Submitted on: 2025, 5th February; revised on: 2025, 4th April; accepted on: 2025, 20th June
Characterization of suitable breeding sites for Asiatic toads (Bufo gargarizans Cantor, 1842) (Anura, Bufonidae) using ecological modeling
1 Department of Biological Sciences, Chonnam National University, 61186 Gwangju, Republic of Korea
2 School of Biological Sciences and Biotechnology, Chonnam National University, 61186 Gwangju, Republic of Korea
3 Gwangyang Greenkorea, 57756, Gwangyang, Republic of Korea
*Corresponding author. Email: shcol2002@jnu.ac.kr
Abstract. Efforts to protect amphibian habitats and breeding sites are increasing, with ecological modeling playing a key role in predicting suitable habitats and informing conservation strategies. The Asiatic toad (Bufo gargarizans) is widely distributed across South Korea and is ideal for establishing comprehensive conservation measures. However, it is increasingly vulnerable to human activities and climate change. In this study, we investigated the breeding site characteristics of B. gargarizans using a generalized linear model (GLM). Field surveys were conducted at 124 reservoirs across three study sites at similar longitudes but different latitudes (Paju, Cheongju, and Gwangyang). To develop 30 candidate GLMs, using model selection based on the Akaike Information Criterion (AIC), 17 variables were collected from field measurements and geographic data. The results identified six factors influencing the suitability of B. gargarizans breeding sites: water temperature, dissolved oxygen, the average water depth 1 m from the shoreline, reservoir area, percentage of barren land within a 1km buffer, and percentage of water within a 1km buffer. The model indicated that areas with a shoreline depth of around 48 cm, low surrounding barren ratios (3%), and more than 3% water bodies are suitable for B. gargarizans breeding. These findings provide valuable insights into the conservation of B. gargarizans and can support the development of effective habitat protection measures.
Keywords. GLM, AIC, conservation management, shoreline depth, barren, water body.
Index
Amphibians are sensitive to subtle habitat changes (Beebee, 1997; Alford and Richards, 1999; Stebbins and Cohen, 2021), and thus face significant threats to their survival, such as habitat fragmentation and alteration (Dixo et al., 2009; Decena et al., 2020). Most amphibians breed and develop in water, undergoing an aquatic larval stage before metamorphosing and moving onto land. Therefore, the quality and stability of their habitats are crucial for survival (Evans et al., 1996) and protecting key breeding habitats (wetlands and reservoirs) is essential for amphibian conservation.
The suitable selection of breeding sites by amphibians significantly impacts their survival and breeding success (Ra et al., 2010; Borzée et al., 2018). Previous studies have identified factors that differentiate breeding sites based on adult responses to different variables such as water-holding capacity (Lin et al., 2008), reproduction avoidance in response to predators (Murphy, 2003 a; Jowers and Dowine, 2005), and negative relationships between conspecific density and breeding site preference (Resetarits and Wilbur, 1989; Crump, 1991; Spieler and Linsenmair, 1997; Murphy, 2003 a). Additionally, for amphibians that breed primarily in permanent water sources, the disappearance of breeding sites due to drying can be a major cause of mortality for the hatched tadpoles (Smith, 1983; Newman, 1988), highlighting the importance of maintaining breeding sites for stable larval growth (Edgerly et al., 1998; Murphy, 2003 b; Rudolf and Rödel, 2005).
Amphibians select breeding sites based on various environmental factors, including water temperature, dissolved oxygen, water depth, aquatic vegetation, and the presence of predators (Skelly et al., 1999; Semlitsch, 2000). The anuran family Bufonidae, commonly known as toads, tend to prefer larger bodies of water with stable environmental conditions, as seen in studies on the Rhinella marina, which selects breeding sites consisting of shallow pools and unvegetated muddy banks (Semeniuk et al., 2007), and Epidalea calamita whose site preference is also influenced by water temperature, chemistry, and the presence of competitor species (Banks and Beebee, 1987). Understanding these factors provides valuable insights into amphibian breeding and conservation efforts. Among the various research methods for identifying these key factors, ecological modeling has been widely used to predict suitable breeding sites by analyzing combinations of variables. Studies have applied habitat suitability models to assess amphibian breeding habitats based on environmental predictors, such as land cover, climate, and topography (Cunningham et al., 2007; Ra et al., 2010; Blank and Blaustein, 2012). These models have proven effective in identifying relationships between species and their environments and predicting species distributions, contributing to conservation planning (Guisan and Zimmermann, 2000; Lunghi et al., 2015; Su et al., 2020).
This study focused on the Asiatic toad (Bufo gargarizans Cantor, 1842). Bufo gargarizans is a species of the family Bufonidae that inhabits the inland areas of the Korean Peninsula and parts of mainland East Asia and breeds in flat water (Lee et al., 2011). A recent taxonomic review of this species was conducted by Matushkina et al. (2022), and based on molecular analyses, proposed reclassifying populations from the Korean Peninsula, northeastern China, and Russia Primorye region as Bufo sachalinensis (Othman et al., 2022). However, the International Union for Conservation of Nature (IUCN) and South Korea’s National Species List has not adopted this classification; therefore, this study will use Bufo gargarizans. According to the IUCN, the B. gargarizans classified as a “Least Concern (LC)” species (IUCN 2022). However, the population of B. gargarizans has been declining due to human activities and climate change, highlighting the need for conservation efforts (Sung et al., 2007; Yang et al., 2020). Identifying suitable habitats is essential to conserve the populations of B. gargarizans (Yang et al., 2023). Additionally, since B. gargarizans is widely distributed across South Korea (Lee et al., 2011), analyzing the key environmental factors of its breeding sites can contribute to broader conservation strategies for amphibians. Therefore, this study aimed to characterize the conditions of suitable breeding sites for B. gargarizans using Generalized Linear Models (GLM) to identify critical variables and their influence on the selection of breeding sites.
