Submitted on: 2024, 11th November; revised on: 2025, 11th May; accepted on: 2025, 23rd July
Spatial and temporal variation in detection probability and abundance of freshwater turtles in a peri-urban area of Midwestern Brazil
1 Programa de Capacitação Institucional, Instituto Nacional de Pesquisas do Pantanal, Av. Fernando Correa da Costa 2367, 78060-900 Cuiabá, Mato Grosso, Brazil
2 Laboratório de Herpetologia, Centro de Biodiversidade, Universidade Federal de Mato Grosso, Av. Fernando Correa da Costa 2367, 78060-900 Cuiabá, Mato Grosso, Brazil
3 Programa de Pós-graduação em Biologia Animal, Instituto de Biociências, Universidade Federal do Mato Grosso do Sul, Av. Costa e Silva 8000, 79070-900 Campo Grande, Mato Grosso do Sul, Brazil
4 Departamento de Ciências Biológicas, Universidade Católica Dom Bosco, Av. Tamandaré 6000, 79117-900 Campo Grande, Mato Grosso do Sul, Brazil
5 Instituto de Biociências, Letras e Ciências Exatas, Universidade Estadual Paulista, Rua Cristóvão Colombo 2265, 15054-000 São José do Rio Preto, São Paulo, Brazil
6 Faculdade de Medicina Veterinária, Universidade Federal de Mato Grosso, Av. Fernando Correa da Costa 2367, 78060-900 Cuiabá, Mato Grosso, Brazil
*Corresponding author. Email: eliz.chelidae@gmail.com
Abstract. This study assessed spatial (across water bodies) and temporal (over months and years) variation in the detection probability and abundance of freshwater turtles, along with the effects of climatic variables such as precipitation and temperature. Fieldwork was conducted in four water bodies located at the headwaters of a stream in the peri-urban area of Campo Grande, Mato Grosso do Sul, in Midwestern Brazil. Over 38 months of sampling (2014–2017), we captured 94 individuals: 73 Phrynops geoffroanus, 19 Mesoclemmys vanderhaegei, and two Trachemys dorbigni. The detection probability of M. vanderhaegei was influenced by both precipitation and temperature, whereas detection of P. geoffroanus was affected only by temperature. The abundance of both species varied across water body types and sampling months, with P. geoffroanus being more abundant in water bodies where M. vanderhaegei was less common. P. geoffroanus reached its highest abundance during the rainy season (November to March), with fluctuations in the subsequent months. In contrast, M. vanderhaegei maintained low abundance throughout most of the year, with a peak in December. To our knowledge, this is the first study to examine population patterns of these two syntopic species. Phrynops geoffroanus, the more abundant species, is often recorded in anthropogenically altered environments and appears to possess greater competitive ability than M. vanderhaegei, which is less abundant and likely more sensitive to environmental degradation. Despite the urban context and the presence of an invasive species (T. dorbigni), the occurrence of M. vanderhaegei suggests that the study area may still retain important elements of habitat quality.
Keywords. Chelidae, chelonians, Testudines, urban ecology, Phrynops geoffroanus, Mesoclemmys vanderhaegei, Trachemys dorbigni.
Index
Characterization of the water bodies
Knowledge about chelonian ecology is still incipient in South America. Population studies are now more frequent, but studies on the structure of assemblages or on the temporal partition of resources of the different species of freshwater turtles are rare, even in the Amazon basin, where the diversity of these reptiles high (Mittermeier et al., 2015). This highlights the challenge of conducting studies on fr∂eshwater turtles in natural environments.
Due to their ease of access, water bodies in urban areas can be considered excellent sites for obtaining information on different aspects of the ecology of freshwater turtles (Rocha et al., 2025; Dorado-Rodrigues et al., 2024). On the other hand, urbanization imposes significant pressure on wildlife, altering natural environments through deforestation, which leads to vegetation fragmentation, species loss, and biodiversity decline (Guzy et al., 2013; Liu et al., 2016; Rocha et al., 2025). Nevertheless, many turtles tolerate, adapt to, and survive in urbanized environments, even those of poor quality, as long as they have access to resources essential for their survival (e.g., food, shelter, and reproductive sites) (Gibbons et al., 2000; Luiselli, 2003; Stokeld et al., 2014; Spencer et al., 2018; Rocha et al., 2025).
Freshwater turtles inhabit different types of habitats, terrestrial and aquatic, which vary according to their basic ecological needs as a function of their semi-aquatic habitat (Bodie and Semlitsch, 2000). Terrestrial and aquatic habitats (whether lentic or lotic) undergo changes due to variations promoted by the hydrological cycle which, in turn, affect the availability of resources, and consequently the quality of the habitat (Alho and Pádua, 1982; Fachín-Terán et al., 2006). As a result of these environmental variations, freshwater turtles adjust their preexisting strategies – such as feeding, thermoregulation, and reproduction – to habitat alterations to ensure their survival (Moll and Moll, 2004). Thus, habitat use by freshwater turtles may vary according to periods of drought and rainfall, since these aspects directly influence the spatial distribution and structure of assemblages by affecting the distribution and supply of resources in the environment (Plummer et al., 1997; Luiselli, 2008).
In this study, we evaluated the effects of population parameters, such as detectability and abundance, for two species of freshwater turtles in relation to temporal (sampling period, rainfall, and temperature) and spatial (water bodies) characteristics. We tested the following hypotheses: (1) the detection probability of freshwater turtles varies with monthly precipitation and temperature; (2) The abundance of freshwater turtles varies spatially among water bodies and temporally with sampling months and temperature, as well as with the interaction between water bodies and sampling months; and (3) the abundance of a species varies with the area of the water body and/or the abundance of syntopic species.
