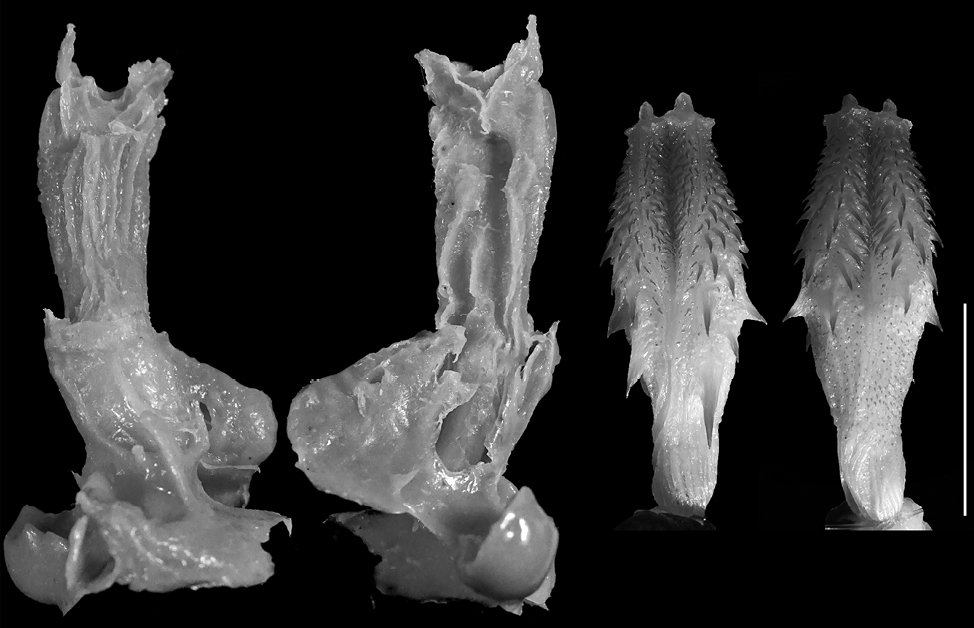
In general, most of the models of the representatives of the family Colubridae show а much higher level of bifurcation and the pouches are longer, in comparison to the base of the cloaca (Fig. 4-7). In the representatives of the family Viperidae (Fig. 8-9), the pouches are significantly shorter, and the female cloaca shape in Eryx jaculus (Fig. 3) differs from the species of both other families. The female genitalia models of species where hemipenes are divided are much less bifurcated, while the genitalia models of species for which the hemipenes are typically simple (no bilobation observed) or slightly bilobed (the bilobation is very low with the body of the hemipenis being more than 75% of the total hemipenial length) have a significant bifurcation and very prominent pouch.
In this study, we describe for the first time the female genital morphology of seven snake species (E. jaculus, D. caspius, Z. longissimus, E. quatuorlineata, C. austriaca, V. ammodytes, and V. berus), using a recently proposed methodology (Granados et al., 2022) with a few modifications. The results show significant variation between some species and suggest different types of adjustment of male and female genitalia during copulation (see Genitalia morphology). The methodology used in the present study also has the potential to reveal valuable information about the process of copulation and might shed light on some of the hypotheses for genital evolution in snakes. Female genital morphology in snakes is considered species-specific (Siegel et al., 2011, 2012), although Showalter et al. (2014) also suggest some intraspecific variation. We did not find intraspecific variation in species where multiple models were made. In particular, analyses of the bigger sample of V. ammodytes models reveal that their general shape does not vary and the proportions between them are similar (see Results). However, a larger sample is required to draw a definitive conclusion. For the current discussion, we assume that the general morphology of female genitalia is species-specific, following Siegel et al. (2012), which is corroborated by the lack of variation in V. ammodytes.
