Submitted on: 2025, 3rd March; revised on: 2025, 26th July; accepted on: 2025, 30th July
Atlas of the herpetofauna of Batna Province and the Belezma Biosphere Reserve, north-eastern Algeria
1 Laboratory “Biodiversity, Biotechnology, and Sustainable Development”, Faculty of Natural and Life Sciences, University of Batna 2, Fesdis 05078, Batna, Algérie
2 Conservation des Forêts de la Wilaya de Batna, Nouvelle cité administrative la verdure, Batna 05000, Batna, Algérie
3 Laboratory “Agro-Biotechnology and Nutrition in Semi-Arid Zones”, Faculty of Natural and Life Sciences, Ibn Khaldoun University of Tiaret, Tiaret 14000, Tiaret, Algérie
4 Conservation des Forêts de la Wilaya de Tamanrasset, Hay-Echoumoua, Tamanrasset 11000, Tamanrasset, Algérie
5 Algerian Wildlife Watchers Association, Cité El-Hay, BT 53, nº 8, Hassi Bahbah 17002, Djelfa, Algérie
6 District des Forêts de Seggana, Seggana 05027, Batna, Algérie
7 Circonscription des Forêts d’Aïn Touta, N°57 Rue Tahar Louchene, Aïn Touta 05002, Batna, Algérie
8 Zana Ouled Sbaa, Zana El Beida 05071, Batna, Algérie
*Corresponding author. Email: idriss.bouam@univ-batna2.dz
Abstract. Algeria, the largest country in Africa, exhibits significant gaps in knowledge regarding species’ geographical distributions, particularly for herpetofauna. This deficiency is attributed in part to the country’s vast geographical expanse, limited local engagement in herpetological research, and persistent underfunding for biodiversity studies. This study presents the first comprehensive atlas of the herpetofauna of Batna Province, northeastern Algeria, including the Belezma Biosphere Reserve, marking a critical step toward developing a national herpetofaunal atlas. Based on 12 years of field surveys and a critical review of historical records spanning nearly 150 years, we document 47 species (four amphibians and 43 reptiles), including seven new provincial records and five species not reported in Batna for over 130 years. Our findings indicate that Batna harbours over two-fifths of Algeria’s known herpetofaunal diversity, highlighting its significance as a biodiversity hotspot. This study also provides Arabic common names for the recorded species to enhance public engagement while offering insights into species richness distribution patterns, conservation, and biogeography. We believe this atlas addresses critical knowledge gaps and will contribute to more accurate biodiversity assessments, while informing effective conservation planning in Algeria and the Mediterranean region.
Keywords. Amphibia, Reptilia, diversity, conservation, biogeography, North Africa.
Index
Unconfirmed or potentially erroneous records
The Wallacean shortfall, which refers to the paucity of information on species’ geographical distribution, poses limitations to both our understanding of biodiversity and the implementation of effective conservation strategies (Hortal et al., 2015). This knowledge gap is particularly evident in least developed countries and those with vast territories and remote locations (Titley et al., 2017), with arid regions being especially illustrative of this shortfall. The Saharo-Arabian Desert, in particular, suffers from strong sampling biases driven by limited accessibility (Velo-Antón et al., 2022). Moreover, within these arid environments, desert ecotones are topographically complex and biologically diverse transitional zones that remain critically under-surveyed despite their high ecological relevance (Liz et al., 2025). In this context, Algeria, Africa’s largest country, over 80% of which lies within the Sahara Desert, epitomizes this knowledge gap when it comes to vertebrates (Brito et al., 2014), notably amphibians and reptiles (Tolley et al., 2016; Beddek et al., 2018). The scarcity of data on Algerian herpetofauna is partly an artefact of the country’s vast geographical expanse and the remoteness of many of its regions (Weiss et al., 2018), both of which make comprehensive data collection challenging. Moreover, there appears to be a lack of interest among local researchers in herpetology, which is further compounded by Algeria’s position as one of the ten most severely underfunded nations for biodiversity conservation (Waldron et al., 2013). Notwithstanding these challenges, the last decade has witnessed a notable local resurgence of interest in Algeria’s herpetofauna, as evidenced by several recent published papers. These works have contributed to expanding our knowledge of range distributions of various herpetofaunal taxa (e.g., Saoudi et al., 2017; Bouam et al., 2019; Bouam and Merzougui, 2021; Bouam et al., 2022, 2024; Chedad et al., 2024), and to the discovery of previously undocumented species within the country (Rouag et al., 2016; Mouane et al., 2021; Boulaouad et al., 2023).
The province of Batna in northeastern Algeria has been the subject of few intermittent herpetological surveys. Noteworthy early explorations include Fernand Lataste’s expedition to Algeria in 1880, which included Batna (Lataste, 1881). Lataste’s notes and collections were later scrutinized by George Albert Boulenger, who reported additional taxa from the province (Boulenger, 1891). In the ensuing decade, Franz Werner conducted two herpetological surveys across eastern Algeria, encompassing Batna as well (Werner, 1892, 1894). Following Werner’s work, herpetofaunal research in Batna remained scarce for much of the 20th century, with only limited records provided by Gauthier (1932) and museum collections. More recent contributions include the investigations undertaken by Ulrich Joger and Wolfgang Bischoff in various parts of the Maghreb region (Joger and Bischoff, 1989). Their findings within Batna were extensively detailed in subsequent work by Bischoff and In Den Bosch (1991). The most comprehensive study on the reptiles of the province was carried out by Laurent Chirio, who conducted intensive surveys in the Aurès region, which covers most of southeastern Batna (Chirio and Blanc, 1997b). The recent phylogeographic study by Beddek et al. (2018) provided additional records for the province. While these contributions have collectively established a foundational understanding of the province’s herpetofaunal diversity, their focus has been largely restricted to mountainous massifs, with steppic plains receiving considerably less attention. Furthermore, much of the data from these studies are constrained by quality issues, including spatial biases such as insufficient geographical details, and taxonomic uncertainties resulting from subsequent nomenclatural and taxonomic changes (e.g., Wade, 2001; Carranza et al., 2006; Wagner et al., 2011; Rato et al., 2012; Denzer, 2021; Martínez-Freiría et al., 2021).
This study aims to provide the first comprehensive atlas of the herpetofauna of Batna Province, including the Belezma Biosphere Reserve. Based on data gathered over a decade of field surveys and a critical review of existing records spanning nearly 150 years, it offers an updated taxonomic list, reliable distribution maps, and insights into species richness patterns, conservation, and biogeography. Additionally, Arabic common names are included to enhance public engagement. This work serves as a foundational reference for future research and conservation efforts in Algeria and the broader Mediterranean region.
The study was conducted in Batna Province (hereafter Batna), northeastern Algeria, which covers 12,039 km² and includes the Belezma Biosphere Reserve (hereafter Belezma BR), recognized as a Key Biodiversity Area (UNESCO, 2015) (Fig. 1). Batna lies at the junction of two major geographical domains: the Tell Atlas to the north and the Saharan Atlas to the south, resulting in significant topographic variation, with elevations ranging from 74 m in the extreme south to 2,322 m at Djebel Mahmel (evaluated from a 30 m Copernicus elevation model; European Space Agency, 2024).
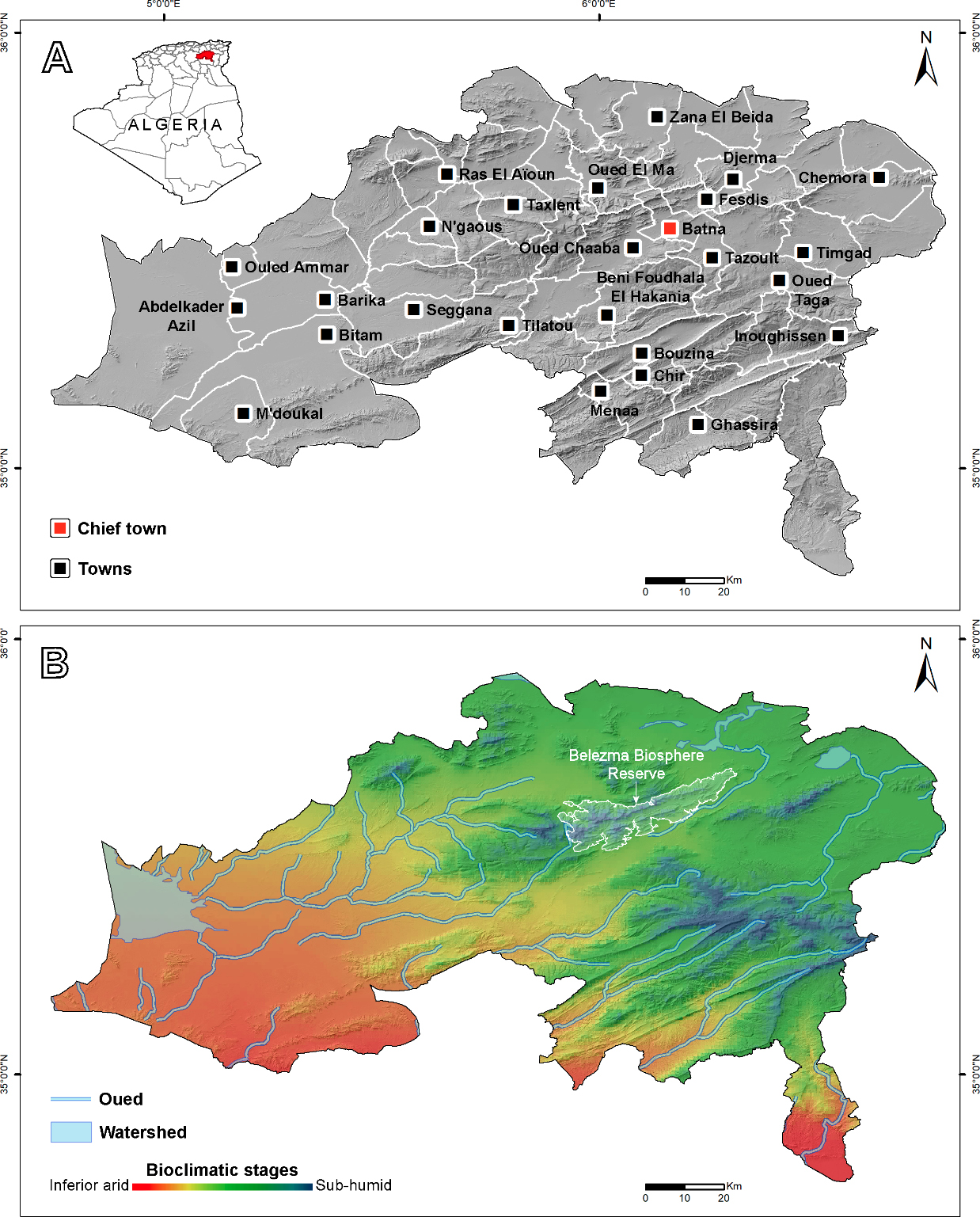
Physiographically, Batna can be broadly divided into three distinct regions: the northern high Tellian plains, characterized by flatlands interspersed with shallow brackish and saline lakes; the western high steppic plains, exhibiting pre-Saharan features and encompassing the eastern extent of Chott El Hodna, a large saline dry lake; and the central and southern mountainous massifs, notably the Aurès and Belezma ranges (Bernard and Ficheur, 1902).