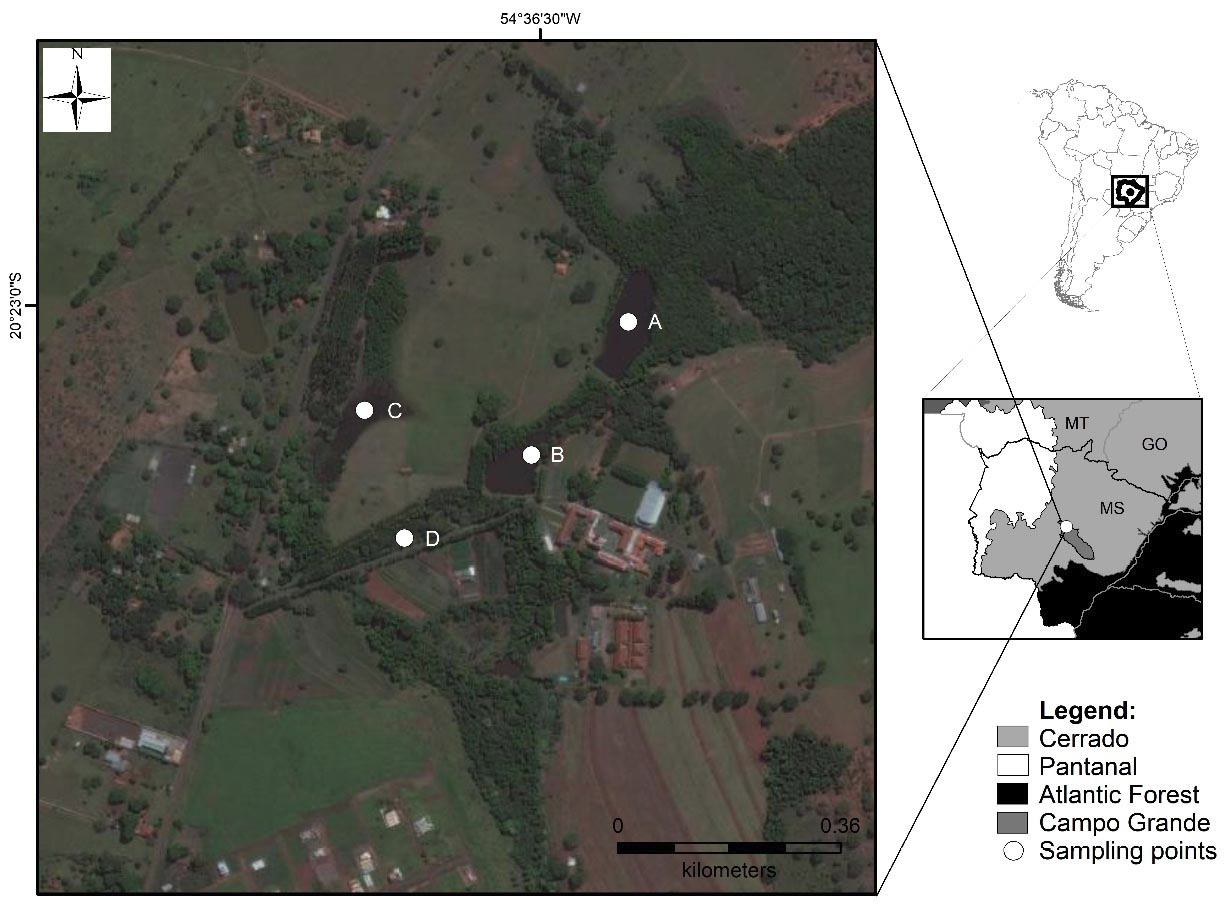
The study area is located in the urban perimeter of the municipality of Campo Grande (Mato Grosso do Sul, Midwestern Brazil), situated in the Cerrado biogeographic domain and the Upper Paraguay river basin (20°23’00”S and 54°36’00”W; Datum WGS84; Fig. 1). The study was conducted in a vegetation fragment of 191 hectares of which 20 hectares are designated for the protection area called legal reserve in Brazil (Cheung et al., 2016; Corral et al., 2021). The fragment is located in an area of urban expansion and is part of a region that includes both forest and open formations. The forest formations range from cerradão – a dense forest formation characterized by tall trees (8–15 m), a closed canopy, a sparse grassy understory, and occurrence in fertile, humid soils – to riparian vegetation, including gallery and riparian forests (Sano et al., 2008; Cheung et al., 2016). The open formations include cerrado stricto sensu and campo sujo. Campo sujo refers to herbaceous-shrubby habitats dominated by grasses, with few trees and shrubs (<2% of the land cover; Furley, 1999).
The climate in the region is of Aw type (tropical climate with a dry winter season), according to the Köppen’s classification (Peel et al., 2007). The rainy (October to March) and dry (April to September) seasons are well defined (see Marcuzzo and Mello, 2011). The average annual temperature is 24°C and the annual rainfall is between 1800 and 2000 mm.
We sampled four water bodies located at the headwaters of the Segredo stream in a landscape with fragmented Cerrado vegetation (Fig. 1). We collected information on the freshwater turtle assemblage between March 2014 and July 2017, using funnel traps 1.2 m in length (see description in Brito et al., 2009; 2018; Balestra et al., 2016). The traps were baited with ground beef and remained in the water diurnally, being reviewed once a day over five consecutive days per month. The number of traps ranged from two to four traps per water body (\). The minimum distance between traps within the same water body was 100 meters, while the maximum distance varied up to 150 meters in water body A, the largest of the four. The traps were installed along the margins of each water body.
| Water bodies | Type | Habitat | Area (m2) | Surrounding matrix | Vegetation cover | Substrate | Funnel traps |
|---|---|---|---|---|---|---|---|
| A | Dam | Lentic | 7543,6 | Riparian forest | Native arboreous plants | Sandy | 4 |
| B | Dam | Lentic | 9171,6 | Riparian forest with exotic vegetation | Exotic arboreous plants | Sandy | 4 |
| C | Dam | Lentic | 8089,1 | Grassland flooded | Herbaceous and aquatic plants | Clay | 2 |
| D | Lagoon | Lentic | 6542,1 | Riparian forest | Native arboreous plants | Sandy | 2 |
Considering the possibility of imperfect detection, we performed five repeated samplings per month (five consecutive days), necessary to estimate the probability of detection. Since each species could be detected (1) or not detected (0), we constructed a detection history denoting the sequence of detections and non-detections for each species (e.g., 10100 - five sampling occasions within a session, where the species was detected on two and not detected on three). The assumption was that the site occupancy status for each species did not change during the period, or that the changes occurred completely at random (i.e., the species present at the sites were constant during sampling) (MacKenzie et al., 2006).