There is a different level of asymmetry in some of the models of the bifurcated pouches. This could be due to uneven inflation by the silicone, although some minor differences between the pouch arms could be expected, considering the similar asymmetry observed in the simple pouch of Afrotyphlops punctatus (Leach, 1819), where only the right pouch arm is present (Gabe and Saint-Girons, 1965). Further studies on more and fresher specimens could reveal additional variation.
With a few exceptions in the early years of research of hemipenial morphology, where male snake copulatory organs are described in situ (i.e., Cope, 1895), hemipenes are usually described in an everted and fully expanded state (i.e., Dowling and Savage, 1960; Pesantes, 1994; Keogh, 1999; Zaher, 1999; Zaher and Prudente, 2003). On the contrary, most descriptions of the female snake cloaca are made by dissecting individuals and examining the morphology in situ (e.g., Pisani, 1976; Siegel et al., 2011, 2012; Showalter et al., 2014), which does not provide a complete 3-D perspective of the examined objects. However, Granados et al. (2022) recently proposed a new method using silicone modeling, which we believe could be modified for the purposes of comparative research on snakes’ male and female genitalia. The silicone models can show the cloacal morphology in detail and reveal information about some of the main genital characteristics, such as the size and topology of the pouch, that might go unnoticed when the genitalia are described in situ. The models also present a clear 3-D perspective on the morphology of the main genital regions. When the genitals are filled with two-component condensation silicone, the filling first enters the cloaca and the pouch. Thus, by suppressing the posterior intestine and coprodaeum, the main part of the filling enters the pouch, and once it hardens, the rest of the silicone fills in the intestine. Since the uterus is narrower and presumably tight, we believe that silicone enters only the pouch region. Unlike Granados et al. (2022), we chose not to tie the oviducts or the intestine. While tying the intestine could seem advantageous – potentially preventing silicone from entering and hardening in a way that might alter the genitalia model – we opted against it to preserve the natural structure and dynamics of the system. In addition, the posterior parts of the oviducts are part of the pouch (Showalter, 2014), and it is speculated that they could have a role in the copulatory adjustment as well (Giacomini, 1893; Ludwig and Rahn, 1943). Therefore, tying the oviducts might lead to losing significant information. Thus, we believe that it is important to model the entire female genitalia, potentially revealing important insights for the male-female genitalia copulatory adjustment. By not tying the intestine and the oviducts, immediately after the cloaca, but further (see Methodology), we are also able to see the full three-dimensional morphology and topology of the organs in this part of the body. Silicone is a semi-liquid paste, and if it is under optimal levels of thickness, a portion of it could enter the intestine. Although this could be considered a disadvantage of the methodology, we believe that it can also reveal important information on the general morphology of the whole internal body region. Without such immediate isolation of the intestine, the general position of all of the organs can be examined (Fig. 2) similar to the lateral descriptions of internal organs made by Gabe and Saint-Girons (1965). We emphasize that the models are extracted from both old fixed museum specimens and recently found dead individuals, and soaking them in KOH cannot completely reverse the tissues to their original state. Still, we think that preserved individuals are suitable enough to be used for the purpose of the study. The lack of visible differences in the general female genital morphology between preserved and fresh specimens in this study corresponds to the results of Andonov et al. (2017), who report no major differences in the hemipenial morphology between old fixed individuals and freshly dead animals.
It should be noted also that the proposed approach reveals the general morphology of female genitalia, allowing comparative studies between sexes, as well as studies on the level of genital adjustment during copulation. However, for a precise investigation of the functionality of genital morphology, combined methodologies should be used (e.g., a combination of histological approach and macroscopic observation of silicone models).
Our study reveals that most of the models of Colubridae species have a deeper level of bifurcation and the pouch arms are longer compared to the base of the cloaca, whereas the pouches of Viperidae species are significantly shorter. The Eryx jaculus female genitalia has a different shape from the representatives of the other families. The female genitalia models in species with bilobed hemipenes are substantially less bifurcated, whereas genitalia models in species with simple or slightly bilobed hemipenes show a significant bifurcation and a highly conspicuous pouch (Fig. 3-9).
