Submitted on: 2024 16th December; revised on: 2025 20 th March; accepted on:2025, 27 th March
Anuran life histories remain consistent across contrasting precipitation regimes in Northeastern Brazilian forests
1 Instituto Federal de Sergipe, Campus Propriá, CEP 49900-000, Propriá, Brasil
2 Departamento de Biologia, Laboratório de Répteis e Anfíbios, Universidade Federal de Sergipe, CEP 49100-000, São Cristovão, Brasil
3 Programa de Pós Graduação em Ecologia e Conservação, Universidade Federal de Sergipe, CEP 49100-000, São Cristovão, Brasil
4 Departamento de Botânica, Ecologia e Zoologia, Universidade Federal do Rio Grande do Norte, CEP 59078-900, Natal, Brasil
5 Programa de Pós-Graduação em Biodiversidade e Evolução, Instituto de Biologia, Universidade Federal da Bahia, CEP 40170-115, Salvador, Brasil
6 Centro de Ciências Agrárias, Laboratório de Microbiologia, Universidade Federal da Paraíba – Campus II, CEP 58397-000, Areia, Brasil
7 Departamento de Sistemática e Ecologia, Universidade Federal da Paraíba, CEP 58059-900 João Pessoa, Paraíba, Brasil
*Corresponding author. Email: francisluiz_bio@hotmail.com
Abstract. Amphibians are inherently dependent on water, and many species characteristics are influenced by this resource, particularly in ecoregions with contrasting precipitation regimes, such as the dry Caatinga and the moist Atlantic Forests of Northeastern Brazil. We compared these environments and hypothesized that anurans, regardless of the species, would be smaller in the Caatinga due to unpredictable duration of the rainy season, which accelerates metamorphosis, and the resulting fluctuations of resources, which limit energy reserves for growth. Furthermore, we anticipated that female reproductive strategies in the Caatinga would be more focused on quality rather than quantity, with small clutches and large oocytes. To test these hypotheses, we sampled areas of Caatinga and Atlantic Forest (two of each) in northeastern Brazil through 15-day consecutive campaigns conducted during the dry and rainy seasons. We combined active searches and pitfall traps to collect the anurans. We performed intraspecific comparisons using morphometric measurements and number and volume of oocytes. Contrary to our expectations, body size did not differ between forests for either sex. In moist environments, the extended reproductive period and the associated energetic demands may constrain growth, imposing costs similar to those in the semiarid environment, resulting in comparable body sizes. Similarly, few differences were observed in oocyte numbers and volumes. Leptodactylus macrosternum and Rhinella diptycha showed fewer and smaller oocytes, respectively, in the Caatinga. Biological factors such as lifespan and body size, and environmental factors such as the duration of the hydroperiod, predation, and insolation can influence oocyte size and numbers in different conditions. However, they likely interact in minimizing energy expenditure, resulting in similar reproductive strategies in contrasting environments.
Keywords. Frogs, local adaptation, abiotic conditions, selective pressures, Ecophysiology, life history, precipitation seasonality.
Understanding how abiotic factors shape species’ life histories is a central question in ecology (Blaustein et al., 1999; Saenz et al., 2006; Pavón‐Vázquez et al., 2022). This issue is particularly relevant for amphibians, given their critical dependence on water for essential biological functions (Pough et al., 1983; Dayton and Fitzgerald, 2006; Brandão et al., 2020). Consequently, life history of these organisms can be directly affected by hydrological dynamics and precipitation regimes (Sullivan and Fernandez, 1999; Navas et al., 2004; Li et al., 2023).
External morphology evolves under selective pressures tied to specific ecological demands, such as increased efficiency in competitive interactions or survival in environments with fluctuating resource availability (Demetrius, 2000; Schäuble, 2004). Consequently, marked morphological variation may arise across populations subject to the selective pressures of different environments. These modifications in life history traits (e.g. body size, clutch size) can result from phenotypic plasticity or local evolutionary adaptation (Pfennig and Murphy, 2002; García et al., 2021; Tiar-Saadi et al., 2022). Howard (1981) suggested that adult body size in amphibians is directly related to reproductive effort, responding to ecological constraints that influence development trajectories of each sex.
In stable environments, with limited resource fluctuations, body size of both sexes increases with the rise in resource acquisition, and sexual selection favors larger individuals, even though dimorphism is preserved (Howard, 1981). Additionally, physiological performance during the larval phase directly influences adult body size (Werner, 1986). Conversely, in unstable environments with high resource fluctuations, anurans encounter sharp increases in prey availability, mainly invertebrates, during the brief and irregular rainy season (Bento et al., 2016; Silva et al., 2021). As a result, post-metamorphic individuals may endure extended periods of reduced resource availability, limiting energy intake for growth and ultimately leading to smaller adults.
Harsh environments can also compromise reproductive output, affecting reproductive investment by imposing significant losses on clutches due to high temperature, solar radiation rates or irregular water availability (Blaustein et al., 1995; Neckel-Oliveira, 2004; Kissel et al., 2019). Life history theory posits that in unstable environments investing in larger oocytes reduces clutch sizes but increases chances of survival of both reproductive females and juveniles (Roff, 1992; Roff, 2002). For example, clutch size in Bufo andrewsi increases with female body size in seasons that favor growth, making them more enduring (Liao et al., 2016). Meanwhile, oocyte size increases during shorter, less favorable periods for resource acquisition regardless of body size (Liao et al., 2016).