We identified, marked, sexed, and performed biometrics on all of the captured turtles. We marked each captured turtle individually using a system of rectangular cuts in marginal scutes of the carapace, adapted from Cagle (1939). We determined the sex of the captured individuals by examining secondary sexual characteristics (males having a more elongated tail than females) and measured carapace length (CL; to the nearest 0.05 mm) with a 150 mm or 300 mm Vernier caliper. The sexed individuals were classified as either female or male, but their sexual maturity status was not determined, as this information is unknown for M. vanderhaegei and P. geoffroanus. We could not determine the sex of some individuals smaller than 100 mm CL for both species investigated. Therefore, we classified them as juveniles. We obtained the body mass with Pesola® spring balances of the following capacities: 2.5 kg (50g precision) and 5.000 g (100g precision).
Finally, we obtained information on monthly precipitation and temperature from the INMET (National Institute of Meteorology) database obtained from the meteorological station in Campo Grande, Mato Grosso do Sul (20°27’00”S; 54°43’12”W), located 25 km from the study area.
Characterization of the water bodies
Among the four water bodies sampled at the headwaters of the Segredo stream, three are artificial dams (A, B and C) and one is a natural pond (D) that remained with water during the entire study period. The area of the water bodies, estimated using Google® Earth Pro software, ranged from 6,542 m² to 9,171 m², are located in the Cerrado. The surrounding matrix is formed by floodable riparian forest and campo sujo, with tree and shrub vegetation, also containing exotic plant species (e.g., bamboo, eucalyptus, and mango trees). The margin and the interior of the water bodies present vegetation cover by herbaceous and aquatic plants and the substrate is sandy or clayey (Table 1). Despite being located in an area of urban expansion, none of the water bodies receive sewage effluents.
Sex ratio. We used a chi-square test to determine whether the sex ratio differed from 1:1 in each of the turtle populations, employing only those individuals sexed and captured for the first time.
Detectability, temporal, and spatial variations. To assess the probability of detection of each species of freshwater turtles, we used the “single-season, single-species” occupancy model, which explicitly accounts for imperfect detection [ψ(.)ρ(.)]. Since our goal is focused on comparing only how much the detection pattern changes according to the temporal characteristics of rainfall and daily temperature occurrence, the detection probability was estimated without the effect of environmental variables on occupancy (ψ Näive). Thus, we tested the effect of rainfall occurrence and daily temperature on the detection probability (from the history of detection [1] and non-detection [0]), considering the other parameters constant [ψ(.)ρ(variable)] (MacKenzie et al., 2006). We considered the most parsimonious detection probability model, with the smoothing method with 100 Bootstrap interactions (to compute the standard error of the estimated derived parameters) (MacKenzie et al., 2006). We obtained the parameter estimates by means of maximum likelihood estimation (MLEs).
We used AIC (Akaike Information Criterion; Burnham and Anderson, 2002) to rank and compare the candidate models for each species. We selected the best models (i.e., the model with the parameter that best describes the data as a function of the variable) by considering those with ΔAIC values ≤ 2. We examined the relevance of the variables in the best selected models for uncertainty from analysis of the 95% confidence interval of the parameter estimates (i.e., the overlap of the confidence interval to zero).
We assessed the normality of species abundance data in relation to water bodies and climatic variables using the Shapiro-Wilk test. To evaluate variations in freshwater turtle abundance (by species and total) in relation to temporal (month sampled, mean daily temperature) and spatial (water bodies) characteristics, we fitted Generalized Linear Mixed Models (GLMMs). The months sampled, mean daily temperature and water bodies, as well as the interaction between them, were considered to be fixed factors. To assess whether species abundance varied across sampling months and between water bodies, random factors, we fitted Generalized Linear Mixed Models (GLMMs) separately for each species and treated fixed and random factors in the same way. Due to the overdispersion in abundance data, we used the quasi-Poisson distribution, which has a better fit to data with inflated variance (Hoef and Boveng, 2007).
To assess whether the abundance of a freshwater turtle is affected by the abundance of another species and whether it varies with the area of the water bodies, we used Generalized Linear Models (GLMs). We evaluated the variation for each sampling year (2014, 2015, 2016, 2017) and considering the total catches taken over the four years. Generalized Linear Models present a major advantage over classical multiple regression approaches in that they integrate data from different statistical distributions with the appropriate modeling of statistical error. We verified by means of the Shapiro-Wilk test the absence of overdispersion in the abundance data, and thus we used the Poisson distribution to estimate abundance.
All statistical analyses were performed in the R 3.6.0 programming environment (R Development Core Team, 2019), using the Occu function in the unmarked package (Fiske and Chandler, 2011) in the AIC analysis, and the MASS package (Venables and Ripley, 2002) for the GLMMs analyses (Venables and Ripley, 2002). We adopted a significance level of P < 0.05.
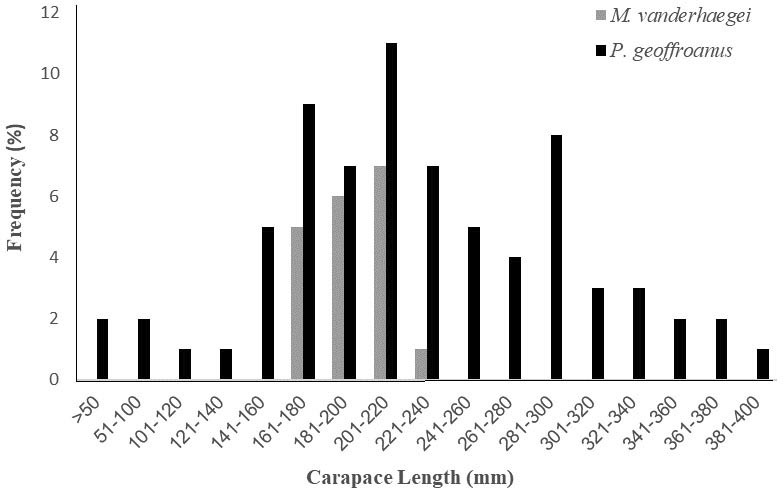
We recorded three species of freshwater turtles in the four water bodies sampled: two native (Phrynops geoffroanus and Mesoclemmys vanderhaegei) and one introduced species (Trachemys dorbigni). In all, we captured 94 individuals: 73 were P. geoffroanus (78% of the total; 31 females, 30 males, and five juveniles), 19 M. vanderhaegei (20% of the total; 10 females, nine males, and one juvenile) and two T. dorbigni (2% of the total). Both T. dorbigni individuals were adults (156 and 190 mm CL), but their sexes were not determined. Sex ratio did not differ from 1:1 for either P. geoffroanus (0.96M:1F; x2 = 0.016, df = 1, P = 0.898) or M. vanderhaegei (1M:1F; x2 = 0, df = 1, P = 1.000). Juveniles represented a small proportion of captures for both P. geoffroanus and M. vanderhaegei (Fig. 2). Of the 94 individuals captured and marked, we recorded 49 (52%) recapture events – 55% involving P. geoffroanus and 47% involving M. vanderhaegei. We did not record recaptures of T. dorbigni (Table 2).