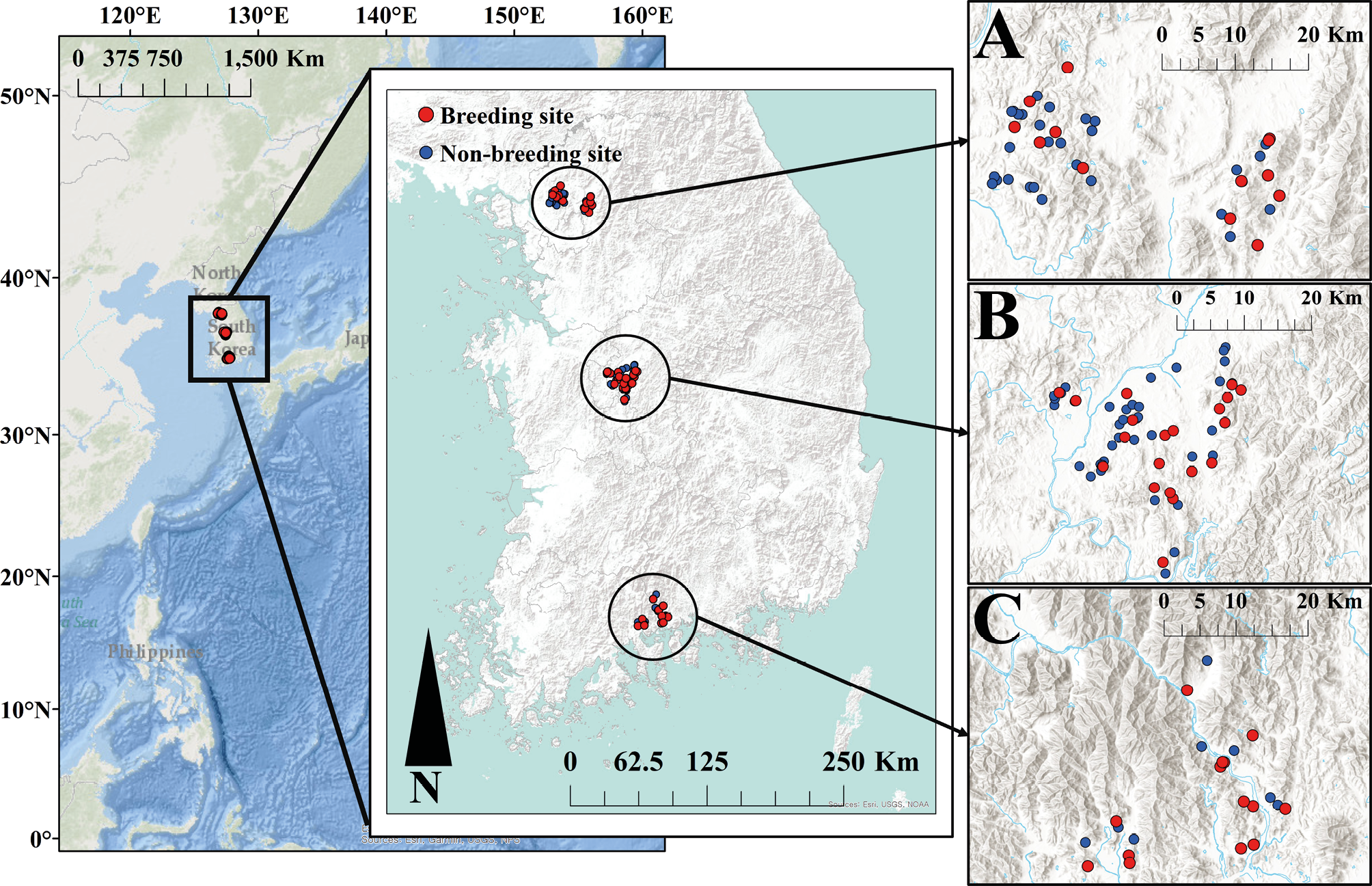
Since B. gargarizans are known to breed in stable water bodies (Lee et al., 2011), only reservoirs were selected as study sites where the water is stable. Data on B. gargarizans breeding sites nationwide were collected through preliminary research and surveys (NIE 2018) to determine the criteria for selecting reservoirs. Based on this data, regions located in the northern, central, and southern parts of the country and situated at 127° longitude were selected (Paju in the North, Cheongju in the Center, and Gwangyang in the South; Fig. 1). After selecting the study sites, a preliminary survey was conducted in January and February to observe the breeding activities and identify breeding and non-breeding sites.