Although we only observed gross morphology and macroscopy morphology of female genitalia, our results partially confirm the conclusion made by Siegel et al. (2012), who state that snake female genitalia have pouches that bifurcate , and the level of separation and pouch length might differ. One small difference is that we observe a slight bifurcation in the Eryx jaculus vaginal pouch (Fig. 3), unlike Siegel et al. (2012), who did not report such bifurcation. We attribute it to the methodological differences, and this is an additional reason to further explore the maximum potential of the herein-proposed methodology.
An interesting trend we observed is that species with bilobed and divided male genitalia (V. ammodytes and V. berus) have a very slightly bifurcated vaginal pouch while the opposite is valid for species with slightly bilobed hemipenes (D. caspius, Z. longissimus, and E. quatuorlineata). We consider that this might reveal an important insight into the anatomy of the snake copulation and could provide information about genital adjustment during the act. In general, a close alignment of the male and female cloaca during copulation, allows the hemipenis to penetrate the cloaca (i.e., Pope, 1941; Pisani, 1976). In some species, this could also be supported by basal hemipenial spines, where they not only serve as an anchor but also help the male to open the female cloaca (Pisani 1976; Friesen et al. 2013). An additional erection of the hemipenis inside the female cloaca follows this opening. Ludwig and Rahn (1943) suggest that after successful penetration, the hemipenis distends and fully closes the vaginal pouches and cloaca, allowing sperm to be deposited by the most apical part of the sulcus spermaticus, based on a study on Crotalus viridis Rafinesque, 1818.
Only a few studies are commenting on the exact adjustment between the different types of species’ genitalia (i.e., Cope, 1898; Pope, 1941; Ludwig and Rahn, 1943; Inger and Marx, 1962). In Pope (1941), the author shows a perfect fit between hemipenis and female genitalia of Erythrolamprus poecilogyrus (Wied-Neuwied, 1824) during copulation. The hemipenis of this species is bilobed and each end of the bifurcated sulcus spermaticus points into each bifurcation of the vaginal pouch and touches the non-glandular uterus. Although not described in detail, from the picture provided, appears that the bifurcation of the pouch is not that prominent. A similar correlation is found in Calamaria lumbricoidea Boie, 1827 (Inger and Marx, 1962), although the conclusion is made after in situ description of female genitalia, therefore it is unclear what the actual genitalia alignment would look like during copulation. In addition, Inger and Marx (1962) find some interspecific variation as well. However, it should be mentioned that the authors study both adults and subadults, so the already described ontogenetic variation in hemipenes by Jadin and King (2012), might have affected the results. Edgren (1953) described a close alignment between the male hemipenis and female genitalia of Heterodon platirhinos Latreille, 1801, but not as aligned as Pope’s (1941) description. Edgren (1953) suggested that the unoccupied part of the area of the simple tall columnar epithelium of the cloaca is later filled with sperm and secretions that likely form a copulatory plug.
Our findings, based on silicone genitalia models, suggest that a similar mechanism might be relevant not only for species described by Pope (1941) and Inger and Marx (1962), but also for species with divided hemipenes, that we studied (V. ammodytes, and V. berus). We might assume, that the hemipenis of these species enters the cloaca during copulation, distends, and fully closes the vaginal pouches and cloaca and the tips of the two lobes touch the non-glandular uterus. Thus, having the divided sulcus spermaticus tightly surrounded by the female genitalia, the semen can be delivered to each oviduct easily with the lobes pointing towards them, as suggested by Ludwig and Rahn (1943).
We suggest that a similar adjustment can be observed in C. austriaca (Fig. 4), a species with bilobed hemipenes, and undivided sulcus spermaticus. Considering the elongation of the vaginal pouches, the lobes of the hemipenis might enter deeper into the pouches during copulation, and the semen is directed into one of the oviducts only, unlike the described adjustment in V. ammodytes and V. berus.