| Sampled water bodies | Phrynops geoffroanus | Mesoclemmys vanderhaegei | Trachemys dorbigni |
|||
|---|---|---|---|---|---|---|
| Capture | Recapture | Capture | Recapture | Capture | Recapture | |
| A | 23 | 10 | 3 | 2 | 0 | 0 |
| B | 23 | 21 | 0 | 2 | 0 | 0 |
| C | 2 | 0 | 14 | 3 | 0 | 0 |
| D | 25 | 9 | 2 | 2 | 2 | 0 |
| Total | 73 | 40 | 19 | 9 | 2 | 0 |
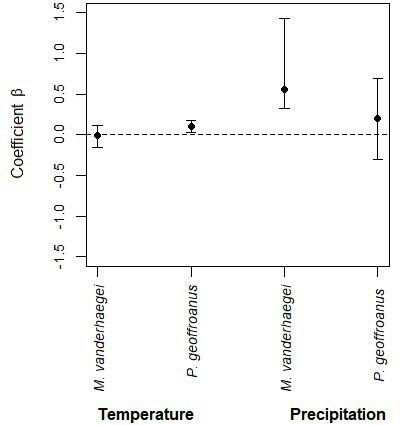
The detection probability of Mesoclemmys vanderhaegei did not include precipitation or temperature in the best model. However, the second-best model, which included precipitation (ΔAIC = 0.05), and the third-best model, which incorporated temperature (ΔAIC = 1.99), were equally informative for the species. On the other hand, Phrynops geoffroanus showed a higher detection probability only with increasing temperature, as both the naïve model and the model including precipitation had ΔAIC > 3, indicating lower support compared to the temperature model (ΔAIC = 0; Table 3; Fig. 3).
| Taxon | K | AIC | ΔAIC | AICw | Beta |
|---|---|---|---|---|---|
| Mesoclemmys vanderhaegei | |||||
| p()Ψ() | 2 | 198.54 | 0 | 0.46 | -3.62 |
| p(prec) Ψ() | 3 | 199.04 | 0.5 | 0.36 | 0.56 |
| p(temp) Ψ() | 3 | 200.53 | 1.99 | 0.17 | -0.0088 |
| Phrynops geoffroanus | |||||
| p(temp) Ψ() | 3 | 490.21 | 0 | 0.756 | 0.103 |
| p() Ψ() | 2 | 493.44 | 3.22 | 0.151 | -2.3 |
| p(prec) Ψ() | 3 | 494.41 | 4.2 | 0.093 | 0.264 |
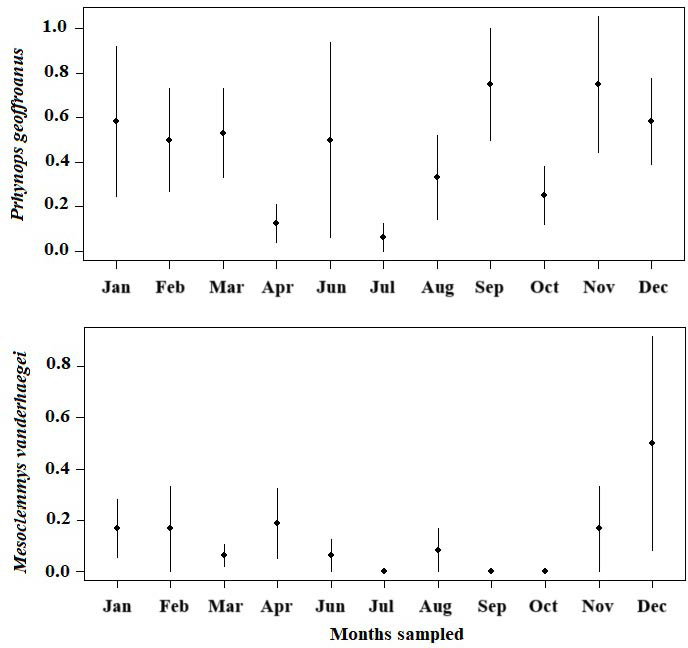
The abundance of M. vanderhaegei and P. geoffroanus varied among water bodies, across sampling months, and also considering the interaction between these two factors (Table 4; Fig. 4, 5). However, the average daily temperature influenced abundance only for M. vanderhaegei (X² = 22.676, df = 2, P < 0.001; Table 4). When both species were modeled together, the average daily temperature also did not have a significant effect on species abundance (X² = 3.3153, df = 1, P = 0.068; Table 4). The variation in abundance over the sampling months was different for P. geoffroanus and M. vanderhaegei, with high capture rates in the wettest and warmest months of the year for P. geoffroanus (November, December, January, February, and March) (Fig. 4). For both species, no new individuals were captured in May (Fig. 4).