Considering the breeding period of B. gargarizans (Lee et al., 2011), a field survey was conducted from March to May. The variables used for the model were referenced from previous studies (Evans et al., 1996; Ra et al., 2010). In previous studies, the variables were categorized into micro, biological, and non-biological categories. However, based on the toxicity of the eggs and tadpoles from the family Bufonidae (Crossland and Alford, 1998; Lim et al., 2005) and the fact that fish tend to avoid or do not prey on Bufonidae (Kruse and Stone, 1984; Kiesecker et al., 1996), this study assumes there is no correlation between Bufonidae larvae and the biological variables. Consequently, biological variables were not collected. Additionally, data collection was conducted exclusively through field observations and did not involve animal capture; therefore, no animal ethics approval was needed.
A total of 17 variables, consisting of 7 micro and 10 macro variables, were collected (Table 1). The micro variables consisted of 7 variables collected in the field: water temperature (Wt), pH (P), dissolved oxygen (Do), the average depth of the water 1 m from the shoreline (Dp), slope (Sl), elevation (El), and the average vegetation cover of the shoreline (Vc). The measurement method involved selecting four points in the east, west, south, and north of each reservoir based on true north and measuring the values at each point. First, water temperature, pH, and dissolved oxygen were measured using a water quality meter (86031 AZ EB; AZ Instrument Corp.; Taiwan) and by scooping water samples into a small box at each point. The slope and elevation at points approximately 1 m above the ground were recorded using a GPS receiver (GPSMAP 64s; Garmin; Switzerland). The average water depth was measured using a plastic ruler 1 m from the shoreline. The average vegetation cover was assessed by placing a 2 m × 2 m grid around the measured water depth and taking photographs. The photographs were then evaluated by two researchers, including the observer, to calculate the average cover. Values recorded at each point were averaged and utilized as variables.
The macro variables consisted of 10 variables (Table 1) and were calculated using ArcMap (ver. 10.7.1; ESRI; USA). The 10 macro variables consisted of reservoir area (Ar), distance to a forest (Df), distance to the water (Dw), distance to a used area (Du), percentage of barren land within a 1km buffer (Pb), percentage of agricultural land within a 1km buffer (Pa), percentage of forest within a 1km buffer (Pf), percentage of wetland and water within a 1km buffer (Pw), percentage of used area within a 1km buffer (Pu), and percentage of grass within a 1km buffer (Pg). First, the coordinates of the reservoirs were collected using Google Earth (ver. Pro; Google; USA). Then, using the Environmental Geographic Information Services, maps containing reservoirs were identified, and the land cover maps were downloaded at a scale of 1:5,000 (EGIS, 2019). Considering the field survey period, maps from 2019 that utilized the primary classification items were used. The classification consisted of 7 items, which were barren land, such as sand or gravel; agricultural lands, such as rice paddies and agricultural fields; forests, such as broadleaf and coniferous forests; used areas, including residential and industrial areas; grasslands (grass with a low proportion of trees); wetlands (consistently saturated and where water accumulates during the rainy season); and water, such as rivers and banks. However, in this study, due to the low proportions of wetlands and water (lower than 1%), they were merged into one variable: water. The reservoir shapes were extracted based on the coordinates, and the reservoir areas were calculated using these shapes. Using the ‘Find Nearest’ function, the distances between the reservoirs and the nearest forest, water, and used areas were calculated. To determine the surrounding landscapes of the reservoirs, the ‘Buffer’ and ‘Intersect’ functions were utilized to extract land use areas from each reservoir. The buffer range was set to 1 km, considering the maximum distance between the breeding site and the microhabitats of B. gargarizans within their home range (Park et al., 2021; 2024; 2025). Since the reservoir sizes varied, the areas within the 1 km buffer were converted into ratios. Based on these area ratios, the percentages of surrounding barren land, agricultural land, forest, water (including wetlands and water), used areas, and grasslands were calculated.
| No. | Variable (unit) | Acronym | Breeding site | Non-breeding site | |
|---|---|---|---|---|---|
| Micro | |||||
| 1 | Water temperature (°C) | Wt | 20.9 ± 3.0 (13.6–29.9) | 20.4 ± 3.8 (13.8–29.5) | |
| 2 | pH | P | 8.3 ± 1.1 (6.2–10.6) | 8.1 ± 1.1 (5.9–10.1) | |