In the Neotropical realm, the dry Caatinga and the moist Atlantic Forest of Brazil are ecoregions with contrasting abiotic conditions. While the former experiences an extended dry season, semiarid climate, reduced and irregular precipitation regimes, and predominance of temporary aquatic environments (Nimer, 1989; Ab’Saber, 1999; Silva et al., 2017), the latter has a humid tropical climate with high and evenly distributed precipitation throughout the year and greater predominance of permanent aquatic environments, due to its proximity to the coast (Ab’Saber, 1977; Tonhasca-Júnior, 2005; Joly et al., 2014). Anurans occurring in these contrasting environments exhibit shorter reproductive periods in the Caatinga, as a result of the reduced spatial and temporal availability of water (Caldas et al., 2016; Caldas et al., 2019). Given the contrasting conditions occupied by frogs in these quite different environments, we hypothesized that: 1) Anuran males and females are smaller in the Caatinga compared to the Atlantic Forest, as water irregularity and the predominance of temporary aquatic environments would accelerate metamorphosis, and marked seasonal fluctuations in resources availability in the post-metamorphic period would reduce the energy stock necessary for growth; 2) Females in the Caatinga are expected to prioritize reproductive quality over quantity, producing fewer but larger oocytes compared to their counterparts in the Atlantic Forest. These strategies could ensure higher chances of survival for larvae in harsh and unstable environments.
To test the hypotheses, we measured morphometric traits of reproductive males and females of species occurring both in Caatinga and Atlantic Forest and counted the mature oocytes of females from both ecoregions. Four species were selected for the morphometric analysis: Boana raniceps (Chaco tree frog), Leptodactylus vastus (Northeastern Pepper Frog), Scinax x-signatus (Venezuela snouted treefrog), and Leptodactylus troglodytes (Pernambuco white-lipped frog). Three species were selected for the reproductive analysis [B. raniceps, Leptodactylus macrosternum (Miranda’s White-lipped Frog) and Rhinella diptycha (cururu toad)]. All species have aquatic reproductive modes and the entire larval period occurs in the water (Nunes-de-Almeida et al., 2021) making them interesting models to evaluate both questions tested in this paper.
The study was conducted with anuran specimens from four protected areas in the Northeast region of Brazil: the Benjamin Maranhão Botanical Garden Permanent Preservation Area, the Seridó and Aiuaba Ecological Stations and the Guaribas Biological Reserve (Fig. 1). The Seridó and Aiuaba Ecological Stations are situated in the Northern Sertaneja Depression, located in the Caatinga ecoregion, part of the biome “Tropical & Subtropical Dry Broadleaf Forests” (Dinerstein et al., 2017). The climate is hot and semi-arid, and the average precipitation is below 800 mm (Velloso et al., 2002). The Guaribas Biological Reserve and the Benjamin Maranhão Botanical Garden are located in the Atlantic Forest ecoregion, part of the biome “Tropical & Subtropical Moist Broadleaf Forests” (Dinerstein et al., 2017) with a tropical rainy climate and a short dry season (Köppen type Am) and have average precipitation above 1500 mm (Nimer, 1979).
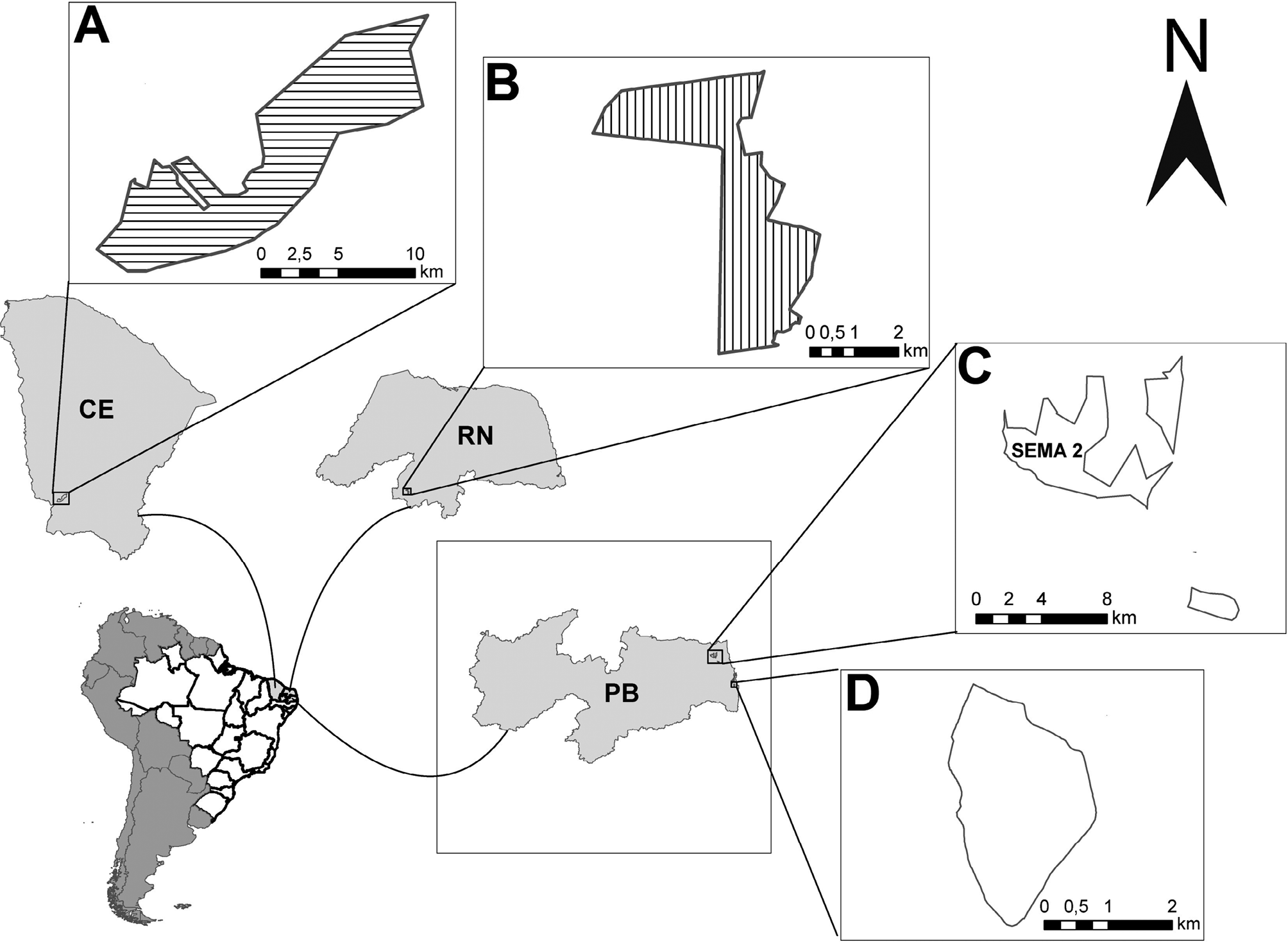
The Seridó Ecological Station (Fig. 1) is located in the state of Rio Grande do Norte (06°35’ to 06°40’S, 37°20’ to 37°39’W), in the municipality of Serra Negra do Norte, totaling an area of approximately 1,166 ha, mainly composed of herbs and shrubs, predominating genus such as Amburana (Fabaceae), Mimosa (Fabaceae), among others, and an endemic species, Gossypium mustelinum (Malvaceae) (Velloso et al., 2002). The Aiuaba Ecological Station (Fig. 1) located in the state of Ceará (06°36’01” to 06°44’35” S and 40°07’15” to 40°19’19” W), in the municipality of Aiuaba, totaling an area of 11,525 ha (Velloso et al., 2002). It includes an arboreal Caatinga remnant with predominantly open vegetation formations with Cnidoscolus–Bursera–Caesalpinia (Andrade-Lima, 1981).