| Variable | df | X2 | P |
|---|---|---|---|
| Abundance of Phrynops geoffroanus | |||
| Water bodies | 3 | 3458.4617 | <0.001 |
| Month | 11 | 1123.1046 | <0.001 |
| Average temperature | 1 | 7.2644 | 0.007 |
| Interaction water body*month | 33 | 4384.3083 | <0.001 |
| Abundance of Mesoclemmys vanderhaegei | |||
| Water bodies | 3 | 12840.363 | <0.001 |
| Month | 11 | 11455.841 | <0.001 |
| Average temperature | 2 | 22.676 | <0.001 |
| Interaction water body*month | 33 | 44687.955 | <0.001 |
| Both species | |||
| Water bodies | 3 | 527.0880 | <0.001 |
| Month | 11 | 1150.3408 | <0.001 |
| Average temperature | 1 | 3.3153 | 0.068 |
| Interaction water body*month | 33 | 5100.7831 | <0.001 |
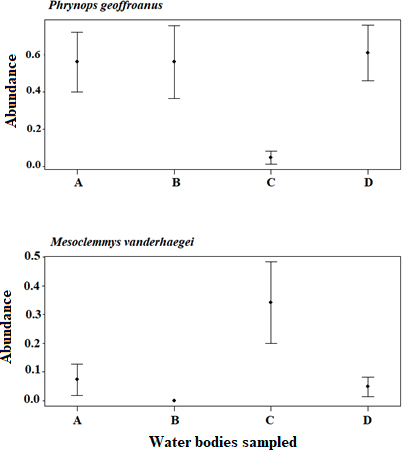
When analyzing, year by year (2014, 2015, and 2017), whether the water bodies (A, B, C, and D) and the abundance of M. vanderhaegei affected the abundance of P. geoffroanus, we observed a significant influence of the area only in 2014 (X² = 1.17, df = 3, P < 0.001). However, when all years were analyzed together, the sampled area did not affect the abundance of P. geoffroanus, which was significantly associated only with the abundance of M. vanderhaegei (X² = -0.15, df = 3, P < 0.001; Table 5). Conversely, when examining whether the abundance of P. geoffroanus or the area affected the abundance of M. vanderhaegei, we found that both the area (X² = 2.74, df = 3, P = 0.01) and the abundance of P. geoffroanus (X² = -0.34, df = 3, P = 0.01) influenced M. vanderhaegei in 2014, but not in the other years when analyzed individually. When all years were considered together, the area did not significantly affect the abundance of M. vanderhaegei (X² = -0.84, df = 3, P = 0.18), which was influenced exclusively by the abundance of P. geoffroanus (X² = -0.12, df = 3, P < 0.001; Table 5). Abundance was not estimated for 2016, as no individuals of M. vanderhaegei were recorded that year. Although the area was not significantly related to the abundance of either species, graphical analysis showed a trend: P. geoffroanus tended to be more abundant in water bodies with fewer individuals of M. vanderhaegei, and similarly, M. vanderhaegei was more abundant where P. geoffroanus occurred at lower abundance (Figure 5).
| Year | Variable | df | X2 | P |
|---|---|---|---|---|
| Abundance of Phrynops geoffroanus | ||||
| 2014 | Water body area | 3 | 1.17 | <0.001 |
| Mesoclemmys vanderhaegei abundance | 3 | 0.02 | 0.81 | |
| 2015 | Water body area | 3 | 0.31 | 0.36 |
| Mesoclemmys vanderhaegei abundance | 3 | -0.06 | 0.46 | |
| 2017 | Water body area | 3 | -0.66 | 0.16 |
| Mesoclemmys vanderhaegei abundance | 3 | 0.03 | 0.92 | |
| Total | Water body area | 3 | -0.19 | 0.10 |
| Mesoclemmys vanderhaegei abundance | 3 | -0.15 | <0.001 | |
| Abundance of Mesoclemmys vanderhaegei | ||||
| 2014 | Water body area | 3 | 2.74 | 0.01 |
| Phrynops geoffroanus abundance | 3 | -0.34 | 0.01 | |
| 2015 | Water body area | 3 | 0.39 | 0.70 |
| Phrynops geoffroanus abundance | 3 | -0.15 | 0.22 | |
| 2017 | Water body area | 3 | 0.18 | 0.79 |
| Phrynops geoffroanus abundance | 3 | -0.06 | 0.77 | |
| Total | Water body area | 3 | -0.84 | 0.18 |
| Phrynops geoffroanus abundance | 3 | -0.12 | <0.001 | |
In the present study, we found a higher abundance of Phrynops geoffroanus compared to Mesoclemmys vanderhaegei. The abundance of P. geoffroanus was higher from November to March, coinciding with the rainy season, while M. vanderhaegei showed a peak in abundance in December and remained at low levels during the other months. The probability of detection was influenced by climatic factors such as precipitation and temperature, but the effect differed between the two most abundant species. Overall, considering all sampling years, the abundance of the two species varied in relation to each other; however, water bodies and sampling months also influenced the individual abundance of each species.
Phrynops geoffroanus was the dominant species in the study. This pattern was expected, since P. geoffroanus seems to be more adapted to urban environments (e.g. Souza and Abe, 2000; Souza et al., 2008). Phrynops geoffroanus does not appear to be selective regarding habitat quality, using both lentic and lotic water bodies of various sizes (Souza et al., 2008; Magalhães et al., 2015; Silva et al., 2016; Rocha et al., 2025). Mesoclemmys vanderhaegei, on the other hand, is often found in oligotrophic, small, lentic and lotic water bodies, in conserved and higher altitude environments (Brito et al., 2009, 2018; Maffei et al., 2016) and, more rarely, in anthropic environments (Marques et al., 2013, 2014).
Precipitation and temperature affected the detection probability of M. vanderhaegei, while for P. geoffroanus, only temperature influenced its detection probability. However, when abundance was considered, average temperature affected only M. vanderhaegei. Freshwater turtles usually respond to climatic variables linked to temperature and humidity (Moll and Moll, 2004; Souza, 2004). Both variables are considered equally important for turtles, as demonstrated in other studies, such as those on flooding cycles for Podocnemis expansa (Eisemberg et al., 2016), or temperature for activity of the turtles in general (Plummer, 2003; Pittman and Dorcas, 2009; Tamplin and Cyr, 2011). Chelonians are ectothermic animals that depend directly on the temperature of the environment to regulate their internal temperature. Some species are more active when the environment is warmer (Tamplin and Cyr, 2011) or, less active during warmer periods of the day to avoid overheating (Hailey and Coulson, 1996). Our study showed that, although temperature and rainfall are equally important for turtle species, different responses to these variables may occur depending on the species analyzed. Additionally, it is important to note that our study area is located in a tropical Aw climate zone, characterized by consistently high temperatures and minimal seasonal variation, with dry winters. Therefore, other environmental factors – such as aquatic habitat type and resource availability – may play a more significant role in determining species abundance.