| 3 | Dissolved Oxygen (ppm) | Do | 9.4 ± 2.3 (4.1–15.6) | 8.5 ± 2.8 (1.7–17.3) | |
| 4 | Average water depth of 1 m from the shoreline (cm) | Dp | 48.0 ± 17.6 (5–88.8) | 57.0 ± 18.7 (13.25–133) | |
| 5 | Slope (°) | Sl | 26.2 ± 11.5 (6.0–90) | 27.5 ± 13.2 (5.3–90) | |
| 6 | Elevation (m) | El | 101.4 ± 89.2 (5.0–383.8) | 83.3 ± 60.9 (0.7–350) | |
| 7 | Average vegetation cover of shoreline (%) | Vc | 30.4 ± 30.5 (0–100) | 28.3 ± 32.3 (0–100) | |
| Macro | |||||
| 8 | Distance to forest (m) | Df | 18.8 ± 40.7 (0–228.2) | 33.8 ± 51.4 (0–249.6) | |
| 9 | Distance to water (m) | Dw | 181.1 ± 250.9 (0–1,153.9) | 192.4 ± 275.4 (0–1,339) | |
| 10 | Distance to used area (m) | Du | 17.5 ± 31.3 (0–147.8) | 17.7 ± 39.1 (0–241.3) | |
| 11 | Reservoir area (m2) | Ar | 5,325.7 ± 7,219.8 (144.8–35,897.5) | 11,289.6 ± 40276.2 (104.6–276,599.7) | |
| Percentage within a 1km buffer | |||||
| 12 | Barren (%) | Pb | 3.6 ± 2.7 (0.3–11.9) | 4.7 ± 3.6 (0.5–22.7) | |
| 13 | Agricultural land (%) | Pa | 19.8 ± 11.5 (1.6–55) | 22.4 ± 13.9 (1.0–58.7) | |
| 14 | Forest (%) | Pf | 50 ± 18.3 (6.6–84.5) | 46.2 ± 20.5 (6.9–95.9) | |
| 15 | Water and wetland (%) | Pw | 3.9 ± 6.4 (0.1–28.7) | 2.4 ± 2.7 (0.3–12.2) | |
| 16 | Used area (%) | Pu | 8.8 ± 7.4 (1.3–37.7) | 9.5 ± 6.3 (0.3–29.6) | |
| 17 | Grass (%) | Pg | 13.9 ± 5.0 (3.5–27) | 14.9 ± 6.0 (1.5–37.9) | |
All statistical analyses were conducted using SPSS (ver. 20.0; IBM; USA). All variables did not exhibit normality in a Kolmogorov-Smirnov test (p < 0.05). Since no significant correlation (Spearman correlation; r > 0.8, p > 0.05) was observed between the variables, and no variable had a correlation coefficient above 0.8, all 17 variables were included in the analysis.
Modeling was conducted using a Generalized Linear Model (binomial logistic model), with breeding site presence as the dependent variable (1 = breeding site, 0 = non-breeding site). Model selection was based on the Akaike Information Criterion (AIC), a widely used method for comparing and evaluating habitat suitability models (Burnham and Anderson, 2004; Hu, 2007). The AIC approach requires species presence-absence data along with habitat information (MacKenzie and Bailey, 2004; Durso et al., 2011), allowing for the estimation of suitable breeding habitats. Following Ra et al. (2010), an a priori model was developed by selecting variables based on their ecological relevance and insights gained from field surveys. While testing all possible variable combinations would provide a more exhaustive evaluation, this approach was impractical due to computational constraints and the risk of model instability (Johnson and Omland, 2004; Diniz‐Filho et al., 2008). Instead, ecologically meaningful combinations were prioritized based on previous research and field observations. To systematically assess the influence of different variable types, the model selection was structured into three groups: (1) 10 models using only microhabitat variables, (2) 10 models using only macrohabitat variables, and (3) 9 models incorporating both. Additionally, a global model including all variables was tested, resulting in 30 model combinations (Table 2). This approach balanced model complexity and ecological interpretability while minimizing the risk of overfitting.
| Model No. | Variable |
|---|---|
| Micro variable combination | |
| 1 | Do, Sl, El, Vc |
| 2 | Do, Dp, El, Vc |
| 3 | Wt, P, Dp |
| 4 | P, Dp, El, Vc |
| 5 | Wt, Do, Dp, Vc |
| 6 | Wt, Do, Sl, El, Vc |
| 7 | Wt, Sl, El, Vc |
| 8 | Dp, Sl, El, Vc |
| 9 | Wt, P, Do, Dp, El, Vc |
| 10 | Wt, P, Do, Dp, Sl, El, Vc |
| Macro variable combination | |
| 11 | Df, Pf, Pg |
| 12 | Dw, Ar, Pa, Pw |
| 13 | Du, Pb, Pu |
| 14 | Df, Ar, Pb, Pw, Pg |
| 15 | Df, Pb, Pa, Pg |
| 16 | Df, Dw, Du, Pf, Pu, Pg |
| 17 | Df, Dw, Ar, Pb, Pa, Pf, Pw, Pg |
| 18 | Ar, Pb, Pw, Pu |
| 19 | Du, Ar, Pb, Pa, Pu, Pg |
| 20 | Df, Dw, Du, Ar, Pb, Pa, Pf, Pw, Pu, Pg |
| All variable combination | |
| 21 | Do, Dp, Pb, Pa, Pg |
| 22 | Do, Dp, Pb, Pf, Pw |
| 23 | Dp, Sl, Df, Du, Ar, Pa, Pf, Pw |
| 24 | Wt, P, Sl, Pb |
| 25 | Wt, Do, Dp, Ar, Pb, Pw |
| 26 | El, Vc, Df, Pf, Pg |
| 27 | Do, Sl, El, Df, Pf |
| 28 | Dp, El, Vc, Df, Pa, Pf, Pg |
| 29 | Do, Dp, Df, Pg |
| Global | |
| 30 | Wt, P, Do, Dp, Sl, El, Vc, Df, Dw, Du, Ar, Pb, Pa, Pf, Pw, Pu, Pg |
Considering the small sample size, the corrected Akaike Information Criterion (AICc) values were used in this study. Generally, the relative likelihood in an AIC model is proportional to the probability of minimizing the information loss for each model (Burnham and Anderson, 2004). The relative likelihood was calculated using the formula:
The calculated values were then used to confirm the model’s explanatory power as weights (wi), ensuring the reliability of the selected suitable model. The selected suitable model evaluated the impact on the breeding sites by assessing the significance of each variable.