The Guaribas Biological Reserve and Benjamin Maranhão Botanical Garden (Fig. 1) are located in Paraíba state. The former is located in the municipalities of Mamanguape (91.59%) and Rio Tinto (8.41%), encompassing areas termed SEMA 01, 02, and 03. We sampled SEMA 2 (06°40’40” a 06°44’59” S e 41°12’47” a 41°07’11” O) which has an area of approximately 3,378 ha (Barbosa et al., 2011). This protected area is highly heterogeneous, comprising tree formations, typical coastal restingas savannas, and an arboreal formation of semi-deciduous seasonal forest (Oliveira-Filho and Carvalho, 1993). In turn, the Botanic Garden is a remnant of forest located in the urban area of the municipality of João Pessoa (07°06’S and 34°52’W). The area is divided by the BR-230 highway, resulting in a larger continuous portion of 471 ha (sampling site) and 11 small fragments located within Campus I of the Federal University of Paraíba. This environment is typical of the Northeast coastal moist forest, featuring secondary growth and a flora of trees, shrubs, and herbaceous plants, with a low sub-canopy layer and few lianas (Dias et al., 2006).
Sampling for this study was conducted in 2013 across the four protected areas. We conducted surveys during the dry season in January (Guaribas), September (Aiuaba), and December (Seridó), while rainy season surveys were made in May (Seridó), April (Aiuaba), and June (Guaribas). For the Benjamin Maranhão Botanical Garden, sampling took place between 2011 and 2012. These data were obtained using the same collection methods as the other sites over a period of 12 months. Since it was possible to filter data covering a similar range of days as the other areas in a seasonal approach, no inconsistencies were caused in the sampling. We selected May, June and July (three of the rainiest months) and October, November, and December (three of the driest months). These intervals showed rainfall averages similar to those of the Guaribas REBIO in 2013 (see National Institute of Meteorology, INMET, 2012). These months were chosen based on seasonal definitions established by Lima and Heckendorff (1985), Aguiar and Martins (2002) and Velloso et al. (2002): in the Benjamim Maranhão Botanic Garden, the period of highest rainfall concentration typically occurs between March and August, in the Guaribas Biological Reserve between April and October, and in the Northern Sertaneja Depression, rainfall occurs from October to April. These patterns have been corroborated by 2013 monthly precipitation averages for each site (data from the Brazilian National Institute of Meteorology, INMET, 2012; INMET, 2013).
Each survey site was sampled for 15 consecutive days per season, resulting in 30 sampling days per site and a total of 90 days across sites. Two methodologies were used for field sampling. The main survey approach involved systematic visual and auditory inspection of the environments for anurans following standard methods (Crump and Scott, 1994), conducted between 18:00 and 22:00 hours. The active search has always been performed by two scientists, totaling 360 hours per person, and after being located, the specimens were manually collected. We tried to survey as many aquatic environments of aquatic environments based on the availability of each site: Guaribas Biological Reserve – five temporary ponds, one perennial stream and one perennial lake; Benjamim Maranhão Botanic Garden – a dammed section of the Jaguaribe River (7°08’42’ ’S and 34°51’54” W); Seridó Ecological Station – three temporary ponds, one intermittent stream, and one perennial lake; Aiuaba Ecological Station – four temporary ponds and one perennial lake. We believe the surveyed environments were adequate to record the species used in the comparisons. Their structural variation among sites was not considered problematic, as the species do not have riparian habits, and all but one (Corythomantis greeningi) reproduce in lentic habitats (Arzabe, 1999; Vieira et al., 2007; Santana et al., 2008).
The complementary survey approach involved the use of pitfall traps. We selected two aquatic environments with distinct conditions (temporary and perennial) in each survey site and install three trapping systems at the margins of each (except at the Botanical Garden, where two areas on the banks of the Jaguaribe River were used). The system consisted of four 30-L buckets buried in the ground and connected by 5-m plastic drift fences arranged in a Y pattern (Cechin and Martins, 2000; Enge, 2001). The traps were checked twice daily for 15 days to prevent animal mortality.