Phrynops geoffroanus was more abundant in water bodies with lower abundances of M. vanderhaegei, particularly in water bodies A, B, and D. In contrast, M. vanderhaegei reached its highest abundance in water body C, where only a few individuals of P. geoffroanus were captured. Behavioral differences, resource use, or morphological traits between the two species may lead to variation in habitat use. Larger species, such as P. geoffroanus, generally require more resources and tend to occupy larger home ranges (Perry and Garland, 2002; Jaeger and Cobb, 2012; Müller et al., 2019). Although the total area of these water bodies is relatively similar, they may vary in depth, resource availability, and degree of eutrophication. For instance, M. vanderhaegei is frequently recorded in small, oligotrophic water bodies, whereas P. geoffroanus appears to be less selective in habitat use, occupying a variety of aquatic environments (Brito et al., 2009, 2018; Souza and Abe, 2000, 2001). However, P. geoffroanus is more commonly found in eutrophic rather than oligotrophic systems (Souza and Abe, 2000; Souza et al., 2008; Ferronato et al., 2009; Rocha et al., 2025). In addition, both species are omnivorous, with a tendency toward carnivory, feeding on aquatic insects, fish, and amphibians (Martins et al., 2010; Brito et al., 2016). In general, species tend to partition resources to coexist, and omnivores generally feed opportunistically, according to food availability. However, interspecific competition can determine exclusion through aggressive interactions or through the exploitation of resources with higher protein content by more competitive species (Luiselli, 2008).
Finally, the presence of Trachemys dorbigni does not seem to be a problem for native species yet, due to its low abundance. However, it is an exotic species, found outside its natural range, and with great potential for dispersal and adaptation, especially in human-altered environments (Rachemys, 2009; Molina et al., 2016; Ciccheto et al., 2018; Fonseca et al., 2021). Trachemys dorbigni occurs naturally in Brazil (Rio Grande do Sul), Uruguay, and Argentina, inhabiting large floodplains (Alcalde et al., 2012). If its population increases outside its natural range, it can quickly occupy the niche of native species, leading to a decrease in native species abundance or even local extinction if population control is not taken (Zalba and Ziller, 2007). Exotic species usually have greater niche width (Pérez-Santigosa et al., 2011), competing directly with native species and affecting the structure of assemblages with changes in the foraging process and the dissemination of pathogens (Lowe et al., 2000; Zalba and Ziller, 2007). In the case of Phrynops geoffroanus, a recent study conducted on a turtle assemblage in an urban area in southern Brazil indicates that the species may alter its activities in response to the coexistence with invasive turtles, such as Trachemys dorbigni and T. scripta elegans (Rocha et al., 2025). This adaptation may occur mainly due to its generalist habits and tolerance to anthropogenic pressure (Souza and Abe, 2001; Rocha et al., 2025). However, in the case of M. vanderhaegei, less abundant than P. geoffroanus, our study indicates that it may be less effective as a competitor and, consequently, less able to compete with T. dorbigni.
We show here the syntopic occurrence of Phrynops geoffroanus and Mesoclemmys vanderhaegei with different patterns of abundance. This varies for both species over time, and the abundance of P. geoffroanus is reflected in the abundance of M. vanderhaegei. Our findings suggest that P. geoffroanus is the dominant species in the study area and, according to other studies in urban areas (Souza and Abe, 2001; Souza et al., 2008; Ferronato et al., 2009; Rocha et al., 2025), should persist in this small protected fragment, as well as in other urban environments near the study area (Souza et al., 2008; Martins et al., 2010; Müller et al, 2019). Despite the challenges of living in fragmented urban environments, the presence of M. vanderhaegei suggests that the area still offers suitable conditions for species with higher habitat quality requirements. Both species seem to have an overlapping trophic niche, and resource partitioning is a hypothesis to be investigated.
The authors would like to thank Kwok Chiu Cheung, colleagues of the Study Group on Fragment Ecology (EcoFrag) and all field assistants. We also thank the team of the Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio) and the National Center for Research and Conservation of Reptiles and Amphibians (RAN) for the permits (SISBio #45297-1). ESB thanks Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for a scholarship (process 300658/2019-2) to the Programa de Capacitação Institucional (PCI) of the Museu Paraense Emilio Goeldi (MPEG); TFDR thanks CNPq for a scholarship (process 302506/2024-1) to the PCI of the Instituto Nacional de Pesquisas do Pantanal (INPP); CS thanks CNPq for a research fellowship (process 2312303/2018-1).
Alcalde, L., Derocco, N.N., Rosset, S.D., Williams, J.D. (2012): Southern-most localities of Trachemys dorbigni and first record of Trachemys scripta elegans for Argentina (Cryptodira, Emydidae). Chelonian Conserv. Biol. 11: 128-133.
Alho, C.J., Pádua, L.F. (1982): Sincronia entre regime de vazante do rio e comportamento de nidificação da tartaruga da Amazônia Podocnemis expansa (Testudinata: Pelomedusidae). Acta Amazon. 1: 323-326.
Balestra, R.A.M., Valadão, R.M., Vogt, R.C., Bernhard, R., Ferrara, C.R., Brito, E.S., Arias, R.B., Malvásio, A., Lustosa, A.P.G., Souza, F.L., Drummond, G.M., Bassetti, L.A.B., Coutinho, M.E., Ferreira-Junior, P.D., Campos, Z.M., Mendonça, S.H.S.T., Rocha, J.M.N., Luz, V.L.F. (2016): Roteiro para Inventários e Monitoramentos de Quelônios Continentais. Biodiv. Bras. (BioBrasil). 1: 114-152.
Brito, E.S.D., Strüssmann, C., Penha, J.M.F. (2009): Population structure of Mesoclemmys vanderhaegei (Bour, 1973) (Testudines: Chelidae) in the Cerrado of Chapada dos Guimarães, Mato Grosso, Brazil. Biota Neotrop. 9: 245-248.
Brito, E., Souza, F., Strüssmann, C. (2016): Feeding habits of Mesoclemmys vanderhaegei (Testudines: Chelidae). Acta Herpetol. 11: 1-13.
Brito, E.S., Vogt, R.C., Valadão, R.M., Franco, L.F., Penha, J., Strüssmann, C. (2018): Population ecology of the freshwater turtle Mesoclemmys vanderhaegei (Testudines: Chelidae). Herpetol. Conserv. Biol. 13: 355-365.