Having a simple or slightly bilobed hemipenis coupled with an undivided sulcus spermaticus is also observed (Andonov et al., 2017). Some of the species that we studied also have such hemipenial morphology (E. jaculus, D. caspius, Z. longissimus, E. quatuorlineata). If the copulation of these species follows the same mechanics and adjustment as for the species with bilobed hemipenis and divided sulcus spermaticus, a perfect closure of the vaginal pouch and cloaca will most likely not be possible and the semen might not be delivered to the pouch directly . This could lead to a reduction in the effectiveness of copulation. Therefore, another mechanism for effective semen delivery might be present. It is possible that during copulation those hemipenes do not constrain penetration to the proctodaeum and urodаeum but enter one of the vaginal pouch branches deeply, reaching the non-glandular uterus very closely. This could ensure successful copulation and semen delivery. An argument for that can be found in the morphology of the apical part of those simple and slightly bilobed hemipenes. All herein studied hemipenes lack any big and visible structures as part of the ornamentation of the apical part or bear just a few structures there. A similar correlation is discussed by Cope (1898), who observed that when the hemipenis is spiny, the cloacal walls are thick, and in cases when the male organ lacks ornamentation, the cloaca walls are thin. The latter is further confirmed by Pope (1941) with his observation on the genitalia of Trimeresurus albolabris Gray, 1842, and T. stejnegeri Schmidt, 1925. An additional argument for our hypothesis, regarding the non-bifurcated hemipenes, can be found by closely observing the genitalia of D. caspius. There is a very close resemblance between the hemipenial shape and one of the vaginal pouch branches, although more individuals should be examined, for making a solid conclusion. It should be added also that the asymmetry of the pouch might be a result of a non-well-inflated pouch (Fig. 4). Actually, the hypothesis that hemipenial lobes can enter the enlarged pouches has been already suggested (Giacomini, 1893; Ludwig and Rahn, 1943). Considering the species studied, we agree with it, but we believe that such penetration is much more likely to happen for species with simple or slightly bilobed hemipenes, rather than for those with divided copulatory organs.
Most of the studies investigate the hemipenial morphology, after removal of the organ. This causes some morphological changes and hinders complete morphological examination (i.e., a lot of the muscle tissue is removed). Thus, it is still uncertain to what extent the sulcus is closed when semen is transferred, and therefore additional methods for hemipenial observation should also be developed and applied, for example everting and inflating the hemipenis while still attached to the body, revealing its original topology. It should be also researched whether there is an additional function of the variable hemipenial morphology, such as stimulative. De-Lima et al. (2019) have explained the function of the deeply bilobed (divided) hemipenes (with structural folds on the apical tips) of the lizard species Tropidurus torquatus (Wied-Neuwied, 1820). They suggest stimulatory function, as the males stimulate the secretion of the female genitalia with the apical parts of the hemipenial lobes. The types of adjustment between snake genitalia could have similar functions as well, and not simply physical fit for semen transportation, but this is a matter that requires deeper investigation.
Our findings suggest that snake species may exhibit distinct patterns of genital alignment during copulation, with possible functional implications. We tentatively present different types of copulatory adjustment in species studied here as follows: adjustment in species with divided hemipenes and slightly bifurcated vaginal pouches, adjustment in species with bilobed hemipenes and highly bifurcated vaginal pouches, and species with simple or slightly bilobed hemipenes and slightly or highly bifurcated vaginal pouches. However, the limitations of the current study do not allow a definitive conclusion or categorization of the types of copulatory adjustment. Thus, more congeneric species should be studied for further confirmation of the herein-described adjustment types. We also assume that the copulation adjustment types are not limited to those observed in this study. It should be noted that there are also various species posing a simple hemipenis with divided sulcus. spermaticus such as Adelphostigma occipitalis (Jan, 1863), Xenopholis undulatus (Jensen, 1900), Dipsas jamespetersi (Orcés and Almendáriz, 1989), etc. (Zaher, 1999; Cadle, 2007). We hypothesize that this could be another group of species with potentially different adjustments, but further research is needed.