We selected four anuran species, B. raniceps, L. vastus, S. x-signatus, and L. troglodytes. The collected specimens were transported to the laboratory, euthanized with 2% lidocaine, fixed in 10% formalin, and then preserved in 70% ethanol (Caldas et al., 2016; Caldas et al., 2019), with each specimen assigned an identification number. After fixation, we measured 11 morphometric traits for each individual using a digital caliper (precision of 0.01 mm): snout-vent length (SVL), head length, head width, inter-eye distance, eye-nostril distance, inter-nostril distance, thigh length, tibia length, foot length, eye diameter, and inter-orbital distance (Napoli and Pimenta, 2009).
Next, we dissected the sampled individuals to determine their sex through direct observation of the gonads. The reproductive condition was assessed by examining the reproductive organs in females and the vocal sacs in males. Females were considered reproductive and suitable for comparison if they had highly convoluted oviducts and developed ovaries, while males were considered reproductive if they displayed fully evident vocal sacs (Mesquita et al., 2004). Individuals lacking these characteristics were classified as non-reproductive and excluded from the analyses.
We selected three species for these analyses: B. raniceps, L. macrosternum, and R. diptycha. They were the only ones that had females with oocytes and occurred in both of the compared ecoregions. We obtained large samples for each species (B. raniceps, n = 73; L. macrosternum, n = 210; R. diptycha, n = 86). However, the proportions of females were low [B. raniceps = 25 (34%); L. macrosternum = 81 (39%); R. diptycha = 24 (28%)], a situation that commonly occur in some anurans (Braun and Braun, 1977; Wiens et al., 2011). Few of these cataloged females contained oocytes (B. raniceps, n = 8 (32%); L. macrosternum, n = 31 (38%); R. diptycha, n = 9 (37%). Due to the smaller number of females and the occasional presence of oocytes in them, it was not possible to obtain information for the last two species at one of the Atlantic Forest sites (APP JBBM). In the laboratory, we dissected the specimens and removed the mature ovarian oocytes (post-vitellogenic) from females. These oocytes were identified by their well-differentiated hemispheres, with the animal pole displaying a dark brown coloration and the vegetative pole appearing milky and ivory in color (Melchiors et al., 2004). The material was then preserved in 70% ethanol.
We counted oocytes using a graduated cylinder filled with water up to a fixed measurement (properly recorded). We submerged the entire mass of oocytes into the container and recorded the displacement of the water column. Subsequently, we removed the mass and reintroduced only enough to displace 1.5 mm3. Next, the oocytes present in this small mass were dissociated by immersing them in a 10% sodium hypochlorite solution for 20 minutes (Melchiors et al., 2004). The dissociation allowed for the individual counting of the oocytes using a stereoscopic magnifying glass. With the total volume of the oocyte mass and the number of oocytes present in 1.5 mm3, it was possible to estimate each oocyte’s total number and average volume through a simple proportion. After completing the procedures, we layed all specimens and oocytes in the Coleção Herpetológica da Universidade Federal da Paraíba.
We initially assessed the normality of the data using the Shapiro-Wilk test to determine whether parametric or nonparametric analyses would be appropriate. Body size was treated as an isometric variable (Rohlf and Bookstein, 1987), following the protocol described by Somers (1986). An isometric eigenvector was calculated with predefined values of p-0.5, where “p” represents the number of variables (Jolicoeur, 1963). The scores for this eigenvector, referred to as “composite body size,” were obtained by multiplying the n x p matrix of log10-transformed data, where “n” is the number of observations. To evaluate differences in composite body size between individuals of the same species across ecoregions, we conducted separate analyses of variance (ANOVA) for males and females.
For the oocyte masses, we calculated the average number of oocytes for species from both the Caatinga and Atlantic Forests. To test whether the total number and average individual volume of oocytes differed significantly between specimens from the two ecoregions, we performed an analysis of variance (ANOVA) (Shapiro-Wilk, p > 0,05) or Mann-Whitney test (Shapiro-Wilk, p < 0,05), depending on the normality of the data. All analyses were conducted using the R program for Windows (R Development Core Team, 2018): package Rcmdr version 2.9-5 for Shapiro-Wilk, ANOVA, and Mann-Whitney (Fox et al., 2020).
We did not find significant differences in body size in the four species between the two ecoregions, regardless of sex. Males and females exhibit similar body sizes in both forests: males of B. raniceps (ANOVA, F1,46: 0.60; P = 0.44), females of B. raniceps (ANOVA, F1,23: 0.01; P = 0.91), and other males: S. x-signatus (ANOVA, F1,47: 0.41; P = 0.52), L. vastus (ANOVA, F1,33: 2.70; P = 0.11), and L. troglodytes (ANOVA, F1,18: 2.10; P = 0.16) (Table 1).
| Variables | B. raniceps n = 24 (49) |
S. x-signatus n = 22 (27) |
L. vastus n = 15 (20) |
L. troglodytes n = 6 ( 15) |
|
|---|---|---|---|---|---|
| males n = 15 (33) |
females n = 9 (16) |
males n = 22 (27) |
males n = 15 (20) |
males n = 6 (15) |
|
| SVL | 68.66 ± 3.42 | 72.57 ± 11.15 | 34.89 ± 2.78 | 135.25 ± 31.87 | 48.33 ± 1.59 |
| (63.00 ± 10.35) | (68.91 ± 7.82) | (33.19 ± 4.12) | (153.30 ± 20.37) | (43.42 ± 6.74) | |
| Composite Body Size | 4.15 ± 0.08 | 4.23 ± 0.30 | 3.07 ± 0.17 | 5.11 ± 0.61 | 3.65 ± 0.09 |
| (4.08 ± 0.30) F1,46 = 0.60 NS |
(4.24 ± 0.14) F1,23 = 0.01 NS |
(3.10 ± 0.16) F1,47 = 0.41 NS |
(5.34 ± 0.14) F1,33 = 2.70 NS |
(3.55 ± 0.15) F1,18 = 2.10 NS |
|
Similarly, the number of oocytes did not differ between ecoregions for B. raniceps and R. diptycha (Table 2). However, we observed differences in the number of oocytes for L. macrosternum, which was lower in the Caatinga (mean = 2,877 ± 1,723) and higher in the Atlantic Forest (mean = 5,255 ± 2,051); (ANOVA, F1,29: 11.41; p = 0.02) (Table 2). The other species did not show variations: B. raniceps (ANOVA, F1,6: 0.15; p = 0.70) and R. diptycha (Mann-Whitney, U1,7: 0.97; p = 0.32) (Table 2).