The survey included 143 reservoirs: 21 in Gwangyang in 2017, 60 in Cheongju in 2018, and 62 in Paju in 2019. Among these, breeding sites were identified in 13 reservoirs in Gwangyang, 20 in Cheongju, and 23 in Paju, totaling 57 breeding sites. For the analysis, modeling was conducted using 123 sites (46 breeding sites, 77 non-breeding sites); the modeling excluded 20 sites in Paju (10 breeding sites, 10 non-breeding sites) that did not have land cover maps.
Among the breeding site models, Model 25 exhibited the highest explanatory power (wi) of 0.6464, and included six variables (Table 3): water temperature (Wt), dissolved oxygen (Do), the average water depth 1 m from the shoreline (Dp), reservoir area (Ar), the percentage of barren land within a 1 km buffer (Pb), and the percentage of water within a 1 km buffer (Pw). Among the variables included in this model, three were significant. The percentage of water within a 1km buffer (Pw) had a positive correlation. In comparison, the average water depth 1 m from the shoreline (Dp) and the percentage of barren land within a 1km buffer (Pb) had negative correlations (Table 4). The average water depth 1 m from the shoreline of breeding sites was 47.9 ± 17.6 cm, and 57.0 ± 18.7 cm for non-breeding sites. The percentage of barren land within a 1km buffer of breeding sites was 3.6 ± 2.7%, and 4.7 ± 3.6% for non-breeding sites. The percentage of water within a 1km buffer of breeding sites was 3.9 ± 6.4%, and 2.4 ± 2.7% for non-breeding sites.
| Model No. | Variable list | k | AICc | ∆AICc | wi |
|---|---|---|---|---|---|
| 25 | Wt, Do, Dp, Ar, Pb, Pw | 8 | 152.797 | 0 | 0.6464 |
| 22 | Do, Dp, Pb, Pf, Pw | 7 | 155.671 | 2.87 | 0.1536 |
| 29 | Do, Dp, Df, Pg | 6 | 158.423 | 5.63 | 0.0387 |
| 2 | Do, Dp, EL, Vc | 6 | 158.659 | 5.86 | 0.0345 |
| 4 | P, Dp, El, Vc | 6 | 158.726 | 5.93 | 0.0334 |
| 21 | Do, Dp, Pb, Pa, Pg | 7 | 159.344 | 6.55 | 0.0245 |
| 5 | Wt, Do, Dp, Vc | 6 | 159.437 | 6.64 | 0.0234 |
| 3 | Wt, P, Dp | 5 | 159.734 | 6.94 | 0.0201 |
| 18 | Ar, Pb, Pw, Pu | 6 | 160.164 | 7.37 | 0.0163 |
| 9 | Wt, P, Do, Dp, El, Vc | 8 | 161.313 | 8.52 | 0.0091 |
| Variable | B | SE | Wald Chi-Square | Sig |
|---|---|---|---|---|
| intercept | 1 | 1.6407 | 0.418 | 0.518 |
| Wt | 0.179 | 0.0617 | 1.620 | 0.203 |
| Do | 1.144 | 0.0795 | 3.263 | 0.071 |
| Dp | -0.035 | 0.0129 | 7.210 | 0.007 |
| Ar | -1.496 | 1.4142 | 1.080 | 0.299 |
| Pb | -0.203 | 0.0915 | 4.921 | 0.027 |
| Pw | 0.118 | 0.0525 | 5.091 | 0.024 |
This study explored the suitable environmental conditions using a relatively simple model for B. gargarizans breeding sites. Based on a GLM, the model provides valuable information on the major environmental predictors affecting the selection of the breeding sites of B. gargarizans. Among the variables showing significant differences, the average water depth 1 m from the shoreline (Dp) was considered an important factor. Since B. gargarizans are known to breed at the shoreline of reservoirs (Lee et al., 2011), it is important to maintain suitable shoreline depths (Jeong, 2017) and permanent waterbodies (Evans et al., 1996). Furthermore, suitable water depths are necessary to ensure the stable occurrence of eggs and larvae (Edgerly et al., 1998; Murphy, 2003b; Rudolf and Rödel, 2005) since lower water depths have lower temperatures, which can negatively affect breeding. In this study, the average shoreline water depth at non-breeding sites was relatively deep at 57 cm, nearly twice the 30 cm reported in a previous study (Jeong, 2017). Therefore, a water depth of 30–48 cm is a suitable shoreline water depth for B. gargarizans breeding sites.
The results of this study indicate that a percentage of barren land within a 1km buffer (Pb) lower than 3% and a percentage of water within a 1km buffer (Pw) higher than 3% are suitable for B. gargarizans breeding. B. gargarizans typically inhabit areas with vegetation cover or grasslands (Yu and Guo, 2010; Su et al., 2020; Park et al., 2024; Park et al., 2025), and are known to use reservoirs, ponds, rice paddies and rice paddy canals as breeding sites (Lee et al., 2011). Bufonids have thicker skin than other frog families (Lee, 2003) and are more resistant to drying out. However, they still prefer habitats with high humidity to retain moisture (Su et al., 2020). Accordingly, the amount of barren land surrounding the breeding sites was relatively low. Additionally, the percentage of water within a 1km buffer (Pw) of breeding sites was relatively high compared with the non-breeding sites. In the post-breeding period, B. gargarizans migrate from breeding sites (reservoirs) to the mountains (Park et al., 2021; 2024). While anurans of the family Bufonidae are known for their drought tolerance (Lee, 2003), maintaining access to water sources is crucial due to the high energy and moisture expenditure required for movement and dispersal (Yu et al., 2009; Luo et al., 2014; 2015). Therefore, additional water bodies near breeding sites likely play a vital role in the survival of metamorphosed tadpoles as they disperse into the forests.