However, there are still only a few species examined with the used methodology, so deeper research is needed to either confirm or reject this hypothesis, which could either group genital fit types as a general rule or on the contrary, reveal a higher variation. Additionally, a higher representative sample is needed for statistical tests, because even though the individuals were very well preserved, internal changes due to decaying processes should not be excluded entirely and should be further investigated. The function and exact adjustment during copulation should also be further studied with more complementary methods (e.g., under high-resolution X-rays), preferably using a single-species approach (Arnqvist, 1997).
In conclusion, our study confirms the importance of the currently proposed silicone model technique for comprehensive analysis of snake genitalia, particularly focusing on the underexplored female cloaca. In addition, the current research proposes important additions to this methodology that might reveal more information about female genitalia morphology. The approach gives extensive insights into female genital morphology, making it a useful tool for comparative studies on male and female reproductive organs. Our findings, which address the scant knowledge of female snake genitalia, provide a groundwork for future research into copulatory adaptations and the co-evolution of male and female genitalia. Furthermore, our suggestion of an adjustment between male hemipenis shape and vaginal pouch bifurcation implies a possible link to snake copulation dynamics. We propose that the significant genital variations observed among the snake species studied here contribute to diverse copulation adjustments, with implications for successful semen delivery and the reproductive strategies of these species.
We thank Dr. Boyan Zlatkov (IBER-BAS, Sofia) for his guidance and helpful comments on the preprint. We thank Dr. Simeon Lukanov and Dr. Simeon Borissov for the English review and improvement of the text. We thank the National Museum of Natural History at the Bulgarian Academy of Sciences for providing the museum specimens. We thank two anonymous reviewers for their detailed and constructive comments and suggestions, which significantly improved the quality of the manuscript.
Supplementary material associated with this article can be found at <http://www-9.unipv.it/webshi/appendix/index.html> manuscript number 16881.
Ah-King, M., Barron, A.B., Herberstein, M.E. (2014): Genital evolution: why are females still understudied? PLOS Biol. 12: e1001851.
Andonov, K., Natchev, N., Kornilev, Y.V., Tzankov, N. (2017): Does sexual selection influence ornamentation of hemipenes in Old World snakes? Anat. Rec. 300: 1680-1694.
Arnqvist, G. (1997): The evolution of animal genitalia: distinguishing between hypothesis by single species studies. Biol. J. Linn. Soc. 60: 365-379.
Arnqvist, G. (1998): Comparative evidence for the evolution of genitalia by sexual selection. Nature 393: 784-786.
Biserkov, V., Naumov, B., Tsankov, N., Stoyanov, A., Petrov, B., Dobrev, D., Stoev, P. (2007): Guide to the amphibians and reptiles in Bulgaria. Sofia, Green Balkans.
Blackburn, D.G. (1998): Structure, function, and evolution of the oviducts of squamate reptiles, with special reference to viviparity and placentation. Jour. of Exp. Zool. 282: 560-617.
Brennan, P.L., Prum, R.O. (2015): Mechanisms and evidence of genital coevolution: The roles of natural selection, mate choice, and sexual conflict. Cold Spring Harb. Perspect. Biol. 7: a017749.
Brennan, P.L. (2022): Evolution and morphology of genitalia in female amniotes. Integr. Comp. Biol. 62: 521-532.
Cadle, J. (2007): The snake genus Sibynomorphus (Colubridae: Dipsadinae: Dipsadini) in Peru and Ecuador, with comments on the systematics of Dipsadini. Bull. Mus. Comp. Zool. 158: 183-283.
Cohn, M.J. (2011): Development of the external genitalia: conserved and divergent mechanisms of appendage patterning. Dev. Dyn. 240: 1108-1115.