The average volume of each oocyte did not differ for B. raniceps (Table 2). However, we observed differences for R. diptycha, which had significantly smaller oocytes in the Caatinga (mean = 0.0015 ± 0.0008 mm3) compared to the Atlantic Forest (mean = 0.0036 ± 0.0013 mm3); (ANOVA, F1,7: 7.76; p = 0.03) (Table 2). The other species did not show variations: B. raniceps (ANOVA, F1,6: 3.55; p = 0.11) and L. macrosternum (ANOVA, F1,29: 0.20; p = 0.66) (Table 2).
| Variables | B. raniceps n = 4 (4) |
L. macrosternum n = 10 (21) |
R. diptycha n = 4 (5) |
|---|---|---|---|
| Average number of oocytes | 4,765 ± 4,634 | 5,255 ± 2,051 | 24,818 ± 10,013 |
| (3,728 ± 2,487) F1,6 = 0.15NS |
(2,877 ± 1,723) F1,29 = 11.41* |
(18,024 ± 17,440) U1,7 = 0.97NS |
|
| Average volume of oocytes | 0.0029 ± 0.0017 mm3 | 0.0018 ± 0.0005 mm3 | 0.0036 ± 0.0013 mm3 |
| (0.0013 ± 0.0002 mm3) F1,6 = 3.55NS |
(0.0019 ± 0.0010 mm3) F1,29: = 0.20NS |
(0.0015 ± 0.0008 mm3) F1,7 = 7.77* |
Contrary to our expectations, neither males nor females differed in size between Caatinga and Atlantic Forest. Similarly, reproductive efforts in the Caatinga did not differ significantly from the Atlantic Forest, except for L. macrosternum and R. diptycha, which showed a lower number and a smaller size of oocytes, respectively. Adult frogs were not smaller in environments with irregular precipitation regimes and reduced resources availability during the extended dry season (Caatinga), suggesting that hydroperiod and prey availability may not be limiting factors for growth. Possibly to avoid compromising growth due to resource limitations, other strategies to minimize energy loss in the dry season seem to have been selected in amphibians from Caatinga, such as aestivation and use of subterranean shelters in periods of reduced activity (Varjão and Ribeiro, 2018; Jared et al., 2019; Moreira et al., 2021).
In the Caatinga, lower precipitation and shorter hydroperiods can hypothetically accelerate metamorphosis and restrict resource availability for adults. Conversely, in the Atlantic Forest the hydrological stability can allow extended reproductive activity, but at the cost of expending more energy in vocalization, territory defense, partner encounter, courtship, and investment in oocytes (Arzabe et al., 1998; Afonso and Eterovick, 2007; Caldas et al., 2016). In both cases, energy deprivation would impact body size, as predicted by life history theory. Given limitations and finite resources, reproduction can incur costs that would ultimately reduce growth rate (Bradshaw and McMahon, 2008). In other words, body size patterns in the two ecoregions would be similar because growth would not be the main focus of energy investment, and thus, selective pressures would not promote an increase in size. In the Caatinga, the limited availability of trophic resources during the short rainy season would provide just enough energy for a rapid reproductive event. In the Atlantic Forest, the greater availability of trophic resources, ensured by annual water stability, would support a prolonged reproductive event. In both cases, the primary objective would be reproduction, and energy investment would be prioritized for this activity.
The expected patterns for hostile environments (Caatinga) and stable environments (Atlantic Forest) were not corroborated for most of the species’ reproductive investment. The number of oocytes did not change in B. raniceps. In contrast, the volume of oocytes in R. diptycha was smaller in the Caatinga, contrary to some expectations for their sizes under harsh conditions (Morrison and Hero, 2003; Liao et al., 2016). Alternatively, harsh environments with short seasons for resource acquisition can result in low annual growth rates. Thus, smaller oocytes or clutches would be compensated by more reproductive events and greater longevity (Sinsch et al., 2015), a pattern common for large-size species (Stark and Meiri, 2018), like Rana temporaria (Sinsch et al., 2015). Indeed, R. diptycha is a large species (Sousa et al., 2022), and longevity has already been indicated as a key factor for the persistence of some bufonids in arid environments (Tevis Jr, 1966). Therefore, slow growth rates and higher longevity resulting in more reproductive events in the Caatinga in R. diptycha may account for its smaller oocytes.
Leptodactylus macrosternum presented a lower number of oocytes in the Caatinga. However, contrary to what would be expected if the species in both regions allocated the same amount of energy for reproduction, these oocytes were not larger than those in the Atlantic Forest. Although it is not clear what drives this difference among areas, other factors can be driving egg number and size in this species (see Horato et al., 2024). The eggs of L. macrosternum are layed in the water (Heyer, 1969), and harsh insolation in arid environments can more severely affect eggs layed outside of water (Caldas et al., 2016). Similarly, there is less predation pressure in temporary aquatic environments subject to prolonged droughts (Woodward, 1983; Dayton and Fitzgerald, 2001; Schiwitz et al., 2020). The combination of such factors could prevent natural selection from favoring an increase in oocyte volume, resistance, and larval size. Thus, the qualitative nature of L. macrosternum reproduction would prevail. This hypothesis needs to be tested for the cause-and-effect relationships we discussed to be confirmed.
Studies focusing on the possible effects of environmental variables on life history traits in amphibian species from the Brazilian Forests remain scarce. Despite our moderate sample sizes, our data suggest that the differences between precipitation regimes are probably insufficient to promote significant reproductive and morphological adjustments in species that can thrive in both Caatinga and Atlantic Forest. However, the observed patterns require further investigation to be completely understood. In particular, future research should focus on other environmental factors and taxa, as well as increasing the number of samples analyzed.