Bodie, J.R., Semlitsch, R.D. (2000): Spatial and temporal use of floodplain habitats by lentic and lotic species of aquatic turtles. Oecologia 122: 138-146.
Burnham, K.P., Anderson, D.R. (2002): Model Selection and Multi-Model Inference: A Practical Information- Theoretic Approach. New York, Springer.
Cagle, F.R. (1939): A system of marking turtles for future identification. Copeia 3: 170-173.
Ciccheto, J.R.M., Grou, C.E.V., Rocha, S.B. (2018): Novos registros de ocorrência de Trachemys dorbigni (Emydidae, Testudines) no Brasil. In: Estudo da herpetofauna brasileira, pp. 42-50. Oldiges, D.P., Ed, Ponta Grossa, Atena Editora.
Cheung, K.C., Koutchin-Reis, L., Jesus, C.C.C. (2016): Análise fitossociológica de um fragmento de cerrado em Campo Grande, MS. Multitemas 21: 241-256.
Corral, A., Valério, L.M., Cheung, K.C., Ferreira, B.H.S., Guerra, A., Szabo, J.K., Koutchin-Reis, L. (2021): Plant-bird mutualistic interactions can contribute to the regeneration of forest and non-forest urban patches in the Brazilian Cerrado. Urban Ecosyst. 24: 205-213.
Eisemberg, C.C., Balestra, R.A.M., Famelli, S., Pereira, F.F., Bernardes, V.C.D., Vogt, R.C. (2016): Vulnerability of Giant South American Turtle (Podocnemis expansa) nesting habitat to climate-change-induced alterations to fluvial cycles. Trop. Conserv. Sci. 9: 1-12.
Fachín-Terán, A., Vogt, R.C., Thorbjarnarson, J.B. (2006): Seasonal movements of Podocnemis sextuberculata (Testudines: Podocnemididae) in the Mamirauá sustainable development reserve, Amazonas, Brazil. Chelonian Conserv.Biol. 5: 18-24.
Ferronato, B.O., Merchant, M.E., Marques, T.S., Verdade, L.M. (2009): Characterization of innate immune activity in Phrynops geoffroanus (Testudines: Chelidae). Zoologia 26: 747-752.
Fiske, I., Chandeler, R. (2011): Unmarked: an R package for fitting hierarchical models of wildlife occurrence and abundance. J. Stat. Softw. 43: 1-23.
Fonseca, É., Both, C., Cechi, S.Z., Winck, G. (2021): Pet distribution modelling: Untangling the invasive potential of Trachemys dorbigni (Emydidae) in the Americas. PLoS ONE 16: e0259626.
Gibbons, J.W., Scott, D.E., Ryan, T.J., Buhlmann, K.A., Tiuberville, T.D., Metts, B.S., Greene, J.L., Mills, T., Leiden, Y., Poppy S., Winne, C.T. (2000): The Global Decline of Reptiles, Déjà Vu Amphibians: Reptile species are declining on a global scale. Six significant threats to reptile populations are habitat loss and degradation, introduced invasive species, environmental pollution, disease, unsustainable use, and global climate change. BioScience 50: 6531-666.
Guzy, J.C., Price, S.J., Dorcas, M.E. (2013): The spatial configuration of greenspace affects semi-aquatic turtle occupancy and species richness in a suburban landscape. Landsc. Urban Plan. 117: 46-56.
Hailey, A., Coulson, I.M. (1996): Temperature and the tropical tortoise Kinixys spekii: constraints on activity level and body temperature. J. Zool. 240: 523-536.
Hoef, J.M., Boveng, P.L. (2007): Quasi‐Poisson vs negative binomial regression: how should we model overdispersed count data? Ecology 88: 27661-2772.
Jaeger, C.P., Cobb, V.A. (2012): Comparative spatial ecologies of female painted turtles (Chrysemys picta) and red-eared sliders (Trachemys scripta) at Reelfoot Lake, Tennessee. Chelonian Conserv. Biol. 11: 591-67.
Liu, Z., He, C., Wu, J. (2016): The relationship between habitat loss and fragmentation during urbanization: an empirical evaluation from 16 world cities. PloS one 11: e0154613.
Lowe, S., Browne, M., Boudjelas, S., De Poorter, M. (2000): 100 of the world’s worst invasive alien species: a selection from the global invasive species database (Vol 12) Auckland: Invasive Species Specialist Group.
Luiselli, L., Akani, G.C. (2003): An indirect assessment of the effects of oil pollution on the diversity and functioning of turtle communities in the Niger Delta, Nigeria. Anim. Biodivers. Conserv. 26: 57-65.
Luiselli, L. (2008): Resource partitioning in freshwater turtle communities: A null model meta-analysis of available data. Acta Oecol. 34: 80-88.
Mackenzie, D.I., Nichols, J.D., Royle, J.A., Pollock, K.H., Bailey, L.L., Hines, J.E. (2006): Occupancy estimation and modeling: Inferring patterns and dynamics of species occurrence. Amsterdam, Elsevier/Academic Press.
Maffei, F., Do Nascimento, B.T.M., Moya, G.M., Donatelli, R.J. (2016): New distribution records of Mesoclemmys vanderhaegei (Testudines: Chelidae) from southeastern Brazil, including observations on reproduction. J. Threat. Taxa 8: 9322-9326.
Magalhães, F.M., Laranjeiras, D.O., Costa, T.B., Juncá, F.A., Mesquita, D.O., Röhr, D.L., Garda, A.A. (2015): Herpetofauna of protected areas in the Caatinga IV: Chapada Diamantina National Park, Bahia, Brazil. Herpetol. Notes 8: 243-261.
Marques, T.S., Lara, N.R., Bassetti, L.A., Ferronato, B.O., Malvásio, A., Verdade, L.M. (2013): Population structure of Mesoclemmys vanderhaegei (Testudines, Chelidae) in a silvicultural system in southeastern Brazil. Herpetol. Notes 6: 179-182.
Marques, T.S., Böhm, S., Brito, E.S., Cabrera, M.R., Verdade, L.M. (2014): Mesoclemmys vanderhaegei (Bour 1973) – Vanderhaege’s Toad-headed Turtle, Karumbé-hy. In: Conservation biology of freshwater turtles and tortoises: A compilation project of the IUCN/SSC Tortoise and Freshwater Turtle Specialist Group. Rhodin, A.G.J., Pritchard, P.C.H., Van Dijk, P.P., Saumure, R.A., Buhlmann, K.A., Iverson, J.B., Mittermeier, R.A. Chelonian Res. Monogr. 5: 0831-8.