Following Emberger’s climatic classification (Daget, 1977), Batna’s climate is predominantly Mediterranean with pronounced Saharan influences, leading to significant climatic variability across the province. The inferior and superior arid stages dominate the southern and western lowlands, the inferior and superior semi-arid stages characterize the northern regions and areas surrounding the mountainous massifs, while the sub-humid bioclimate prevails at higher elevations (Fig. 1B).
The hydrographic network of Batna is shaped by a relatively dense system of oueds – an Arabic term referring to river channels that experience brief, intense run-off during rainfall and prolonged dry periods. These oueds originate primarily in the mountains and drain into three main watersheds: Chott El Hodna, the shallow lakes of the high Tellian plains, and Chott Melrhir, located beyond the province’s southern boundary. Most oueds exhibit weak, highly seasonal flow, with the entire network dependent on precipitation and significantly affected by high evaporative rates (Mebarki, 2007).
Batna encompasses four ecoregions: Mediterranean conifer and mixed forests, Mediterranean woodlands and forests, Mediterranean dry woodlands and steppe, and North Saharan steppe and woodlands (Olson et al., 2001). These ecoregions host diverse habitats, from alpine grasslands and Atlas cedar forests at higher elevations to desert landscapes in the western lowlands (Fig. 2).

Data for this study were collected through opportunistic observations and systematic fieldwork by the authors from 2012 to 2024. Surveys were conducted diurnally and nocturnally employing various sampling methods, primarily visual surveys, rock flipping, and hand capture (McDiarmid et al., 2012). The dataset also included identifiable dead specimens and snake shed skins. Most individuals were photographed, and all had their precise geographic locations recorded via Global Positioning System (GPS) with corresponding dates. When multiple individuals of the same species were found nearby, a single spatial data point was recorded. This methodology resulted in 565 observations. Additionally, 15 observations were provided by local residents and colleagues through identifiable species photographs.
To complement our database, we integrated data from GBIF (2025), available literature, and undigitized museum or institutional collections (Appendices 1-2). Throughout the text, the following institutional abbreviations are used for museum collections, following Sabaj (2020): Museum of Comparative Zoology, Harvard University, Cambridge, USA (MCZ), Musée des Confluences, Lyon, France (MHNL), Muséum National d’Histoire Naturelle, Paris, France (MNHN), Natural History Museum, London, UK (NHMUK), Naturhistorisches Museum Basel, Basel, Switzerland (NMB), Natuurhistorisch Museum Rotterdam, Rotterdam, Netherlands (NMR), Zoologisches Forschungsmuseum Alexander Koenig, Bonn, Germany (ZFMK). Following Chowdhury et al. (2024), we also incorporated data sourced from Facebook. Species identification for photographic records, primarily from iNaturalist via GBIF (2025) and Facebook, was double-checked. Photographs that were of poor quality, lacked key diagnostic features, or corresponded to individuals that could not be reliably identified were systematically excluded. Only records with confident taxonomic identification, precise locality descriptions, and/or GPS coordinates with at least two decimal digits were retained. Records with coordinate uncertainties exceeding 10 km or those generically referencing “Batna” were also excluded, and duplicate records were removed. This process yielded a dataset of 231 occurrence records, with 49% originating from museum and institutional collections, 29% from online repositories (GBIF and Facebook), and 22% from published literature.
Taxonomy and nomenclature followed Frost (2024) for amphibians and Uetz et al. (2024) for reptiles. Authorship references for all taxa cited are provided in Appendix 3. English common names were sourced from Frost (2024), IUCN (2024), and Uetz et al. (2024). Arabic common names were primarily derived from El Maalouf (1985) and Ghaleb (1986). While several species had Arabic names listed in these references with scientific names consistent with current taxonomy, others appeared under synonyms, were represented only at the genus level, or were entirely absent. In the latter two cases, we retained the available Arabic name for the genus and translated the species epithet from the scientific or English name. For taxa not listed at all, complete Arabic names were constructed through direct translation from their scientific or English equivalents. IUCN conservation categories for the species reported in this study were obtained from the IUCN Red List of Threatened Species website (IUCN, 2024).
Chorotypes for each species were assigned following Chirio and Blanc (1997a), who applied this classification to the reptile fauna of the Aurès region, providing a relevant biogeographic framework for the study area. Eight distinct affinity groups were identified: Mediterranean taxa sensu stricto (MSS), distributed throughout the entire Mediterranean region; Mediterranean taxa sensu lato (MSL), extending their range eastward to the Caspian Sea region; West Mediterranean taxa (MWE), restricted to the western Mediterranean; Mediterranean taxa endemic to the Maghreb region (MAG); Saharan taxa sensu stricto (SSS), exclusively distributed in the African Sahara; Saharan taxa sensu lato (SSL), whose range extends into the deserts of Asia Minor; West Saharan taxa (SWE), confined to the western part of the Sahara; and Tropical taxa (TRO), predominantly found in the steppes and savannas south of the Sahara. For species exhibiting multiple chorotypes, we retained only the dominant biogeographic affinity that corresponds to the largest portion of the species’ natural range, while marginal affinities were not considered.
Distribution maps for individual species, the number and dominant source of observations, species richness, and chorotypes were generated using 6-arc-minute grids (~11 × 9 km), yielding 162 cells. This resolution offers a balance between spatial detail and sampling adequacy, minimizing false absences without compromising data quality, as recommended for atlas-based mapping (Robertson et al., 2010). Moreover, a similar grid size has been applied in herpetofaunal atlases for regions of comparable extent (Sillero et al., 2005; Cogălniceanu et al., 2014). All spatial analyses were conducted using ArcGIS v.10.8.
A total of 47 herpetofaunal species were documented from Batna. These are systematically presented in Table 1, along with their common names, IUCN conservation categories, and chorotypic classifications. The amphibian assemblage includes four anuran species – two frogs and two toads – from three families: Alytidae Fitzinger, 1843, Bufonidae Gray, 1825, and Ranidae Batsch, 1796, all of which occur in Belezma BR. The reptilian fauna comprises 43 species, 20 of which occur in Belezma BR. Snakes (Serpentes Linnaeus, 1758) account for 32.55% of the reptile species, spanning seven families: Colubridae Oppel, 1811 (seven species), Viperidae Oppel, 1811 (three species), and Erycidae Bonaparte, 1831, Lamprophiidae Fitzinger, 1843, Psammophiidae Dowling, 1967, and Elapidae Boie, 1827 each represented by a single species. Lacertid lizards (Lacertidae Batsch, 1788) follow at 27.90% (12 species), geckos (Phyllodactylidae Gamble, Bauer, Greenbaum & Jackman, 2008 and Gekkonidae Oppel, 1811) at 11.62% (five species), skinks (Scincidae Gray, 1825) at 9.30% (four species), and agamids (Agamidae Spix, 1825) at 6.97% (three species). The families Testudinidae Batsch, 1788, Geoemydidae Theobald, 1868, Trogonophidae Bonaparte, 1838, Chamaeleonidae Gray, 1825, and Varanidae Gray, 1827 are each represented by a single species.
In terms of conservation status, the herpetofauna of Batna encompasses six IUCN categories. Most species (74.46%, 35 species) are classified as Least Concern, while 10.63% (five species) are Near Threatened. Testudo graeca Linnaeus, 1758 and Vipera monticola Saint Girons, 1953 are listed as Vulnerable, Acanthodactylus blanci Doumergue, 1901 as Endangered, and Spalerosophis dolichospilus (Werner, 1923) as Data Deficient. Three species, namely Tarentola sp., Stenodactylus mauritanicus Guichenot, 1850, and Tropiocolotes chirioi Ribeiro-Júnior, Koch, Flecks, Calvo & Meiri, 2022, remain Not Evaluated.
Our results show that 105 out of 162 grid cells (64.81%) were sampled (Fig. 3). Most of the province was covered, with unsampled grids mainly along the borders and additional gaps in Chott El Hodna in western Batna. Despite these gaps, the overall sampling effort is considered substantial.
The number of observations per grid cell ranged from 1 to 44 (Fig. S1A), with original field records representing the dominant data source in most cells, whereas museum and institutional records predominated in parts of the Aurès region; cells where online and literature-based records were dominant were comparatively scarce and spatially scattered (Fig. S1B). Species richness per cell varied from 1 to 17 species (Fig. 3A). The spatial patterns of both observation counts and species richness were broadly congruent, with the highest concentrations within and around the Belezma range, extending south to the northern Aurès Mountains. Notable richness also occurs near Tazoult and within the quadrilateral delineated by Seggana, Djebel Metlili in Tilatou, Bitam, and Barika. The remaining areas of the province exhibit moderate to low species richness, with a marked contrast between the Aurès and the arid western regions, the latter displaying lower herpetofauna richness.