We thank the Biodiversity Information and Authorization System (Sistema de Autorização e Informação em Biodiversidade – SISBIO) for granting the collection license (number 36095-8). This study received support from the managers and staff of all conservation units, especially J. L. Nascimento, G. Freitas, M. Kluppel (all from Guaribas Biological Reserve), G. Stephenson (Seridó Ecological Station), and M. Souza (Aiuaba Ecological Station). The Federal University of Paraíba (Universidade Federal da Paraíba–UFPB) provided logistical support during the study, and the Federal University of Rio Grande do Norte (Universidade Federal do Rio Grande do Norte–UFRN) and the Federal University of Sergipe (Universidade Federal de Sergipe–UFS) provided significant assistance during the project. Finally, we thank the Coordination for the Improvement of Higher Education Personnel – CAPES for the research scholarship and CNPq/ICMBIo for the financial support to AAG, which partially funded this study (# 552031/2011-9). AAG and DOM thank CNPq for financial support (research productivity grants numbers 309531/2015-2 and 303610/2014-0/306541/2017-3, 304715/2021-2, respectively). DOM acknowledges FAPESQ-PB for financial support (PRONEX and Universal). FLSC also thanks CNPQ for financial support in the postdoctoral period (numbers 150827/2018-0 and 150063/2022-9).
Ab’Saber, A.N. (1977): Os domínios morfoclimáticos na América do Sul: primeira aproximação. Geomorfologia 52: 1-121.
Ab’Saber, A.N. (1999): Sertões e sertanejos: uma geografia humana sofrida. Estud. Av. 13: 7-59.
Afonso, L.G., Eterovick, P.C. (2007): Spatial and temporal distribution of breeding anurans in streams in southeastern Brazil. J. Nat. Hist. 41: 949-963.
Aguiar, A.J.C., Martins, C.F. (2002): Abelhas e vespas solitárias em ninhos-armadilha na Reserva Biológica Guaribas (Mamanguape, Paraíba, Brasil). Rev. Bras. Zool. 19: 101-116.
Andrade-Lima, D. (1981): The caatingas dominium. Rev. Bras. Bot. 4: 149-163.
Arzabe, C., Carvalho, C.X., Costa, M.A.G. (1998): Anuran assemblages in Crasto Forest ponds (Sergipe State, Brazil): comparative structure and calling activity patterns. Herpetol. J. 8: 111-113.
Arzabe, C. (1999): Reproductive activity patterns of anurans in two different altitudinal sites within the Brazilian Caatinga. Rev. Bras. Zool. 16: 851-864.
Barbosa, M.R.V., Thomas, W.W., Zárate, E.L.P., Lima, R.B., Agra, M.F., Lima, I.B., Pessoa, M.C.R., Lourenço, A.R.L., Delgado Júnior, G.C., Pontes, R.A., Chagas, E.C.O., Viana, J.L., Neto, P.C.G., Araújo, C.M.L.R., Araújo, A.A.M., Freita, G.B., Lima, J.R., Silva, F.O., Vieira, L.A.F., Pereira, L.A., Costa, R.M.T., Duré, R.C., Sá, M.G.V. (2011): Checklist of the vascular plants of the Guaribas Biological Reserve, Paraíba, Brazil. Rev. Nord. Biol. 20: 79-106.
Bento, D.M., Ferreira, R.L., Prous, X., Souza-Silva, M., Bellini, B.C., Vasconcellos, A. (2016): Seasonal variations in cave invertebrate communities in the semiarid Caatinga, Brazil. J. Cave Karst Stud. 78: 61-71.
Blaustein, A.R., Edmond, B., Kiesecker, J.M., Beatty, J.J., Hokit, D.G. (1995): Ambient ultraviolet radiation causes mortality in salamander eggs. Ecol. Appl. 5: 740-743.
Blaustein, L., Garb, J.E., Shebitz, D., Nevo, E. (1999): Microclimate, developmental plasticity and community structure in artificial temporary pools. Hydrobiologia 392: 187-196.
Bradshaw, C.J.A., McMahon, C.R. (2008): Fecundity. In: Encyclopedia of Ecology, pp. 1535-1543. Amsterdam, Elsevier Inc.
Brandão, R.A., Fenker, J., Lopes, B.E.P.C., Sena, V.M.A., Vasconcelos, B.D. (2020): Diet of terrestrial anurans in an ephemeral and simplified habitat during the dry season in the Brazilian Cerrado. Ethol. Ecol. Evol. 1: 1-24.
Braun, P.C., Braun, C.A.S. (1977): Nova espécie de Hyla do Estado do Rio Grande do Sul, Brasil (Anura, Hylidae). Rev. Bras. Biol. 37: 853-857.
Caldas, F.L.S., Silva, B.D., Santos, R.A., De-Carvalho, C.B., Santana, D.O., Gomes, F.F.A., Faria, R.G. (2016): Autoecology of Phyllomedusa nordestina (Anura: Hylidae) in areas of the Caatinga and Atlantic Forest in the State of Sergipe, Brazil. North-West. J. Zool. 12: 271-285.
Caldas, F.L.S., Garda, A.A., Cavalcanti, L.B.Q., Leite-Filho, E., Faria, R.G., Mesquita, D.O. (2019): Spatial and trophic structure of anuran assemblages in environments with different seasonal regimes in the Brazilian Northeast Region. Copeia 107: 567-584.
Cechin, S.Z., Martins, M. (2000): Eficiência de armadilhas de queda (pitfall traps) em amostragens de anfíbios e répteis no Brasil. Rev. Bras. Zool. 17: 729-740.