Among the six variables included in the best-suitable model, dissolved oxygen (Do) had a p-value of 0.071, indicating marginal significance. However, it appeared in seven of the top ten models and was statistically significant (P < 0.05) in four of them (models 29, 2, 21, and 5), suggesting potential ecological relevance. The typical dissolved oxygen range in water is 7–10 ppm. In this study, dissolved oxygen levels were 9.4 ± 2.3 ppm in breeding sites and 8.5 ± 2.8 ppm for non-breeding sites, although the mean comparison between the two site types did not reach statistical significance (T-test, t = 1.875, P = 0.063).Previous studies have shown that toads prefer sites with higher dissolved oxygen levels (Noland and Ultsch, 1981; Semeniuk et al., 2007). While dissolved oxygen was included in 7 of the top 10 models, no significant differences were found between breeding and non-breeding sites. However, it remains an important environmental factor influencing breeding site selection. Dissolved oxygen levels can be affected by factors such as water flow, water depth, water temperature, and aquatic vegetation, which could be explored further in future studies.
The results of this study provide meaningful implications for the conservation and ecological management of B. gargarizans breeding habitats. Key factors such as shoreline water depth, the percentage of barren land surrounding the breeding sites, and nearby water bodies suggest practical strategies for breeding site restoration. For instance, altering artificial reservoirs with overly steep or deep shorelines to include shallower zones with depths around 48 cm may enhance breeding habitat suitability. Furthermore, improving dissolved oxygen concentrations could contribute to improving breeding site quality (Semeniuk et al., 2007). The presence of nearby water bodies for post-breeding dispersal further highlights the necessity of landscape-level conservation efforts. Preserving small wetlands and canals may facilitate metamorphism juvenile movement and connect habitats (Yu et al., 2009; Luo et al., 2014).
Nevertheless, considering the overlapping breeding periods with other amphibian species (Hynobius spp., Rana dybowskii, R. huanrenensis, and R. coreana; Lee et al., 2011), potential ecological interactions and impacts should be carefully evaluated. Moreover, B. gargarizans in South Korea primarily utilizes artificial reservoirs for breeding, but the effects on other organisms, including amphibians, are unknown. Future research should investigate whether these anthropogenic habitats confer ecological advantages compared with their natural breeding sites, which may, in turn, influence long-term population dynamics.
This study was supported by the Animal Behavior and Ecology Laboratory at Chonnam National University. All authors declare no conflicts of interest, and an anonymous reviewer is thanked for the helpful comments on a previous version of the manuscript.
Alford, R.A., Richards, S.J. (1999): Global amphibian declines: A problem in applied ecology. Annu. Rev. Ecol. Syst. 30: 133-165.
Banks, B., Beebee, T.J. (1987): Factors influencing breeding site choice by the pioneering amphibian Bufo calamita. Ecography. 10: 14-21.
Beebee, T.J. (1997): Changes in dewpond numbers and amphibian diversity over 20 years on chalk downland in Sussex, England. Biol. Conserv. 81: 215-219.
Blank, L., Blaustein, L. (2012): Using ecological niche modeling to predict the distributions of two endangered amphibian species in aquatic breeding sites. Hydrobiologia 693: 157-167.
Borzée, A., Heo, K., Jang, Y. (2018): Relationship between agro-environmental variables and breeding Hylids in rice paddies. Sci. Rep. 8: 8049.
Burnham, K.P., Anderson, D.R. (2004): Multimodel inference: Understanding AIC and BIC in model selection. SMR 33: 261-304.
Crossland, M.R., Alford, R.A. (1998): Evaluation of the toxicity of eggs, hatchlings and tadpoles of the introduced toad Bufo marinus (Anura: Bufonidae) to native Australian aquatic predators. Aust. J. Ecol. 23: 129-137.
Crump, M.L. (1991): Choice of oviposition site and egg load assessment by a treefrog. Herpetol. 47: 308-315.
Cunningham, J.M., Calhoun, A.J., Glanz, W.E. (2007): Pond‐breeding amphibian species richness and habitat selection in a beaver‐modified landscape. J. Wildl. Manag. 71: 2517-2526.
Decena, S.C.P., Avorque, C.A., Decena, I.C.P., Asis, P.D., Pacle, B. (2020): Impact of habitat alteration on amphibian diversity and species composition in a lowland tropical rainforest in Northeastern Leyte, Philippines. Sci. Rep. 10: 10547.
Diniz‐Filho, J.A.F., Rangel, T.F.L., Bini, L.M. (2008): Model selection and information theory in geographical ecology. Glob. Ecol. Biogeogr. 17: 479-488.