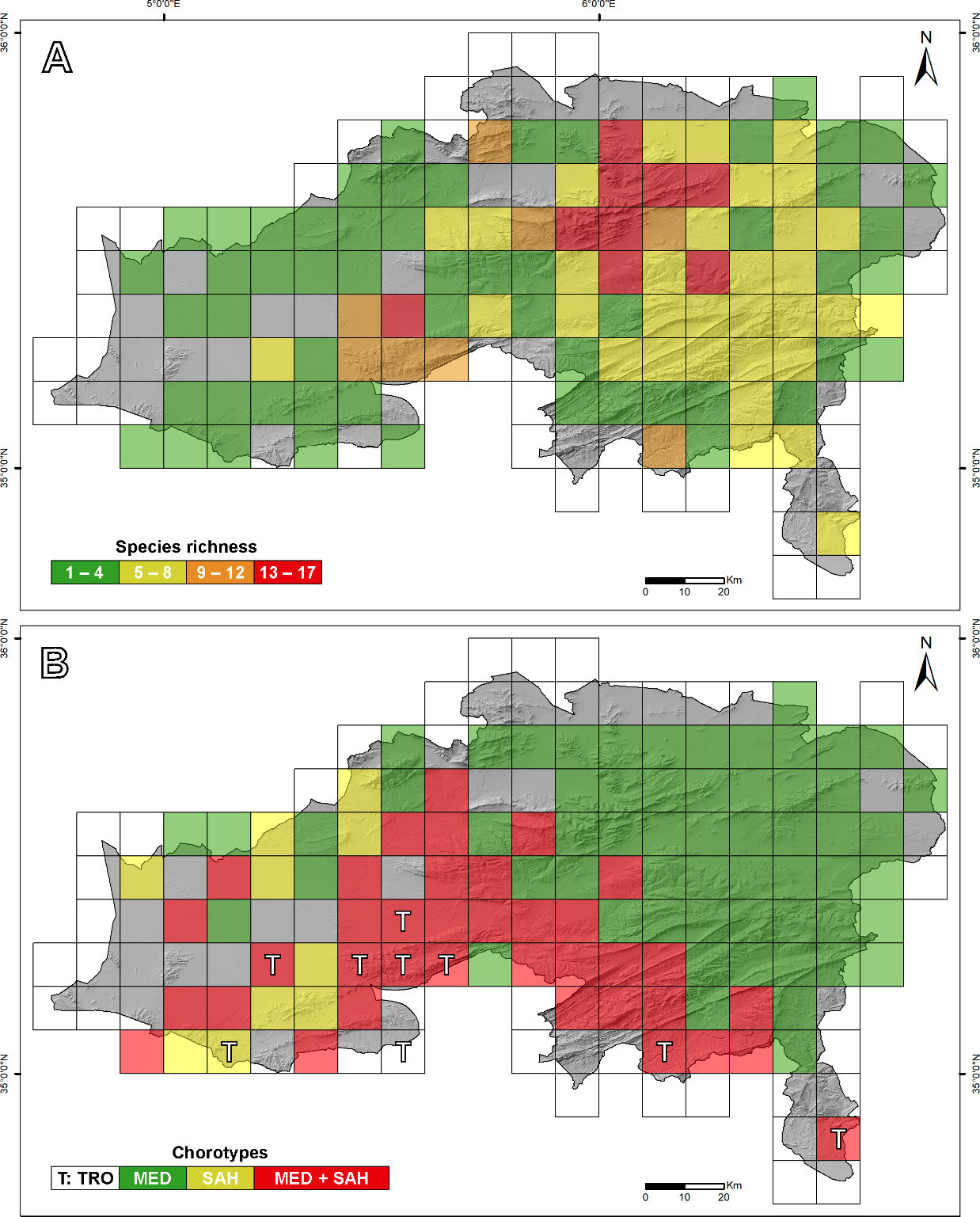
Biogeographically, Batna’s herpetofauna is predominantly Mediterranean, comprising about two-thirds of the recorded species, while Saharan taxa account for the remaining third. One species of tropical origin, Naja haje (Linnaeus, 1758), was also documented. Among Mediterranean taxa, Maghrebian species are the most prevalent, representing approximately 40%. Within Saharan taxa, all chorotypic categories are evenly distributed (Table 1). A clear spatial separation is observed (Fig. 3B), with Mediterranean species concentrated in the eastern half, while Saharan and Tropical species are primarily found in the southern and western regions. Notably, these chorotypes interdigitate along the western periphery of the Aurès and Belezma mountain ranges.
| Taxa | English name | Arabic name | IUCN Status | Chorotype |
|---|---|---|---|---|
| Class: Amphibia | ||||
| Order: Anura | ||||
| Family: Alytidae | ||||
| Discoglossus pictus Otth, 1837* | Painted Frog |  (Ḍaffāj Mulawwan) (Ḍaffāj Mulawwan) |
LC | MWE |
| Family: Bufonidae | ||||
| Sclerophrys mauritanica (Schlegel, 1841)* | Moorish Toad | 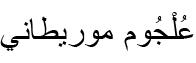 (ʿUlǧūm Mūrīṭānī) (ʿUlǧūm Mūrīṭānī) |
LC | MAG |
| Bufotes boulengeri (Lataste, 1879)* | African Green Toad |  (ʿUlǧūm Būlūnjī) (ʿUlǧūm Būlūnjī) |
LC | MSS |
| Family: Ranidae | ||||
| Pelophylax saharicus (Boulenger, 1913)* | North African Green Frog |  (Ḍifdaʿ ʾAkhḍar Shamāl ʾIfriqī) (Ḍifdaʿ ʾAkhḍar Shamāl ʾIfriqī) |
LC | MSS |
| Class: Reptilia | ||||
| Order: Testudines | ||||
| Family: Testudinidae | ||||
| Testudo graeca Linnaeus, 1758* | Spur-thighed Tortoise |  (Sulḥafāh Ighrīqiyyah) (Sulḥafāh Ighrīqiyyah) |
VU | MSL |
| Family: Geoemydidae | ||||
| Mauremys leprosa (Schweigger, 1812)* | Mediterranean Pond Turtle |  (Ḥamasah Mutawassiṭiyyah) (Ḥamasah Mutawassiṭiyyah) |
NT | MWE |
| Order: Squamata | ||||
| Suborder: Amphisbaenia | ||||
| Family: Trogonophidae | ||||
| Trogonophis wiegmanni Kaup, 1830* | Checkerboard Worm Lizard |  (Quhayqarān Wayghmān) (Quhayqarān Wayghmān) |
LC | MAG |
| Suborder: Lacertilia | ||||
| Family: Agamidae | ||||
| Agama bibronii A. Duméril in Duméril & Duméril, 1851* | Bibron’s Agama |  (Ḥirḏawn Bībrūn) (Ḥirḏawn Bībrūn) |
LC | MAG |
| Trapelus mutabilis (Merrem, 1820)**, ‡ | Desert Agama |  (ʿAḍrafūṭ Mutalawwin) (ʿAḍrafūṭ Mutalawwin) |
LC | SSS |
| Uromastyx acanthinura Bell, 1825** | North African Spiny-tailed Lizard |  (Ḍabb Shamāl ʾIfriqī) (Ḍabb Shamāl ʾIfriqī) |
NT | SWE |
| Family: Chamaeleonidae | ||||
| Chamaeleo chamaeleon (Linnaeus, 1758)* | Common Chameleon |  (Ḥirbāʾ Shāʾiʿah) (Ḥirbāʾ Shāʾiʿah) |
LC | MSS |
| Family: Phyllodactylidae | ||||
| Ptyodactylus oudrii Lataste, 1880 | Oudri’s Fan-footed Gecko |  (Abū Burayṣ ʾAudrī Marwḥī al-Arjul) (Abū Burayṣ ʾAudrī Marwḥī al-Arjul) |
LC | SWE |
| Tarentola deserti Boulenger, 1891 | Desert Wall Gecko |  (Abū Burayṣ Ṣaḥrāwī) (Abū Burayṣ Ṣaḥrāwī) |
LC | SWE |
| Tarentola sp.* | NA | NA | NE | MAG |
| Family: Gekkonidae | ||||
| Stenodactylus mauritanicus Guichenot, 1850 | Moorish Sand Gecko |  (Abū Burayṣ Ramlī Mūrīṭānī) (Abū Burayṣ Ramlī Mūrīṭānī) |
NE | MSS |
| Tropiocolotes chirioi Ribeiro-Júnior, Koch, Flecks, Calvo & Meiri, 2022**, ‡ | Chirio’s Gecko |  (Abū Burayṣ Shīryū) (Abū Burayṣ Shīryū) |
NE | SWE |
| Family: Varanidae | ||||
| Varanus griseus (Daudin, 1803)** | Desert Monitor |  (Warl Ṣaḥrāwī) (Warl Ṣaḥrāwī) |
LC | SSL |
| Family: Lacertidae | ||||
| Acanthodactylus boskianus (Daudin, 1802) | Bosc’s Fringe-toed Lizard |  (Thuʿbah Būsk) (Thuʿbah Būsk) |
LC | SSL |
| Acanthodactylus blanci Doumergue, 1901 | Blanc’s Fringe-toed Lizard |  (Thuʿbah Blān) (Thuʿbah Blān) |
EN | MAG |
| Acanthodactylus erythrurus (Schinz, 1833)* | Spiny-footed Lizard |  (Thuʿbah Ḥamrāʾ al-Dhayl) (Thuʿbah Ḥamrāʾ al-Dhayl) |
LC | MWE |
| Acanthodactylus bedriagai Lataste, 1881 | Bedriaga’s Fringe-fingered Lizard |  (Thuʿbah Badriyāghā) (Thuʿbah Badriyāghā) |
NT | MAG |
| Mesalina guttulata (Lichtenstein, 1823) | Small-spotted Desert Racer |  (Waḥīrah Raqṭāʾ) (Waḥīrah Raqṭāʾ) |
LC | SSS |
| Mesalina olivieri (Audouin, 1829)* | Olivier’s Desert Racer |  (Waḥīrah ʾŪlīfiyyī) (Waḥīrah ʾŪlīfiyyī) |
LC | MSS |
| Ophisops elegans Ménétries, 1832 | Snake-eyed Lizard |  (Siḥliyyat ʿAyn al-Thuʿbān al-ʾAnīqah) (Siḥliyyat ʿAyn al-Thuʿbān al-ʾAnīqah) |
LC | MSL |
| Ophisops occidentalis (Boulenger, 1887)* | Western Snake-eyed Lizard |  (Siḥliyyat ʿAyn al-Thuʿbān al-Gharbiyyah) (Siḥliyyat ʿAyn al-Thuʿbān al-Gharbiyyah) |
LC | MAG |
| Podarcis vaucheri (Boulenger, 1905)* | Vaucher’s Wall Lizard |  (Siḥliyyat Fūshī al-Jidāriyyah) (Siḥliyyat Fūshī al-Jidāriyyah) |
LC | MWE |
| Psammodromus algirus (Linnaeus, 1758)* | Algerian Sand Racer |  (Sunduwāh Jazāʾiriyyah) (Sunduwāh Jazāʾiriyyah) |
LC | MWE |
| Psammodromus blanci (Lataste, 1880)* | Blanc’s Sand Racer |  (Sunduwāh Blān) (Sunduwāh Blān) |
NT | MAG |
| Timon pater (Lataste, 1880)* | North African Ocellated Lizard |  (Siḥliyyat Maʿyūnah Shamāl Ifrīqiyyah) (Siḥliyyat Maʿyūnah Shamāl Ifrīqiyyah) |
LC | MAG |
| Family: Scincidae | ||||
| Chalcides mertensi Klausewitz, 1954 | Algerian Three-toed Skink |  (Dassāsah Jazāʾiriyyah Thulāthiyyat al-Aṣābiʿ) (Dassāsah Jazāʾiriyyah Thulāthiyyat al-Aṣābiʿ) |
LC | MAG |
| Chalcides ocellatus (Forskål, 1775)* | Ocellated Skink |  (Dassāsah Maʿyūnah) (Dassāsah Maʿyūnah) |
LC | MSS |
| Heremites vittatus (Olivier, 1804) | Bridled Skink |  (Ḥukʾah Mukhaṭṭaṭah) (Ḥukʾah Mukhaṭṭaṭah) |
LC | MSS |
| Scincus scincus (Linnaeus, 1758)** | Common Skink |  (Saqanqūr Shāʾiʿ) (Saqanqūr Shāʾiʿ) |
LC | SSL |
| Suborder: Serpentes | ||||
| Family: Erycidae | ||||
| Eryx jaculus (Linnaeus, 1758) | Javelin Sand Boa |  (Dassās Rumḥī) (Dassās Rumḥī) |
LC | MSL |
| Family: Lamprophiidae | ||||
| Malpolon insignitus (Geoffroy Saint-Hilaire, 1809)* | Eastern Montpellier Snake |  (Khuḍārī Mūnblīyah Sharqī) (Khuḍārī Mūnblīyah Sharqī) |
LC | MSL |
| Family: Psammophiidae | ||||
| Psammophis schokari (Forskål, 1775) | Schokari Sand Racer |  (Shuqārīy) (Shuqārīy) |
LC | SSL |
| Family: Colubridae | ||||
| Coronella girondica (Daudin, 1803)* | Southern Smooth Snake |  (Ḥuffāth Amghar) (Ḥuffāth Amghar) |
LC | MWE |
| Hemorrhois algirus (Jan, 1863) | Algerian Whip Snake |  (Ḥanash Jazāʾirī) (Ḥanash Jazāʾirī) |
LC | SSS |
| Hemorrhois hippocrepis (Linnaeus, 1758)* | Horseshoe Whip Snake |  (Ḥanash Ḥidwat al-Ḥiṣān) (Ḥanash Ḥidwat al-Ḥiṣān) |
LC | MWE |