Marcuzzo, F.F.N., Melo, D.C.D.R., Rocha, H.M. (2011): Distribuição espaço-temporal e sazonalidade das chuvas no Estado do Mato Grosso. Rev. Bras. Recur. Hídr. 16: 157-167.
Martins, F.I., Souza, F.L., Da Costa, H.T.M. (2010): Feeding habits of Phrynops geoffroanus (Chelidae) in an urban river in Central Brazil. Chelonian Conserv. Biol. 9: 294-297.
Mittermeier, R.A., Van Dijk, P.P., Rhodin, A.G., Nash, S.D. (2015): Turtle hotspots: an analysis of the occurrence of tortoises and freshwater turtles in biodiversity hotspots, high-biodiversity wilderness areas, and turtle priority areas. Chelonian Conserv. Biol. 14: 2-10.
Moll, D., Moll, E.O. (2004): The ecology, exploitation, and conservation of river turtles. New York, Oxford University Press.
Molina, F.B., Ferronato, B.O., Souza, F.L. (2016): Freshwater turtles in natural, rural and urban ecosystems in São Paulo State, Southeastern Brazil. In: Biodiversity in agricultural landscapes of Southeastern Brazil. pp. 151-162. Gheler-Costa, C., Lyra-Jorge, M.C., Verdade, L.M., Eds, Berlin, De Gruyter Open.
Müller, M.M.P., Ortega, Z., Antunes, P.C., Seino, L.L., Hammarstron, M.J., Balbino-Silva, A.C.F., Oliveira-Santos, L.G.R. (2019): The home range of adult Phrynops geoffroanus (Testudines, Chelidae) in relation to sex and body mass. Herpetozoa 32: 259-265.
Peel, M.C., Finlayson, B.L., Mcmahon, T.A. (2007): Updated world map of the Köppen-Geiger climate classification. Hydrol. Earth Syst. Sci. 11: 1633-1644.
Perry, G., Garland, J.R.T. (2002): Lizard home ranges revisited: effects of sex, body size, diet, habitat, and phylogeny. Ecology 83: 1870-1885.
Pérez-Santigosa, N., Florencio, M., Hidalgo-Vila, J., Días-Paniagua, C. (2011): Does the exotic invader turtle, Trachemys scripta elegans, compete for food with coexisting native turtles? Amphib.-Reptil. 32: 167-175.
Pittman, S.E., Dorcas, M.E. (2009): Movements, habitat use, and thermal ecology of an isolated population of bog turtles (Glyptemys hlenbergii). Copeia 2009: 781-790.
Plummer, M.V., Mills, N.E., Allen, S.L. (1997): Activity, habitat, and movement patterns of softshell turtles (Trionyx spiniferus) in a small stream. Chelonian Conserv. Biol. 2: 514-520.
Plummer, M.V. (2003): Activity and thermal ecology of the box turtle, Terrapene ornata, at its southwestern range limit in Arizona. Chelonian Conserv. Biol. 4: 569-577.
R DEVELOPMENT CORE TEAM. (2019): R: a language and en- vironment for statistical computing. Available at http:// wwwR-projectorg/
Rachemys, T. (2009): First record of Trachemys dorbigni (Duméril & Bibron, 1835) (Testudines, Emydidae) in a remnant of Mesophytic Semideciduous Forest of São Paulo State, southeastern. Herpetol. Bull. 108: 27-30.
Rocha, S.B., Rouco, C., Grou, C.E.V., Ferreira, V.L. (2025): Freshwater turtle (Reptilia: Testudines) assemblage from an urban protected Atlantic Forest remnant: population density, movement and habitat preference. Zoologia (Curitiba) 42: e24009.
Silva, M.B.D., Da Rocha, W.A., Nogueira-Paranhos, J.D. (2016): Checklist of reptiles of the Amazonia-Caatinga-Cerrado ecotonal zone in eastern Maranhão, Brazil. Herpetol. Notes 9: 7-14.
Sano, S.M., Almeida, S.P., Ribeiro, J.F. (2008): Cerrado: ecologia e flora. Brasília, DF: Embrapa informação tecnológica. Planaltina, Distrito Federal, Embrapa Cerrados.
Souza, F.L., Abe, A.S. (2000): Feeding ecology, density and biomass of the freshwater turtle, Phrynops geoffroanus, inhabiting a polluted urban river in south-eastern Brazil. J. Zool. 252: 437-446.
Souza, F.L., Abe, A.S. (2001): Population structure and reproductive aspects of the freshwater turtle, Phrynops geoffroanus, inhabiting an urban river in southeastern Brazil. Stud. Neotrop. Fauna Environ. 36: 57-62.
Souza, F.L. (2004): Uma revisão sobre padrões de atividade, reprodução e alimentação de cágados brasileiros (Testudines, Chelidae). Phyllomedusa 3: 15-27.
Souza, F.L., Raizer, J., Costa, H.Y.M., Martins, F.I. (2008): Dispersal of Phrynops geoffroanus (Chelidae) in an urban river in central Brazil. Chelonian Conserv. Biol 7: 257-61.
Spencer, R.J., Dykea, J.V., Petrova, K., Ferronato, B., Mcdougalla, F., Austina, M., Keiteld, C., Georges, A. (2018): Profiling a possible rapid extinction event in a long-lived species. Biol. Conserv. 221: 190-197.
Stokeld, D., Hamer, A.J., Van Der Ree, R., Pettigrove, V., Gillespie, G. (2014): Factors influencing occurrence of a freshwater turtle in an urban landscape: a resilient species? Wildl. Res. 41: 163-171.
Tamplin, J.W., Cyr, A.B. (2011): Effects of acclimation and egg-incubation temperature on selected temperature by hatchling western painted turtles (Chrysemys picta bellii). J. Therm. Biol. 36: 507-514.
Venables, W.N., Ripley, B.D. (2002): Modern applied statistics with S-Plus. New York, Springer.
Zalba, S., Ziller, S.R. (2007): Adaptive management of alien invasive species: putting the theory into practice. Natureza & Conservação 5: 86-92.