Dixo, M., Metzger, J.P., Morgante, J.S., Zamudio, K.R. (2009): Habitat fragmentation reduces genetic diversity and connectivity among toad populations in the Brazilian Atlantic Coastal Forest. Biol. Conserv. 142: 1560-1569.
Durso, A.M., Willson, J.D., Winne, C.T. (2011): Needles in haystacks: Estimating detection probability and occupancy of rare and cryptic snakes. Biol. Conserv. 144: 1508-1515.
Edgerly, J.S., McFarland, M., Morgan, P., Livdahl, T. (1998): A seasonal shift in egg‐laying behaviour in response to cues of future competition in a treehole mosquito. J. Anim. Ecol. 67: 805-818.
Environmental Geographic Information Service (EGIS). (2019): Land cover map Subdivision. Sejong, South Korea. URL: https://egis.me.go.kr/intro/land.do/ Accessed 20 September 2024.
Evans, M., Yáber, C., Hero, J.M. (1996): Factors influencing choice of breeding site by Bufo marinus in its natural habitat. Copeia 1996: 904-912.
Fleishman, E., Murphy, D.D., Sjögren-Gulve, P. (2002): Modeling species richness and habitat suitability for taxa of conservation interest. In: Predicting Species Occurrences: Issues of Accuracy and Scale, pp. 506–517. Scott, J.M., Heglund, P.J., Morrison, M.L., Eds, Washington, DC, Island Press.
Guisan, A., Zimmermann, N.E. (2000): Predictive habitat distribution models in ecology. Ecol. Model. 135: 147-186.
Hirzel, A.H., Hausser, J., Chessel, D., Perrin, N. (2002): Ecological‐niche factor analysis: How to compute habitat‐suitability maps without absence data?. Ecol. 83: 2027-2036.
Hu, S. (2007): Akaike information criterion. CRSC 93: 1-20.
International Union for Conservation of Nature (IUCN): (2022): Bufo gargarizans. IUCN red list Threatened Species. Cambridge, UK. URL: https://www.iucnredlist.org/species/78017839/197248539/ Accessed 20 September 2024.
Jeong, J.S. (2017): On the population density of Bufo bufo gargarizans in the wetland of nearby Seomjin river and its habitats. Master’s Theses. Kong Ju National University, Kong Ju.
Johnson, J.B., Omland, K.S. (2004): Model selection in ecology and evolution. TREE. 19: 101-108.
Jowers, M.J., Downie, J.R. (2005): Tadpole deposition behaviour in male stream frogs Mannophryne trinitatis (Anura: Dendrobatidae). J. Nat. Hist. 39: 3013-3027.
Kiesecker, J.M., Chivers, D.P., Blaustein, A.R. (1996): The use of chemical cues in predator recognition by western toad tadpoles. Anim. Behav. 52: 1237-1245.
Kruse, K.C., Stone, B.M. (1984): Largemouth bass (Micropterus salmoides) learn to avoid feeding on toad (Bufo) tadpoles. Anim. Behav. 32: 1035-1039.
Lee, J.H., Jang, H.J., Seo, J.H. (2011): Ecological guide book of herpetofauna in Korea, pp. 64-66. Incheon, Republic of Korea. National Institute of Environmental Research.
Lee, S.H. (2003): A comparative histological study on some salientia inhabitated in Korea. Gwangju, Honam University Academic papers 24: 289-303.
Lim, M.A., Choi, W.K., Eo, S.H., Choi, H.Y., Lee, J.S., Yang, W.K., Woo, S.H., Park, Y.S. (2005): Detection of bufotoxins in toad venom and toad eggs. Kor. J. Forensic Sci. 6: 125-130.
Lin, Y.S., Lehtinen, R.M., Kam, Y.C. (2008): Time-and context-dependent oviposition site selection of a phytotelm-breeding frog in relation to habitat characteristics and conspecific cues. Herpetol. 64: 413-421.
Lunghi, E., Manenti, R., Ficetola, G.F. (2015): Seasonal variation in microhabitat of salamanders: Environmental variation or shift of habitat selection? PeerJ 3: e1122.
Luo, Z., Hu, M., Hong, M., Li, C., Gu, Q., Gu, Z., Liao, C., Zhao, M., Wu, H. (2015): Outbreeding avoidance as probable driver of mate choice in the Asiatic toad. J. Zool. 295: 223-231.
Luo, Z., Li, C., Wang, H., Zhao, M., Gu, Q., Gu, Z., Liao, C., Wu, H. (2014): Mutual mate choice in the Asiatic toad, Bufo gargarizans, exerts stabilizing selection on body size. Chin. Sci. Bull. 59: 38-45.
MacKenzie, D.I., Bailey, L.L. (2004): Assessing the fit of site-occupancy models. JABES 9: 300-318.
Matushkina, K.A., Makarov, O., Afrin, K., Kidov, A., Litvinchuk, S. (2022): Patterns of reproduction, growth and development in Asiatic toads, (Bufo gargarizans Cantor, 1842) from Russia and China: An experimental comparison. Herpetol. Notes. 15: 123-138.
Murphy, P.J. (2003a): Does reproductive site choice in a neotropical frog mirror variable risks facing offspring?. Ecol. Monogr. 73: 45-67.
Murphy, P.J. (2003b). Context-dependent reproductive site choice in a Neotropical frog. Behav. Ecol. 14: 626-633.