| Lytorhynchus diadema (Duméril, Bibron & Duméril, 1854) | Crowned Leaf-nosed Snake |  (Ḥawfath Mutawwaǧ) (Ḥawfath Mutawwaǧ) |
LC | SSL |
| Macroprotodon mauritanicus Guichenot, 1850* | Moorish False Smooth Snake | 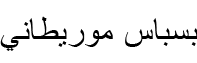 (Bisbās Mūrītānī) (Bisbās Mūrītānī) |
LC | MAG |
| Natrix maura (Linnaeus, 1758)* | Viperine Snake |  (Ḥinfish) (Ḥinfish) |
LC | MWE |
| Spalerosophis dolichospilus (Werner, 1923)** | Werner’s Diadem Snake |  (Arqam Wārnar) (Arqam Wārnar) |
DD | SWE |
| Family: Viperidae | ||||
| Cerastes cerastes (Linnaeus, 1758) | Desert Horned Viper |  (Afʿā Qarnāʾ) (Afʿā Qarnāʾ) |
LC | SSL |
| Daboia mauritanica (Gray, 1849)* | Moorish Viper | 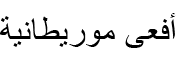 (Afʿā Mūrītānīyah) (Afʿā Mūrītānīyah) |
NT | MAG |
| Vipera monticola Saint Girons, 1953 | Mountain Viper | 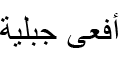 (Afʿā Jabalīyah) (Afʿā Jabalīyah) |
VU | MAG |
| Family: Elapidae | ||||
| Naja haje (Linnaeus, 1758)** | Egyptian Cobra |  (Ṣill Miṣrī) (Ṣill Miṣrī) |
LC | TRO |
Discoglossus pictus Otth, 1837 (Figs 4A; S2A). Algerian populations belong to a single lineage, except those in the extreme northwest (Beddek et al., 2018). In Batna, the species was first recorded by Lataste in Boulenger (1891), followed by Werner (1892, 1894) and Olivier (1894). The painted frog is the least frequently encountered amphibian in Batna, with a seemingly restricted distribution limited to mountain streams in sub-humid areas.
Sclerophrys mauritanica (Schlegel, 1841) (Figs 4B; S2B). Harris and Perera (2009) included a specimen from Batna in their genetic study, revealing minimal intraspecific variation across the species’ range in the Maghreb. Werner (1892) provided the earliest record, referring to it as Bufo mauritanicus Schlegel, 1841. The Moorish toad is common throughout the province and was recorded in a variety of habitats, particularly agricultural landscapes and stagnant sections of streams, and was occasionally found road-killed along mountain roads.
Bufotes boulengeri (Lataste, 1879) (Figs 4C; S2C). Dufresnes et al. (2019) confirmed that North African populations belong to the nominotypical subspecies, reporting three localities in Batna. The African green toad is relatively common in the province, occurring across arid to sub-humid areas.
Pelophylax saharicus (Boulenger in Hartert, 1913) (Figs 4D; S2D). Molecular studies (Beddek et al., 2018; Dufresnes et al., 2024) identified two lineages in Algeria: a western and an eastern lineage, the latter including Batna populations. Werner (1892) first recorded the species in Batna as Rana esculenta var. ridibunda Boettger, 1880. The North African green frog is the most common amphibian in the province and is encountered in virtually all types of aquatic habitats.

Testudo graeca Linnaeus, 1758 (Figs 5A; S2E). Molecular analyses identified two subspecies in Algeria, including Testudo graeca whitei Bennett in White, 1836 in Batna (Escoriza et al., 2022). Werner (1892, 1894) first documented the species in Lambesa (presently and hereafter referred to as Tazoult) as Testudo ibera Pallas, 1814, later reassigned to Testudo graeca graeca Linnaeus, 1758 by Loveridge and Williams (1957). The spur-thighed tortoise is increasingly rare in Batna, with remaining populations severely threatened by illegal wildlife trade driven by local demand for pet tortoises.
Mauremys leprosa (Schweigger, 1812) (Figs 5B; S2F). Algerian populations are recognized as the subspecies Mauremys leprosa saharica Schleich, 1996 (Bertolero and Busack, 2017). In Batna, it was first recorded by Lataste in Boulenger (1891) and later by Gauthier (1932) as Clemmys leprosa Schoepff in Schweigger, 1812. Subsequently, Loveridge and Williams (1957) assigned Batna’s populations to the subspecies Clemmys caspica leprosa Schoepff in Schweigger, 1812. The Mediterranean pond turtle persists in small, fragmented populations within a few northeastern wetlands.

Trogonophis wiegmanni Kaup, 1830 (Figs 6A; S2G). Phylogenetic analyses suggest that northeastern Algerian populations belong to the nominotypical subspecies (Salvi et al., 2018). The species’ earliest record in Batna dates to Strauch (1882), who reported two specimens acquired from Mr. Deyrolle in 1879. Strauch (1882) also mentioned Amphisbaena cinerea Vandelli, 1797 [=Blanus cinereus (Vandelli, 1797)] in Batna, a species confined to the Iberian Peninsula (Uetz et al., 2024). Boulenger (1891) noted that Fernand Lataste had not found B. cinereus in Algeria, suggesting a misidentification with T. wiegmanni. This was corroborated by Ineich et al. (2003) through re-examination of two specimens from Batna housed in the MHNL, originally catalogued as B. cinereus, which they identified as T. wiegmanni. The checkerboard worm lizard is sporadically encountered in Batna, where both the ‘mauve’ and ‘yellow’ forms, sensu Salvi et al. (2018), are documented.
Agama bibronii A. Duméril in Duméril & Duméril, 1851 (Figs 6A; S2H). Denzer (2021) posits that A. bibronii is the correct nomenclature, rather than Agama impalearis Boettger, 1874. The first formal record in Batna was documented by Saoudi et al. (2017). This species is common in rocky habitats but absent from the arid steppic plains in the province’s west. Batna populations may represent the species’ easternmost limit, as no records exist further east.
Trapelus mutabilis (Merrem, 1820) (Figs 6C; S3A). We report the first record of this species from Batna, specifically from multiple localities in the arid, rocky habitats of the province’s western region. Wagner et al. (2011) did not include genetic data from northeastern Algeria in their revision of the North African T. mutabilis complex; later, Wagner and Wilms (2013) provisionally assigned populations from this region to Trapelus boehmei Wagner, Melville, Wilms & Schmitz, 2011, though they acknowledged uncertainty. The closest populations, from Biskra – a province bordering Batna to the south – were identified by Werner (1893) as Agama aspera Daudin, 1802, a taxon Wagner et al. (2011) considered likely valid and distinct from T. boehmei. Chirio and Blanc (1997b) identified Biskra’s populations as T. mutabilis, although Wagner et al. (2021) questioned its occurrence in Algeria, while acknowledging its possible presence near the Libya-Tunisia-Algeria border. Given this ongoing taxonomic uncertainty, we provisionally assign Batna’s populations to T. mutabilis, pending further research.