Crump, M.L., Scott, N.J.J. (1994): Measuring and monitoring biological diversity: standard methods for amphibians. In: Standard Techniques for Inventory and Monitoring, pp. 76-141. Heyer, W.R., Donnelly, M.A., Mcdiarmid, R.W., Hayek, L.C., Foster, M.S., Eds., Washington, D.C., Eds, Smithsonian Institution Press.
Dayton, G.H., Fitzgerald, L.A. (2001): Competition, predation, and the distributions of four desert anurans. Oecologia 129: 430-435.
Dayton, G.H., Fitzgerald, L.A. (2006): Habitat suitability models for desert amphibians. Biol. Cons. 132: 40-49.
Demetrius, L. (2000): Directionality theory and the evolution of body size. Proc. R. Soc. B. 267: 2385-2391.
Dias, S.C., Candido, D.M., Brescovit, A.D. (2006): Scorpions from Mata do Buraquinho, João Pessoa, Paraíba, Brazil, with ecological notes on a population of Ananteris mauryi Lourenço (Scorpiones, Buthidae). Rev. Bras. Zool. 23: 707-710.
Dinerstein, E., Olson, D., Joshi, A., Vynne, C., Burgess, N., Wikramanayake, E., Saleem, M. (2017): Ecoregions 2017. Resolve. https://ecoregions.appspot.com/.
Enge, K.M. (2001): The pitfalls of pitfall traps. J. Herpetol. 35: 467-478.
Fox, J., Marquez, M.M., Bouchet-Valat, M. (2024). Rcmdr: R Commander. R package version 2.9-5, https://github.com/RCmdr-Project/rcmdr.
García, J., Arizaga, J., Rodríguez, J.I., Alonso, D., Suárez‐Seoane, S. (2021): Morphological differentiation in a migratory bird across geographic gradients in mountains of southern Europe. J. Biogeogr. 48: 2828-2838.
Heyer, W.R. (1969): The adaptive ecology of the species groups of the genus Leptodactylus (Amphibia, Leptodactylidae). Evolution 23: 421-428.
Horato, M.N., Almeida-Santos, M., Rocha, C.F.D., Sabagh, L.T. (2024): Reproductive ecology of treefrogs: egg size promotes reproductive effort differences between females. Evol. Ecol. 38: 481-493.
Howard, R.D. (1981): Sexual dimorphism in bullfrogs. Ecology 62: 303-310.
INMET (2012): Instituto Nacional de Meteorologia. https://portal.inmet.gov.br.
INMET (2013): Instituto Nacional de Meteorologia. https://portal.inmet.gov.br.
Jared, C., Mailho‐Fontana, P.L., Mendelson, J., Antoniazzi, M.M. (2019): Life history of frogs of the Brazilian semi‐arid (Caatinga), with emphasis in aestivation. Acta Zool. 101: 302-310.
Jolicoeur, P. (1963): The multivariate generalization of the allometry equation. Biometrics 19: 497-499.
Joly, C.A., Metzger, J.P., Tabarelli, M. (2014): Experiences from the Brazilian Atlantic Forest: ecological findings and conservation initiatives. New Phytol. 204: 459-473.
Kissel, A.M., Palen, W.J., Ryan, M.E., Adams, M.J. (2019): Compounding effects of climate change reduce population viability of a montane amphibian. Ecol. Appl. 29: 1-12.
Li, H., Cui, C., Shen, H., Zhu, Y., Chen, Z., Chen, X. (2023): Morphological variation and its correlation with bioclimatic factors in Odorrana graminea sensu stricto. Front. Ecol. Evol. 11: 1-11.
Liao, W.B., Luo, Y., Lou, S.L., Lu, D., Jehle, R. (2016): Geographic variation in life-history traits: growth season affects age structure, egg size and clutch size in Andrew’s toad (Bufo andrewsi). Front. Zool. 13: 1-9.
Lima, P.J., Heckendorff, W.D. (1985): Climatologia. In: Atlas Geográfico do Estado da Paraíba, pp. 34-43. UFPB, Ed., João Pessoa, Governo do Estado da Paraíba.
Melchiors, J., Di-Bernardo, M., Pontes, G.M.F., de Oliveira, R.B., Solé, M., Kwet, A. (2004): Reproduction of Pseudis minuta (Anura, Hylidae) in southern Brazil. Phyllomedusa 3: 61-68.
Mesquita, D.O., Costa, G.C., Zatz, M.G. (2004): Ecological aspects of the casque-headed frog Aparasphenodon brunoi (Anura, Hylidae) in a Restinga habitat in southeastern Brazil. Phyllomedusa 3: 51-59.
Moreira, D.C., Carvajalino-Fernández, J.M., Navas, C.A., Carvalho, J.E., Hermes-Lima, M. (2021): Metabolic and redox biomarkers in skeletal muscle underlie physiological adaptations of two estivating anuran species in a South American semi-arid environment. Front. Physiol. 12: 1-13.
Morrison, C., Hero, J.M. (2003): Geographic variation in life‐history characteristics of amphibians: a review. J. Anim. Ecol. 72: 270-279.
Napoli, M.F., Pimenta, B.V.S. (2009): A new species of the Bokermannohyla circumdata group (Anura: Hylidae) from the coastal forests of Bahia, Northeastern Brazil. Copeia 2009: 674-683.
Navas, C.A., Antoniazzi, M.M., Jared, C. (2004): A preliminary assessment of anuran physiological and morphological adaptation to the Caatinga, a Brazilian semi-arid environment. Int. Congr. Ser. 1275: 298-305.
Neckel-Oliveira, S. (2004): Effects of landscape change on clutches of Phyllomedusa tarsius, a neotropical treefrog. Biol. Cons. 118: 109-116.
Nimer, E. (1979): Climatologia do Brasil. Rio de Janeiro, IBGE.
Nimer, E. (1989): Climatologia da região Nordeste. In: Climatologia do Brasil, p. 315-361. IBGE, DRNEA, Eds., Rio de Janeiro, Fundação Instituto Brasileiro de Geografia e Estatística.