Cope, E.D. (1895): The classification of the Ophidia. Trans. Amer. Philos. Soc. 18: 186-219.
Cope, E.D. (1898): The Crocodilians, Lizards, and Snakes of North America. Kept. US Nat. Mus. 1898: 153-1270.
Darwin, C. (1871): The descent of man and selection in relation to sex. Modern Library, New York.
De-Lima, A.K.S., Paschoaletto, I.P., Pinho, L.dO., Benmamman, P., Klaczko, J. (2019): Are hemipenial traits under sexual selection in Tropidurus lizards? Hemipenial development, male and female genital morphology, allometry and coevolution in Tropidurus torquatus (Squamata: Tropiduridae). PLOS ONE 14: e0219053.
Dowling, H.G., Savage, J.M. (1960): A guide to the snake hemipenis: a survey of basic structure and systematic characters. Zool. 45: 17-28.
Dufour, L. (1844): Anatomie générale des diptères. Ann. Sci. Nat. 1: 244-264.
East, M.L., Hofer, H., Wickler, W. (1993): The erect ‘penis’ is a flag of submission in a female-dominated society: greetings in Serengeti spotted hyenas. Behav. Ecol. Sociobiol. 33: 355-370.
Eberhard, W.G. (1985): Sexual selection and the evolution of animal genitalia. Harvard University Press, Cambridge, MA.
Eberhard, W.G. (2010): Evolution of genitalia: theories, evidence, and new directions. Genetica 138: 5-18.
Edgren, R.A. (1953): Copulatory adjustment in snakes and its evolutionary implications. Copeia 1953: 162-164.
Friesen, C.R., Uhrig, E.J., Squire, M.K., Mason, R.T., Brennan, P.L.R. (2013): Sexual conflict over mating in red-sided garter snakes (Thamnophis sirtalis) as indicated by experimental manipulation of genitalia. P. Roy. Soc. B. Biol. Sci. 281: 20132694.
Gabe, M., Saint-Girons, H. (1965): Contribution à la morphologie comparée du cloaque et des glandes épidermoïdes de la région cloacale chez les lépidosauriens. Mém. Mus. Hist. Nat. XXXIII: 149-332
Giacomini, E. (1893): Sull’ovidutto del Sauropsidi. Monit. Zool. Ital. 4: 202-265.
Granados, G.L., Greenwood, L., Secor, S., Shan, S., Hedrick, B.P., Brennan, P.L. (2022): Examining the shape and size of female and male genitalia in snakes using three-dimensional geometric morphometrics, Biol. J. Linn. Soc. 136: 466-476.
Gredler, M.L., Larkins, C.E., Leal, F., Lewis, A.K., Herrera, A.M., Perriton, C.L., Sanger, T.J., Cohn, M.J. (2014): Evolution of external genitalia: insights from reptilian development. Sex. Dev. 8: 311-326.
Hosken, D.J., Stockley, P. (2004): Sexual selection and genital evolution. Trends. Ecol. Evol. 19: 87-93.
Inger, R.F., Marx, H. (1962): Variation of hemipenis and cloaca in the colubrid snake Calamaria lumbricoidea. Syst. Zool. 11: 32-39.
Jadin, R.C., King, R.B. (2012): Ontogenetic effects on snake hemipenial morphology. J. Herpetol. 46: 393-395.
Jurkfitz, R.C., Silva, K.M.P., Almeida-Santos, S.M. (2023): Sperm storage in Crotalus durissus (Serpentes: Crotalinae): histological insights about the female reproductive tract of pit vipers. Zoomorphology, 142: 487-496.
Keogh, J.S. (1999): Evolutionary implications of hemipenial morphology in the terrestrial Australian elapid snakes. Zool. J. Linn. Soc. Lond. 125: 239-278.