Uromastyx acanthinura Bell, 1825 (Figs 6D; S3B). Tamar et al. (2018) assigned northeastern Algerian populations to U. acanthinura. Although documented in the neighbouring provinces of M’sila (Benelkadi et al., 2021) and Biskra (Chirio and Blanc, 1997b; as Uromastyx acanthinurus Bell, 1825), it had not been recorded in Batna. Our study provides the first formal record of this species in Batna, where it is notably prevalent in the rocky habitats of the western region.
Chamaeleo chamaeleon (Linnaeus, 1758) (Figs 6E; S3C). Although historical records of this species in Batna are scarce – limited to four specimens at the NMB collected by K. Seiler in 1946 without precise locality data, and a single specimen at the MNHN (RA-1997.6480) collected by Laurent Chirio – recent iNaturalist records (GBIF, 2025) and our observations indicate that it is more widespread than previously documented, occurring in arboreal habitats across all bioclimatic zones.

Ptyodactylus oudrii Lataste, 1880 (Figs 7A; S3D). Molecular studies identified North Algerian populations of the genus as belonging to P. oudrii, while also suggesting it may represent a species complex (Perera and Harris, 2010; Metallinou et al., 2015). The first record in Batna was documented by Lataste in Boulenger (1891), based on specimens collected in 1880 between Biskra and Batna, initially identified as Ptyodactylus lobatus (Geoffroy Saint-Hilaire, 1809) [=Ptyodactylus hasselquistii (Donndorff, 1798)]. Strauch (1887) referred to a specimen obtained from Mr. Deyrolle in 1879 from Batna as Ptyodactylus gecko Hasselq. [=Ptyodactylus hasselquistii], while another, collected by Ms. Westphal in 1893, is preserved at the ZFMK (HERP 002294) under P. oudrii. Loveridge (1947) treated Batna’s populations as the subspecies Ptyodactylus hasselquistii oudrii Anderson, 1898. Subsequent records from Batna (Chirio and Blanc, 1997b; Perera and Harris, 2010; Beddek et al., 2018) consistently identified it as P. oudrii. In Batna, Oudri’s fan-footed gecko is common in rocky habitats within semi-arid to arid bioclimatic zones, especially in the south.
Genus Tarentola Gray, 1825 (Figs 7B-C; S3E-F). Rato et al. (2012) included two specimens from Batna in their genetic analyses – one from Belezma BR and another from M’doukal in southwestern Batna. Both specimens were assigned to Clade XV sensu Rato et al. (2012) within the Tarentola fascicularis/deserti complex, a distinct lineage spanning Djelfa to Batna, with morphological evidence supporting its recognition as a separate species. Historically, two species have been recognized in Batna; Tarentola mauritanica (Linnaeus, 1758) (Werner, 1894; Bischoff and In Den Bosch, 1991; Chirio and Blanc, 1997b) and Tarentola deserti Boulenger, 1891 (MNHN-RA-1997.5057; MNHN-RA-1999.9460). Chirio and Blanc (1997b) assigned T. mauritanica from Batna to the nominotypical subspecies but noted that individuals from the eastern Aurès exhibited intermediate traits between T. mauritanica and Tarentola fascicularis (Daudin, 1802). Pending further research, and based on preliminary genetic analyses (pers. obs.), we provisionally recognize two species in Batna: Tarentola sp., encompassing all prior records of T. mauritanica, and T. deserti.
Stenodactylus mauritanicus Guichenot, 1850 (Figs 7D; S3G). Previously considered a subspecies of Stenodactylus sthenodactylus (Lichtenstein, 1823), this species was later confirmed as distinct through morphological and genetic analyses (Baha El Din, 2006; Metallinou et al., 2012). Historical records from Batna include Strauch’s (1887) report of two species, Stenodactylus wilkinsonii (Gray, 1842) and Stenodactylus guttatus Cuvier, 1829, based on specimens obtained from Mr. Deyrolle in 1879. Boulenger (1891) later synonymized both under S. guttatus [=S. sthenodactylus]. Olivier (1894) also reported S. guttatus from Batna based on a specimen collected by Mr. Pic. However, none of these historical records provide precise locality data. Our study confirms the presence of the Moorish sand gecko in several areas of the arid western steppes of Batna, where it is frequently observed at night.
Tropiocolotes chirioi Ribeiro-Júnior, Koch, Flecks, Calvo & Meiri, 2022 (Figs 7E; S3H). Described by Ribeiro-Júnior et al. (2022) based solely on morphological data, this micro-endemic species was initially known only from Biskra Province. Chirio and Blanc (1997b) had previously identified these populations as the nominotypical subspecies of Tropiocolotes tripolitanus Peters, 1880. Following Ribeiro-Júnior et al. (2022), we tentatively assign the Batna populations to T. chirioi due to their close geographic proximity to the type locality in Biskra, pending further phylogenetic analyses to confirm their taxonomic status. Our study provides the first record of Chirio’s Gecko in Batna, documenting multiple individuals under stones in rocky habitats across three localities in the southwestern region near the Biskra border.
Varanus griseus (Daudin, 1803) (Figs 7F; S4A). The desert monitor was recorded during our surveys in the arid steppic plains of the province’s western region. These observations represent the first formal records of the species in Batna. Notably, one specimen was found as a roadkill.

Acanthodactylus boskianus (Daudin, 1802) (Figs 8A; S4B). Acanthodactylus boskianus constitutes a species complex comprising considerable undescribed diversity, including nine potential candidate species (Liz et al., 2021). Tamar et al. (2016) assigned Algerian populations to the subspecies Acanthodactylus boskianus asper (Audouin, 1829). Lataste in Boulenger (1891) documented a single occurrence from N’gaous, referring to it as the ‘variety’ asper. In Batna, Bosc’s fringe-toed lizard predominantly inhabits sandy areas in the western region, where several populations appear well established.
Acanthodactylus erythrurus (Schinz, 1833) species complex (Figs 8B-C; S4C-D). The taxonomy of this species complex remains unresolved (Fonseca et al., 2009; Tamar et al., 2016; Miralles et al., 2020; Harris et al., 2024). Miralles et al. (2020) identified five main lineages across the Maghreb, suggesting the existence of at least five species, two of which occur in Algeria. Genetic analyses have included only a single specimen from Batna, which was assigned to the Central Algeria clade (Beddek et al., 2018; Harris et al., 2024). Based on available data, we provisionally recognize two taxa from this complex in Batna: Acanthodactylus blanci and Acanthodactylus erythrurus.
Acanthodactylus blanci, which may represent a valid species (Miralles et al., 2020), is rare in Batna. Two specimens (MNHN-RA-1997.6308 and RA-1997.6317) were collected by Laurent Chirio in 1993 from Col de Teniet El Hamadi in Chir. In 2024, we recorded a juvenile at this locality, identified as A. blanci by Philippe Geniez.
Populations of A. erythrurus from Batna are assigned to the subspecies Acanthodactylus erythrurus belli Gray, 1845 (Salvador, 1982; Chirio and Blanc, 1997b; Beddek et al., 2018), distinguished by extensive contact between the subocular scale and the upper lip (bellii conformation sensu Miralles et al. 2020). The first record of this species from Batna dates to 1893, based on a specimen housed at the ZFMK (HERP 022724), collected by Alexander Koenig in Tazoult. The species inhabits the eastern mountainous regions of Batna, particularly in semi-arid to sub-humid areas, and is the only member of the genus recorded within Belezma BR.
Acanthodactylus bedriagai Lataste, 1881 (Figs 8D; S4E). Endemic to Algeria (Nouira et al., 2022), this species belongs to the Acanthodactylus pardalis (Lichtenstein, 1823) complex, a species-group with intricate phylogenetic relationships (Fonseca et al., 2008; Tamar et al., 2016). Lataste (1881) first described A. bedriagai from multiple localities, including Batna, but later reclassified it as a ‘variety’ of Acanthodactylus savignyi (Audouin, 1829) (Lataste, 1885). Boulenger (1891, 1921) treated it as a ‘variety’ of A. pardalis, while Salvador (1982) reinstated it to species rank, including specimens from Batna. Chirio and Blanc (1997b) considered Batna populations as a subspecies of Acanthodactylus maculatus (Gray, 1838). Bedriaga’s fringe-fingered lizard is relatively common across the high Tellian plains and foothills of northern Batna but does not extend into the adjacent mountainous massifs.
Mesalina guttulata (Lichtenstein, 1823) (Figs 8E; S4F). Genetic studies (Kapli et al., 2008, 2015) indicate that M. guttulata represents a species complex. Sindaco et al. (2018) restricted its distribution to Africa, uncovering significant genetic variability and unresolved taxonomic issues. In Batna, only two specimens are preserved in the MNHN: one collected by Henri Martin without a precise locality (RA-0.8561), identified as Eremias guttulata watsonana (Stoliczka, 1872) [=Mesalina watsonana (Stoliczka, 1872)], and another from southern Batna collected by Laurent Chirio in 1984 (RA-1997.5864). We document several additional localities for the small-spotted desert racer, mainly in the southern half of the province.
Mesalina olivieri (Audouin, 1829) (Figs 8F; S4G). The Mesalina olivieri species complex exhibits considerable, yet undescribed, intraspecific diversity (Kapli et al., 2015; Pizzigalli et al., 2021). In Batna, it is known from few MNHN specimens collected by Laurent Chirio in the southeastern province, identified as the nominotypical subspecies. We report two additional localities, including one within the Belezma BR, where the species was observed in open habitats with patchy shrub cover.

Ophisops elegans Ménétries, 1832 (Figs 9A; S4H). It was first reported from Batna by Chirio and Blanc (1993), who identified it as the nominotypical subspecies, distinguishing it from Ophisops occidentalis (Boulenger, 1887) by its higher counts of supraocular scales and femoral pores. This species forms an isolated population in the Aurès Mountains, approximately 1,500 km from the nearest population in Libya. In Batna, it is confined to the southeastern mountain ranges, inhabiting drier, more steppic biotopes than O. occidentalis.