National Institute of Ecology (NIE). (2018): The 4th National Natural Environment Survey Amphibians and Reptiles. National Institute of Ecology. Seocheon, Republic of Korea.
Newman, R.A. (1988): Adaptive plasticity in development of Scaphiopus couchii tadpoles in desert ponds. Evol. 42: 774-783.
Noland, R., Ultsch, G.R. (1981): The roles of temperature and dissolved oxygen in microhabitat selection by the tadpoles of a frog (Rana pipiens) and a toad (Bufo terrestris). Copeia 1981: 645-652.
Othman, S.N., Litvinchuk, S.N., Maslova, I., Dahn, H., Messenger, K.R., Andersen, D., Jowers, M.J., Kojima, Y., Skorinov, D.V., Yasumiba, K., Chuang, M., Chen, Y., Bae, Y., Hoti, J., Jang, Y., Borzee, A. (2022): From Gondwana to the Yellow Sea, evolutionary diversifications of true toads Bufo sp. in the Eastern Palearctic and a revisit of species boundaries for Asian lineages. Elife. 11: e70494.
Park, S.M., Kang, H.J., Ham, C.H., Lee, J.E., Park, S.W., Sung, H.C. (2021): A case study of the movement and behavior of Asian toads (Bufo gargarizans) during post-breeding season in the spring. Korean J. Herpetol. 12: 9-15.
Park, S.M., Rahman, M.M., Ham, C.H., Park, S.W., Kang, H.J., Sung, H.C. (2025): Movement patterns and habitat use within the home range of the Asiatic toad (Bufo gargarizans) prior to hibernation. Herpetozoa. 38: 77-85.
Park, S.M., Rahman, M.M., Park, H.R., Cheon, S.J., Kang, H.J., Park, S.W., Ham, C.H., Sung, H.C. (2024): Resting site selection and microhabitat use of an Asiatic toad (Bufo gargarizans; Anura: Amphibia) in South Korea. Curr. Herpetol. 43: 203-213.
Pearl, C.A., Adams, M.J., Leuthold, N., Bury, R.B. (2005): Amphibian occurrence and aquatic invaders in a changing landscape: Implications for wetland mitigation in the Willamette Valley, Oregon, USA. Wetlands 25: 76-88.
Ra, N.Y., Park, D., Cheong, S., Kim, N.S., Sung, H.C. (2010): Habitat associations of the endangered gold-spotted pond frog (Rana chosenica). Zool. Sci. 27: 396-401.
Resetarits Jr, W.J., Wilbur, H.M. (1989): Choice of oviposition site by Hyla chrysoscelis: Role of predators and competitors. Ecol. 70: 220-228.
Rudolf, V.H., Rödel, M.O. (2005): Oviposition site selection in a complex and variable environment: The role of habitat quality and conspecific cues. Oecologia 142: 316-325.
Semeniuk, M., Lemckert, F., Shine, R. (2007): Breeding-site selection by cane toads (Bufo marinus) and native frogs in northern New South Wales, Australia. Wildl. Res. 34: 59-66.
Semlitsch, R.D. (2000): Principles for management of aquatic-breeding amphibians. J. Wildl Manage. 64: 615-631.
Skelly, D.K., Werner, E.E., Cortwright, S.A. (1999): Long‐term distributional dynamics of a Michigan amphibian assemblage. Ecology. 80: 2326-2337.
Smith, D.C. (1983): Factors controlling tadpole populations of the chorus frog (Pseudacris triseriata) on Isle Royale, Michigan. Ecol. 64: 501-510.
Spieler, M., Linsenmair, K.E. (1997): Choice of optimal oviposition sites by Hoplobatrachus occipitalis (Anura: Ranidae) in an unpredictable and patchy environment. Oecologia 109: 184-199.
Stebbins, R.C., Cohen, N.W. (2021): A natural history of amphibians. Princeton University Press, Princeton, New Jersey.
Su, J., Han, M., Zhu, X., Liao, C., Tu, S., Luo, Z. (2020): Habitat selection of the Asiatic toad (Bufo gargarizans) during hibernation in the Badagongshan National Nature Reserve, central China. Salamandra 56: 16-26.
Sung, H.C., Park, O.H., Kim, S.K., Park, D.S., Park, S.R. (2007): Abundance and breeding migration of the Asian toad (Bufo gargarizans). J Ecol Environ 30: 287-292.
Yang, L., Huang, L., Zhang, H., Lee, P., Zhang, N., Cai, R., Li, E., Pan, T., Wu, X. (2023): Habitat suitability and distribution pattern response to global climate change in a widespread species, the Asiatic toad (Bufo Gargarizans). AHR 14:138-146.
Yang, L., Zhang, H., Zhang, N., Cai, R., Yan, P., Li, E., Pan, T., Wu, X. (2020): Population and habitat responses to the global climate change in a widespread species, the Asiatic toad (Bufo gargarizans). Authorea Preprints.
Yu, T.L., Gu, Y.S., Du, J., Lu, X. (2009): Seasonal variation and ontogenetic change in the diet of a population of Bufo gargarizans from the farmland, Sichuan, China. Biharean Biol. 3: 99-104.
Yu, T.L., Guo, Y.S. (2010): Overwintering habitat selection of Asiatic toad, Bufo gargarizans in southwestern China. Biharean Biol. 4: 15-18.