Nunes-de-Almeida, C.H.L., Haddad, C.F.B., Toledo, L.F. (2021): A revised classification of the amphibian reproductive modes. Salamandra, 57: 413-427.
Oliveira-Filho, A., Carvalho, D. (1993): Floristica e fisionomia da vegetação no extremo norte do litoral da Paraíba. Rev. Bras. Bot. 16: 115-130.
Pavón‐Vázquez, C.J., Brennan, I.G., Skeels, A., Keogh, J.S. (2022): Competition and geography underlie speciation and morphological evolution in Indo‐Australasian monitor lizards. Evolution 76: 476-495.
Pfennig, D.W., Murphy, P.J. (2002): How fluctuating competition and phenotypic plasticity mediate species divergence. Evolution 56: 1217-1228.
Pough, F.H., Taigen, T.L., Stewart, M.M., Brussard, P.F. (1983): Behavioral modification of evaporative water loss by a Puerto Rican frog. Ecology 64: 244-252.
R Development Core Team (2018): R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/.
Roff, D.A. (1992): Evolution of Life Histories: theory and Analysis. New York, Chapman e Hall.
Roff, D.A. (2002): Life-history Evolution. Sunderland, Sinauer Associates.
Rohlf, F.J., Bookstein, F.L. (1987): A comment on shearing as a method for “size correction”. Syst. Biol. 36: 356-367.
Saenz, D., Fitzgerald, L.A., Baum, K.A., Conner, R.N., Adams, D. (2006): Abiotic correlates of anuran calling phenology: the importance of rain, temperature, and season. Herpetol. Monogr. 20: 64-82.
Santana, G.G., Vieira, W.L., Pereira-Filho, G.A., Delfim, F.R., Lima, Y.C., Vieira, K.S. (2008): Herpetofauna em um fragmento de Floresta Atlântica no estado da Paraíba, Região Nordeste do Brasil. Biotemas 21: 75-84.
Schäuble, C.S. (2004): Variation in body size and sexual dimorphism across geographical and environmental space in the frogs Limnodynastes tasmaniensis and L. peronii. Biol. J. Linn. Soc. 82: 39-56.
Schiwitz, N.C., Schalk, C.M., Saenz, D. (2020): Activity level-predation risk tradeoff in a tadpole guild: implications for community organization along the hydroperiod gradient. Am. Midl. Nat. 183: 223-232.
Silva, I.S., Lucena, E.F., Moura, F.M.S., Vasconcellos, A. (2021): Termite flights seasonally promote nutrient pulses in the Caatinga dry forest in northeastern Brazil. Appl. Soil Ecol. 166: 1-6.
Silva, J.M.C., Barbosa, L.C.F., Leal, I.R., Tabarelli, M. (2017): The Caatinga: understanding the challenges. In: Caatinga: the Largest Tropical Dry Forest Region in South America, pp. 3-19. Silva, J.M.C., Leal, I.R., Tabarelli, M., Eds, Gewerbestrasse, Springer International Publishing.
Sinsch, U., Pelster, B., Ludwig, G. (2015): Large-scale variation of size- and age-related life-history traits in the common frog: a sensitive test case for macroecological rules. J. Zool. 297: 32-43.
Somers, K.M. (1986): Multivariate allometry and removal of size with principal components analysis. Syst. Biol. 35: 359-368.
Sousa, T.R., Benício, R.A., Fonseca, M.G. (2022): Polimorfismo em Rhinella diptycha (Anura: Bufonidae) em uma área de Caatinga, estado do Piauí, Nordeste do Brasil. Biosphere 1: 16-22.
Stark, G., Meiri, S. (2018): Cold and dark captivity: Drivers of amphibian longevity. Glob. Ecol. Biogeogr. 27: 1384-1397.
Sullivan, B.K., Fernandez, P.J. (1999): Breeding activity, estimated age-structure, and growth in sonoran desert anurans. Herpetologica 55: 334-343.
Tevis Jr, L. (1966): Unsuccessful breeding by desert toads (Bufo punctatus) at the limit of their ecological tolerance. Ecology 47: 766-775.
Tiar-Saadi, M., Tiar, G., Bouslama, Z., Široký, P. (2022): Mechanisms determining body size and shape difference in Algerian spur-thighed tortoises (Testudo graeca). Animals 12: 1-21.
Tonhasca-Júnior, A. (2005): Ecologia e História Natural da Mata Atlântica. Rio de Janeiro, Interciência.
Varjão, I.C.G., Ribeiro, L.B. (2018): Field record of aestivation with formation of cocoon in the frog Leptodactylus fuscus (Anura: Leptodactylidae) in a semiarid region of northeastern Brazil. Phyllomedusa 17: 135-138.
Velloso, A.L., Sampaio, E.V., Giulietti, A.M., Barbosa, M.R.V., Castro, A.A.J.F., Queiroz, L.P., Fernandes, A., Oren, D.C., Cestaro, L.A., Carvalho, A.J.E. (2002): Ecorregiões Propostas para o Bioma Caatinga. Recife, Flamar Gráfica e Editora.
Vieira, W.L.S., Arzabe, C., Santana, G.G. (2007): Composição e distribuição espaço-temporal de anuros no Cariri paraibano, Nordeste do Brasil. Oecol. Bras. 11: 383-396.
Werner, E.E. (1986): Amphibian metamorphosis: growth rate, predation risk, and the optimal size at transformation. Amer. Nat. 128: 319-341.
Wiens, J.J., Pyron, R.A., Moen, D.S. (2011): Phylogenetic origins of local‐scale diversity patterns and the causes of Amazonian megadiversity. Ecol. Lett. 14: 643-652.
Woodward, B.D. (1983): Predator-prey interactions and breeding-pond use of temporary-pond species in a desert anuran community. Ecology 64: 1549-1555.