Ophisops occidentalis (Boulenger, 1887) (Figs 9B; S5A). It was first reported from Batna by Lataste in Boulenger (1891) as Ophiops occidentalis, based on specimens collected in 1880, followed by Werner (1892). Subsequent records include Bischoff and In Den Bosch (1991) and Chirio and Blanc (1993). The Western snake-eyed lizard is the most frequently observed reptile in Batna, occupying diverse biotopes but absent from the western arid steppic plains and higher elevations.
Podarcis vaucheri (Boulenger, 1905) (Figs 9C; S5B). Phylogeographic studies of the Podarcis hispanicus (Steindachner, 1870) complex suggest that Batna’s populations represents a distinct lineage (Lima et al., 2009; Kaliontzopoulou et al., 2011; Caeiro-Dias et al., 2018), which remains undescribed (Nouira et al., 2022). Due to their genetic and morphological similarity to P. vaucheri (Kaliontzopoulou et al., 2012), we provisionally assign these populations to this species. The earliest records from Batna, collected by Alexander Koenig in 1893, are housed in the ZFMK. Subsequent studies referred to Batna specimens as a subspecies of P. hispanicus (Bischoff and In Den Bosch, 1991; Chirio and Blanc, 1997b), and later P. vaucheri (Beddek et al., 2018). Vaucher’s wall lizard is common in Batna, particularly in the eastern half of the province, where it primarily inhabits elevated mountainous areas.
Psammodromus algirus (Linnaeus, 1758) (Figs 9D; S5C). Phylogenetic analyses confirm that all North African populations of P. algirus belong to the African clade (Verdú-Ricoy et al., 2010; Mendes et al., 2017, Faria and Harris, 2020). This species was initially documented in Batna by Werner (1892), with subsequent records provided by Bischoff and In Den Bosch (1991), Chirio and Blanc (1997b), Bouam et al. (2016), and Khelfaoui et al. (2023). The Algerian sand racer is mainly found in forested habitats across the mountain ranges of the eastern half of the province.
Psammodromus blanci (Lataste, 1880) (Figs 9E; S5D). In their phylogeographic study of the genus, Mendes et al. (2017) included two specimens of P. blanci from Belezma BR, revealing significant genetic differentiation between Batna and Tunisian populations. Originally described as Zerzoumia blanci Lataste, 1880 from Algiers and Batna, Boulenger (1891) later reclassified it as P. blanci. A specimen collected by Henri Martin in Batna in 1881 is housed in the MNHN (RA-0.8551). Early records from Batna, mainly from Tazoult, were documented by Lataste (1880a) and Werner (1892, 1894), with specimens deposited in NHMUK and ZFMK, including those collected by Alexander Koenig between 1892 and 1893. More recent records include specimens collected by Laurent Chirio (1984–1993) in the MNHN and those reported by Bischoff and In Den Bosch (1991), with several deposited in the ZFMK. Blanc’s sand racer has a limited distribution in Batna, primarily inhabiting Atlas cedar forests at higher elevations in the eastern half of the province. While Joger et al. (2009) reported an upper elevation limit of 1,200 m for P. blanci, we observed the species at 1,892 m on Djebel Chélia.
Timon pater (Lataste, 1880) (Figs 9F; S5E). Ahmadzadeh et al. (2016) confirmed the Batna populations as T. pater through phylogenetic analysis. The species was first reported in Batna by Lataste (1880b) as Lacerta ocellata pater Lataste, 1880. Subsequent records identified it as Lacerta pater Lataste, 1880 (Werner, 1892, 1894; Bischoff and In Den Bosch, 1991), Lacerta lepida pater (Lataste, 1880) (Chirio and Blanc, 1997b), and T. pater (Ineich et al., 2005). The North African ocellated lizard has a continuous distribution across the sub-humid mountain ranges of the eastern province, where it favours forested, elevated habitats (Bouam et al., 2017).

Chalcides mertensi Klausewitz, 1954 (Figs 10A; S5F). Werner (1894) first recorded this species in Batna, from Tazoult, as Chalcides tridactylus Laurenti, 1768 [=Chalcides chalcides (Linnaeus, 1758)]. Chirio and Blanc (1997b) later identified it as C. mertensi from the same locality. Our findings confirm its presence in this area and document a new locality in southwestern Batna. According to Chirio and Blanc (1997a), the Algerian three-toed skink likely represents relictual populations in Batna, persisting in humid microhabitats within the province.
Chalcides ocellatus (Forskål, 1775) (Figs 10B; S5G). Werner (1894) first reported this species from Batna. Chirio and Blanc (1997b) assigned all specimens to the subspecies Chalcides ocellatus tiligugu (Hermann, 1783), distinguished by dark and light longitudinal laterodorsal bands. However, a specimen from Ouled Ammar (NMR 998800000275) was identified as Chalcides ocellatus subtypicus Werner, 1931, suggesting the presence of both subspecies. The ocellated skink is widespread across Batna, commonly found under stones or near rocky structures.
Heremites vittatus (Olivier, 1804) (Figs 10C; S5H). Baier et al. (2017) suggested that H. vittatus may represent a species complex, with populations from Tunisia, Libya, and likely Algeria forming a distinct mitochondrial lineage. While common in the Eastern Mediterranean, its distribution in North Africa is limited, with few records from Algeria. In Batna, a historical specimen (MNHN-RA-1974.322) collected by Henri Martin in 1884 was identified as Mabuya vittata (Olivier, 1804), although it lacks precise locality data. We confirm the species’ presence in Batna through an observation of three individuals along an irrigation canal in the western arid steppes.
Scincus scincus (Linnaeus, 1758) (Figs 10D; S6A). The northernmost record of S. scincus in Algeria was reported by Olivier (1894) as Scincus officinalis Laurenti, 1768 from Oumache in southern Biskra Province. However, Chirio and Blanc (1997b) questioned its presence there, suggesting its northern limit lies in the Great Oriental Erg, about 100 km south of Biskra. We document the first confirmed record of the common skink in Batna, from an isolated sand dune area approximately 70 km northwest of Oumache. This population appears well-established and likely represents an isolated population due to the absence of suitable sand dune habitats in the surrounding areas.

Eryx jaculus (Linnaeus, 1758) (Figs 11A; S6B). Lataste in Boulenger (1891) first reported the Javelin sand boa in Batna from Barika and N’gaous. We observed this species only twice during our surveys. Its distribution in the province appears sporadic, likely due to its cryptic behaviour (Geniez, 2015).
Malpolon insignitus (Geoffroy Saint-Hilaire, 1809) (Figs 11B; S6C). The molecular study by Carranza et al. (2006) elevated M. insignitus to species status. Morphologically, it differs from Malpolon monspessulanus (Hermann, 1804) by the absence of a dark ‘saddle’ marking on the foreparts of males. Geniez (2015) and Trape (2023) assigned the northeastern Algerian populations to M. insignitus, although the recent IUCN assessment (Aghasyan et al., 2021) inadvertently reported the species as absent from Algeria. The first record of M. insignitus in Batna was provided by Werner (1894) as Coelopeltis lacertina (Wagler, 1830), with later reports by Bischoff and In Den Bosch (1991) as M. monspessulanus and Chirio and Blanc (1997b) as the subspecies Malpolon monspessulanus insignitus (Geoffroy Saint-Hilaire, 1809). In Batna, the Eastern Montpellier snake is relatively common, although road mortality poses a significant threat to its population.
Psammophis schokari (Forskål, 1775) (Figs 11C; S6D). A specimen collected from Ghoufi in Ghassira, southern Batna, was included in the genetic analyses by Rato et al. (2007) and Gonçalves et al. (2018), confirming that Algerian populations form a monophyletic lineage. This specimen represents the only recorded instance of the species in Batna. Our documentation is limited to three sightings in semi-arid to arid areas in the southern and northwestern parts of the province.

Coronella girondica (Daudin, 1803) (Figs 12A; S6E). The presence of this species in Batna is documented by two specimens housed at the ZFMK (HERP 023220–023221), collected by Alexander Koenig between 1892 and 1893, without specific locality data. The Southern smooth snake appears rare in Batna, as we encountered it only twice during our surveys at relatively high elevations of 1,433 and 1,833 m within the Belezma mountain ranges.
Hemorrhois algirus (Jan, 1863) (Figs 12B; S6F). The only previous record of this species in Batna was from Ghoufi in the province’s south (Abreu, 2017). We document several new localities in semi-arid and arid environments across the southern half of Batna, including individuals with the characteristic ‘horseshoe’ mark and others with uniformly darker heads.
Hemorrhois hippocrepis (Linnaeus, 1758) (Figs 12C; S6G). This species was first recorded in Batna as Zamenis hippocrepis (Linnaeus, 1758) by Lataste in Boulenger (1891) and later by Werner (1892). Chirio and Blanc (1997b) documented it as Coluber hippocrepis Linnaeus, 1758 from two localities, with an additional site reported by Abreu (2017). The horseshoe whip snake is prevalent in the northeastern part of the province but absent from the arid western and southern regions, where H. algirus appears to replace it. It is frequently observed near urban areas and is often persecuted due to its large size and human fear, despite being non-venomous.
Lytorhynchus diadema (Duméril, Bibron & Duméril, 1854) (Figs 12D; S6H). This species, associated with desert sandy habitats (Geniez, 2015), was reported in Batna by Boulenger (1891) based on a specimen in the Saint Petersburg Museum, though specific details were not provided. The crowned leaf-nosed snake appears rare in Batna, as we documented it only once during a nocturnal survey in a sandy area of the province’s western arid region.
Macroprotodon mauritanicus Guichenot, 1850 (Figs 12E; S7A). Molecular analyses by Carranza et al. (2004) identified the northeastern Algerian populations as M. mauritanicus. Initially reported in Batna by Lataste in Boulenger (1891) as Macroprotodon cucullatus (Geoffroy Saint-Hilaire, 1809), subsequent studies (Wade, 1988; Busack and McCoy, 1990) assigned Batna specimens to Macroprotodon cucullatus mauritanicus Guichenot, 1850. Wade (2001) later elevated this subspecies to species rank, considering six specimens from Batna, including Lataste’s collections. Bischoff and In Den Bosch (1991) provided precise locality records, with one specimen preserved in ZFMK (HERP 049602). The Moorish false smooth snake is relatively common in Batna but appears absent in arid regions.