King, R.B., Jadin, R.C., Grue, M., Walley, H.D. (2009): Behavioural correlates with hemipenis morphology in New World natricine snakes. Biol. J. Linn. Soc. 98: 110-120.
Langerhans, R.B., Anderson, C.M., Heinen-Kay, J.L. (2016): Causes and consequences of genital evolution. Integr. Comp. Biol. 56: 741-751
Ludwig, M., Rahn, H. (1943): Sperm storage and copulatory adjustment in the prairie rattlesnake. Copeia 1943: 15-18.
Mayr, E. (1963): Animal species and evolution. Cambridge, Mass: Harvard University Press.
Moore, B.C., Kelly, D.A., Piva, M., Does, M., Kim, D.K., Simoncini, M., Leiva, P.M.L., Pina, C.I. (2022): Genital anatomy and copulatory interactions in the broad snouted Caiman (Caiman latirostris). The. Anat. Rec. 305: 3075-3087.
Myers, C.W., McDowell, S.B. (2014): New taxa and cryptic species of neotropical snakes (Xenodontinae), with commentary on hemipenes as generic and specific characters. Bull. A. Mus. Nat. His. 385: 1-112.
Orbach, D.N., Hedrick, B.P., Würsig, B., Mesnick, S.L., Brennan, P.L.R. (2018): The evolution of genital shape variation in female cetaceans. Evolution 72: 261-273
Orbach, D.N., Brassey, C.A., Gardiner, J.D., Brennan, P.L.R. (2021): 3D genital shape complexity in female marine mammals. Ecol. Evol. 11: 3210-3218.
Pesantes, O.S. (1994): A method for preparing the hemipenis of preserved snakes. J. Herpetol. 28: 93-95.
Pisani, G.R. (1976): Comments on the courtship and mating mechanics of Thamnophis (Reptilia, Serpentes, Colubridae). J. Herpetol. 10: 139-142.
Pope, C.H. (1941): Copulatory adjustment in snakes. Fieldiana Zool. 24: 149-152.
Shapiro, A.M., Porter, A.H. (1989): The lock-and-key hypothesis: evolutionary and biosystematic interpretation of insect genitalia. Annu. Rev. Entomol. 34: 231-245.
Showalter, I., Todd, B.D., Brennan, P.L.R. (2014): Intraspecific and interspecific variation of female genitalia in two species of watersnake. Biol. J. Linn. Soc. 111: 183-191.
Siegel, D.S., Miralles, A., Chabarria, R.E., Aldridge, R.D. (2011): Female reproductive anatomy: cloaca, oviduct, and sperm storage. Chapter 9. In: Reproductive biology and phylogeny of snakes, pp 347-409. Aldridge R., Sever D.M. Eds, Boca Raton, Florida, CRC Press.
Siegel, D.S., Miralles, A., Trauth, S.E., Aldridge, R.D. (2012) The phylogenetic distribution and morphological variation of the ‘pouch’ in female snakes. Acta Zool. 93: 400-408.
Stojanov, A., Tzankov, N., Naumov, B. (2011): Die Amphibien und Reptilien Bulgariens. Chimaira, Frankfurt am Main.
Thornhill, R. (1983): Cryptic female choice and its implications in the scorpionfly Harpobittacus nigriceps. Am. Nat. 122: 765-788.
Ware, A., Opell, B.D. (1989): A test of the mechanical isolation hypothesis in two similar spider species. J. Arachnol. 17: 149-162.
Zaher, H. (1999): Hemipenial morphology of the South American xenodontine snakes: with a proposal for a monophyletic Xenodontinae and a reappraisal of colubroid hemipenes. Bull. Am. Mus. Nat. 240: 1-168.
Zaher, H., Prudente, A.L.C. (2003): Hemipenes of Siphlophis (Serpentes, Xenodontinae) and techniques of hemipenial preparation in snakes: a response to Dowling. Herpetol. Rev. 34: 302-307.