Natrix maura (Linnaeus, 1758) (Figs 12F; S7B). Genetic analyses by Barata et al. (2008), including a specimen from Batna, revealed significant genetic divergence between populations from eastern Algeria and Tunisia compared to those in western Algeria, Morocco, and Europe. Historical records of N. maura in Batna include a specimen collected by Alexander Koenig in 1893 and Werner’s (1894) report from Tazoult as Tropidonotus viperinus (Sonnini & Latreille, 1802). The viperine snake is the only semi-aquatic snake species in Batna, frequently observed in aquatic habitats.
Spalerosophis dolichospilus (Werner, 1923) (Figs 12G; S7C). This species remains one of the least studied within its genus (Yadollahvandmiandoab et al., 2023) and had not been previously reported in Batna. It appears rare in the province, with only two nocturnal observations provided by local residents from the western arid regions. We recorded a single individual in the same locality indicated by one of these residents, in an arid, stony area sparsely vegetated with shrubs.

Cerastes cerastes (Linnaeus, 1758) (Figs 13A; S7D). Strauch (1862) mentioned this species in Batna, referring to it as Vipera cerastes (Linnaeus, 1758) and noting its purported abundance, though without personal observations or specific locality data. Our findings confirm the presence of the desert horned viper in the arid western and southern regions, where it primarily inhabits sandy substrates and gravel plains with sparse vegetation.
Daboia mauritanica (Gray, 1849) (Figs 13B; S7E). Martínez-Freiría et al. (2017) proposed treating all North African populations as D. mauritanica, invalidating Daboia deserti (Anderson, 1892). Lataste in Boulenger (1891) first reported this species near Batna as Vipera lebetina (Linnaeus, 1758) [=Macrovipera lebetinus (Linnaeus, 1758)] from M. Hénon’s collection. Its presence in Batna was formally confirmed over a century later from a single documented locality north of the Belezma BR (Martínez-Freiría et al., 2017). Our findings indicate the Moorish viper is relatively common in rocky habitats across the province’s eastern half.
Vipera monticola Saint Girons, 1953 (Figs 13C; S7F). Martínez-Freiría et al. (2021) confirmed that North African populations belong to V. monticola, while Vipera latastei Bosca, 1878 is restricted to the Iberian Peninsula, assigning Algerian populations to the subspecies Vipera monticola saintgironsi Martínez-Freiría, Fahd, Larbes & Brito in Martínez-Freiría et al., 2021. In Batna, the mountain viper has only been documented from Djebel Chélia in the eastern part of the province (Bouam et al., 2019), which remains the sole confirmed locality.
Naja haje (Linnaeus, 1758) (Figs 13D; S7G). This species has not been previously documented in Batna. We report its occurrence in the arid southern and western regions, particularly in palm groves and rocky landscapes. These records represent the first formal documentation of the Egyptian cobra in the province. Unfortunately, many observed specimens were deceased, often killed by local residents.

Unconfirmed or potentially erroneous records
Emys orbicularis (Linnaeus, 1758). Gauthier (1932) reported several individuals from “Fontaine-Chaude” (now Ain Skhouna) in northeastern Batna. However, despite extensive surveys, we found no evidence of this species, and the region’s arid climate appears unsuitable for its persistence (Gherbi et al., 2023). Given its resemblance to M. leprosa (Schleich et al., 1996), the reported individuals were likely misidentified.
Chalcides boulengeri Anderson, 1892. Two historical specimens of Sphenops boulengeri (Anderson, 1892) [=Chalcides boulengeri] are tentatively attributed to Batna. The first, housed at the MNHN (RA-0.8578), was collected by Henri Martin in 1884 and labelled “Batna”. The second, held at the MCZ (R-112204), was also collected by H. Martin but recorded as from “Biskra (province of Batna)”. The latter locality suggests that the historical records of C. boulengeri in Batna likely reflect the administrative organization of Algeria during the French colonial period, when Biskra was included as a district within the department of Batna. The occurrence of C. boulengeri in Biskra was confirmed by Chirio and Blanc (1997b), but we found no evidence of the species within Batna’s current boundaries.
Tarentola neglecta Strauch, 1887. Strauch (1887) described Tarentola neglecta and Tarentola angusticeps Strauch, 1887 from Batna, based on two specimens purchased in Paris from Mr. Deyrolle, who claimed they originated from Batna. However, Strauch explicitly noted that he could not verify the accuracy of the locality. Boulenger (1887) considered both specimens to represent T. neglecta, while Olivier (1894) questioned their origin, arguing that T. neglecta is strictly desertic. Given the uncertain provenance, the species’ current known distribution (Rato et al., 2012), and the results of our field surveys, we suggest that the occurrence of T. neglecta in Batna likely reflects an imprecise locality record.
Stenodactylus petrii Anderson, 1896. This species was reported from Batna by Angel (1923) and Loveridge (1947), both relying on Strauch (1887). Strauch, as in the case of Tarentola neglecta, obtained specimens from Mr. Deyrolle, identifying them as Stenodactylus wilkinsonii and Stenodactylus guttatus. However, Boulenger (1891) synonymized these ‘forms’ under S. guttatus [= Stenodactylus sthenodactylus]. Loveridge (1947) also suggested that Strauch’s records likely originated from southern Algeria rather than Batna. Given uncertainties in specimen identification and locality, combined with our fieldwork findings, we propose that the occurrence of S. petrii in Batna likely reflects a misidentification with S. mauritanicus or an imprecise locality record.
This study marks a significant contribution to the understanding of Algerian herpetofauna, particularly given the lack of a national atlas and the reliance on distributional data from general works on North Africa (e.g., Schleich et al., 1996; Sindaco and Jeremčenko, 2008, Geniez, 2015; Trape, 2023), which, while providing valuable insights, are often based on outdated literature and museum collections, limiting their contemporary relevance.
The present work provides extensive spatial coverage, encompassing approximately two-thirds of the province’s area. This estimate is conservative, as distinguishing between unsampled grid cells and those with no observations was challenging due to the opportunistic nature of many records. Most empty grids are located along the province’s borders, a common limitation in atlases (e.g., Cogălniceanu et al., 2014; Burriel-Carranza et al., 2019). Since border grids cover less provincial area than interior grids, the actual spatial coverage is likely more comprehensive.
Original field data constituted the dominant source of observations in the majority of grid cells, reflecting the substantial sampling effort conducted across the province. The prevalence of museum and institutional records in parts of the Aurès region is unsurprising, reflecting the intensive herpetological work carried out in this region by Chirio and Blanc (1997a, b), whose surveys yielded numerous specimens now housed in the collections of the MNHN. Grid cells dominated by records from online or literature sources were both scarce and spatially scattered, emphasizing their more localized and opportunistic nature. These areas, where original field data remain limited, should be prioritized in future surveys to improve spatial completeness and data consistency.
The observed pattern of species richness across Batna is largely congruent with the distribution of observation records, which is expected given that species richness generally increases with sampling effort and spatial extent, highlighting the need for intensified surveys in the less-sampled western region. However, this apparent pattern should be interpreted with caution, as the number of records per grid cell is not always a reliable proxy for actual sampling effort in atlases (Robertson et al., 2010), and should therefore be assessed in conjunction with the individual species distribution maps, which in our study display contiguous patterns for numerous species, indicative of well-documented ranges across the province.
The amphibian assemblage of Batna, though seemingly species-poor, reflects the naturally depauperate amphibian diversity characteristic of North Africa (Escoriza and Ben Hassine, 2019). Nonetheless, these species collectively represent 25% of Algeria’s documented amphibian fauna (Frost, 2024), highlighting the regional significance of Batna’s amphibian diversity within the national context.
In contrast, the reptile assemblage of Batna comprises 43 species, including seven newly recorded for the province: Trapelus mutabilis, Uromastyx acanthinura, Tropiocolotes chirioi, Varanus griseus, Scincus scincus, Spalerosophis dolichospilus, and Naja haje, all representing genera previously unrecorded in the province, with T. mutabilis and T. chirioi tentatively assigned pending further taxonomic confirmation. With the exception of the Tropical Naja haje, these species are of Saharan chorotypes and were predominantly recorded in the arid western regions of Batna – an area that has remained virtually unexplored, with only sporadic historical records from the localities of Barika, M’doukal, N’gaous, and Ouled Ammar (Boulenger, 1891; Olivier, 1894; Salvador, 1982; Rato et al., 2012; GBIF, 2025). This finding represents a significant 19.44% increase in the known reptile diversity of Batna, further corroborating the pronounced Wallacean shortfall across the Algerian Sahara and its adjacent arid zones (Brito et al., 2014; Tolley et al., 2016). Additionally, our study confirmed the presence of Acanthodactylus boskianus, Heremites vittatus, Coronella girondica, Lytorhynchus diadema, and Cerastes cerastes, species that had not been reported in the region for over 130 years. Conversely, historical records of Emys orbicularis, Chalcides boulengeri, Tarentola neglecta, and Stenodactylus petrii were excluded due to potential misidentifications, vague locality data, or insufficient supporting evidence. However, the possible presence of C. boulengeri in the province merits further investigation, particularly in the sandy arid landscapes of the western region, where Scincus scincus – a species with comparable ecological requirements – is currently established.
The reptile fauna documented in Batna represents 42.57% of Algeria’s known terrestrial reptile diversity (Rouag et al., 2024), an exceptional proportion considering that the province accounts for only 0.5% of the country’s area. This richness surpasses that reported in the few recent regional herpetofaunal surveys conducted in Algeria (Rouag and Benyacoub, 2006; Benelkadi et al., 2021; Bezaz et al., 2021; Mouane et al., 2024), despite the limited number of such studies for comparison. Remarkably, Batna’s reptile diversity also exceeds that of several Mediterranean countries (Cox et al., 2006), further highlighting its significance as a biodiversity hotspot for reptiles both within Algeria and across the Mediterranean region. Despite this exceptional diversity, substantial gaps remain in the genetic characterization of Batna’s herpetofauna, a deficiency observed across much of Algeria (Beddek et al., 2018). Future molecular studies should focus on Trapelus, Tarentola, Tropiocolotes, Acanthodactylus, and Ophisops to resolve their unclear phylogenetic relationships.
Eight species (17.02%) of Batna’s herpetofauna have an unfavourable conservation status, with Acanthodactylus blanci, Testudo graeca, and Vipera monticola being the most threatened. Acanthodactylus blanci and V. monticola are at risk due to their restricted extent of occurrence (Nouira and Joger, 2006; Martínez-Freiría et al. 2024), though no immediate threats were observed at their single recorded localities. In contrast, T. graeca faces significant pressure from illegal wildlife trade, a widespread issue across the Mediterranean region (Nijman and Bergin, 2017). Despite its legal protection in Algeria, its persistent presence in Batna’s traditional markets (pers. obs.) underscores the urgent need for stricter law enforcement. In addition, the provincial records of species newly documented in Batna presented in this study provide crucial distributional data that can contribute significantly to future IUCN assessments, offering more accurate estimates of Extent of Occurrence (EOO) and Area of Occupancy (AOO), two critical parameters in Red List evaluations.
Human persecution, particularly targeting snakes, and road mortality were among the most pervasive threats observed during our fieldwork. These pressures affect numerous species, including those currently classified as Least Concern, highlighting the need for a national Red List assessment for Algerian herpetofauna to better inform conservation priorities. Moreover, increasing public awareness on snake conservation – including educational campaigns promoting the distinction between venomous and non-venomous species – and implementing road mortality mitigation measures are crucial to addressing these threats (Colley et al., 2017; Roshnath and Divakar, 2019).
The Belezma BR is the only protected area within Batna, supporting 20 of the 47 documented herpetofaunal species and representing the most species-rich area in the province (Fig. 3A). Notably, it harbours 50% of species with unfavourable conservation statuses, all belonging to the Mediterranean chorotype. In contrast, the conservation of Saharan species should prioritize areas such as Djebel Metlili in Tilatou and the sand dunes of Bitam and M’doukal, and further west toward the Abdelkader Azil region. These unique ecological zones support species of particular conservation concern, including the Data Deficient Spalerosophis dolichospilus, the micro-endemic Tropiocolotes chirioi, and the only known populations of Heremites vittatus, Scincus scincus and Lytorhynchus diadema in the province.
The east/west distribution of chorotypes across Batna (Fig. 3B) highlights the role of the Aurès and Belezma ranges as biogeographic barriers, restricting herpetofaunal dispersal and contributing to regional faunal differentiation. During hyper-arid phases of the Quaternary, Mediterranean habitats in North Africa were largely confined to montane refugia, including these massifs, profoundly shaping the region’s biogeographic and taxonomic structure (Husemann et al., 2014). The humid montane environments of the Aurès and Belezma served as glacial refugia, sustaining relict populations of moisture-dependent species such as Chalcides mertensi, Coronella girondica, and Vipera monticola (Chirio and Blanc, 1997a; Bouam et al., 2019). Additionally, prolonged geographic isolation within these ranges likely fostered genetic divergence in reptile lineages, as evidenced by studies on the Podarcis hispanicus complex and Psammodromus blanci (Kaliontzopoulou et al., 2011; Mendes et al., 2017). In contrast, the arid western region of Batna may have acted as a climatic refugium for hyper-arid specialists such as Scincus scincus, consistent with evidence that peripheral Saharan zones have served as refugia fostering the persistence of isolated populations, as supported by paleodistribution models in wide-ranging taxa (Liz et al., 2024).
This study provides a critical piece of the puzzle toward the development of a national atlas of Algerian herpetofauna. However, achieving comprehensive coverage remains challenging due to limited national and international funding for biodiversity research (Waldron et al., 2013). The growing involvement of naturalist photographers in documenting Algeria’s biodiversity offers a promising avenue to address these gaps. We also advocate for public institution-led initiatives, such as coordinated mapping campaigns, to facilitate systematic surveys across other provinces, thereby enhancing knowledge not only of Algeria’s herpetofauna but also of its overall biodiversity.
We thank Ayachi Nacer, Chemseddine Zeroual, Farid Benzeroual, Hichem Nacer, Mohamed Ali Ramoul, Oussama Saadi, Rabeh Bentouati, Rachid Ait Medjber, Ramzi Bekkache, and local residents for their invaluable assistance during fieldwork. We are grateful to Aziz Hadj Aissa for providing photographs of a species not encountered during our surveys. Special thanks go to Souhil Zaaboubi, former Conservator of the Forest Conservation of Batna Province, for facilitating logistical support. We thank Philippe Geniez for assisting with specimen identification and Nicolas Vidal (MNHN) and Morris Flecks (ZFMK) for providing photographs of museum specimens. We are also grateful to the two anonymous reviewers for their constructive comments and suggestions, which helped improve the quality of the manuscript. Fieldwork involved photographing specimens in situ and, in rare occasions, briefly capturing individuals by hand for identification, followed by immediate release at the capture site. No permits were required to conduct these activities.
Supplementary material associated with this article can be found at: https://oaj.fupress.net/index.php/ah/article/view/17427/14573.
Abreu, J.M.B. (2017): Phylogenetic and diversity patterns of the Algerian whip snake Hemorrhois algirus. Unpublished master dissertation. University of Porto, Porto.
Aghasyan, A., Avci, A., Tuniyev, B., Lymberakis, P., Andrén, C., Cogălniceanu, D., Wilkinson, J., Ananjeva, N.B., Üzüm, N., Orlov, N.L., Podloucky, R., Tuniyev, S., Kaya, U., Vogrin, M., Baha El Din, S. (2021): Malpolon insignitus. The IUCN Red List of Threatened Species 2021: e.T157253A744783.
Ahmadzadeh, F., Flecks, M., Carretero, M.A., Böhme, W., Ihlow, F., Kapli, P., Miraldo, A., Rödder, D. (2016): Separate histories in both sides of the Mediterranean: phylogeny and niche evolution of ocellated lizards. J. Biogeogr. 43: 1242-1253.
Angel, F. (1923): Reptiles du Sahara, rapportés par la mission du colonel Hovart. Description d’un Ophidien nouveau du genre Rhamphiophis. Bull. Mus. Natl. Hist. Nat. 29: 205-208.
Baha El Din, S. (2006): A guide to the reptiles and amphibians of Egypt. Cairo, The American University in Cairo Press.
Baier, F., Schmitz, A., Sauer-Gürth, H., Wink, M. (2017): Pre-Quaternary divergence and subsequent radiation explain longitudinal patterns of genetic and morphological variation in the striped skink, Heremites vittatus. BMC Evol. Biol. 17: 132.
Barata, M., Harris, D.J., Castilho, R. (2008): Comparative phylogeography of northwest African Natrix maura (Serpentes: Colubridae) inferred from mtDNA sequences. Afr. Zool. 43: 1-7.
Beddek, M., Zenboudji-Beddek, S., Geniez, P., Fathalla, R., Sourouille, P., Arnal, V., Dellaoui, B., Koudache, F., Telailia, S., Peyre, O., Crochet, P-A. (2018): Comparative phylogeography of amphibians and reptiles in Algeria suggests common causes for the east-west phylogeographic breaks in the Maghreb. PLoS ONE 13: e0201218.
Benelkadi, H.A., Mammeri, A., Amroun, M. (2021): Biogeography, inventory and new data on reptiles of M’sila region, Algeria. Zool. Ecol. 31: 86-98.
Bernard, A., Ficheur, É. (1902): Les régions naturelles de l’Algérie: Second article. Ann. Géogr. 11: 339-365.
Bertolero, A., Busack, S.D. (2017): Mauremys leprosa (Schoepff in Schweigger 1812) – Mediterranean pond turtle, Spanish terrapin, Mediterranean stripe-necked terrapin. In: Conservation biology of freshwater turtles and tortoises: A compilation project of the IUCN/SSC Tortoise and Freshwater Turtle Specialist Group. Chelon. Res. Monogr. 5: 102, pp. 1-19. Rhodin, A.G.J., Iverson, J.B., van Dijk, P.P., Buhlmann, K.A., Pritchard, P.C.H., Mittermeier, R.A., Eds, Arlington, Chelonian Research Foundation and Turtle Conservancy.
Bezaz, Y.I., Hadjab, R., Khammar, H., Redjaimia, L., Saheb, M. (2021): First data on the diversity of the herpetofauna of the Oum El Bouaghi Region (Northeast of Algeria). Ecol. Environ. Conserv. 27: 983-989.
Bischoff, W., In Den Bosch, H.A.J. (1991): Zur Kenntnis von Psammodromus blanci (Lataste, 1880): Morphologie, Verbreitung, Ökologie und Paarungsbiologie. Salamandra 27: 163-180.
Bouam, I., Benmokhtar, E., Guechi, R. (2019): A fortuitous encounter with the vulnerable Vipera latastei: a new locality record from Algeria and distributional range extension. Herpetol. Notes 12: 809-812.
Bouam, I., Chedad, A., Sadine, S.E. (2024): Swimming in the Sahara: a new locality record and range extension of Natrix maura (Linnaeus, 1758) (Squamata, Colubridae) from central Algeria. Arx. Misc. Zool. 22: 129-142.
Bouam, I., Khelfaoui, F., Saoudi, M. (2022): Inferring probable distributional gaps and climate change impacts on the medically important viper Echis leucogaster in the western Sahara-Sahel: An ecological niche modeling approach. Biodiversitas 23: 5175-5183.
Bouam, I., Merzougui, S. (2021): A new provincial record and an updated distribution map for Pleurodeles nebulosus (Guichenot, 1850) in Algeria, with a new elevational record for the species (Amphibia: Salamandridae). Herpetol. Notes 14: 927-931.
Bouam, I., Necer, A., Saoudi, M. (2017): Highest altitudinal record of the ocellated lizard Timon pater (Lataste, 1880) (Squamata: Lacertidae). Herpetol. Notes 10: 101-102.
Bouam, I., Necer, A., Saoudi, M., Tahar-Chaouch, L., Khelfaoui, F. (2016): Diet and daily activity patterns of the lacertid lizard Psammodromus algirus (Sauria: Lacertidae) in a semi-arid Mediterranean region. Zool. Ecol. 26: 244-252.
Boulaouad, B.A., Bakhouche, B., Friel, B., Escoriza, D. (2023): African spurred tortoise Centrochelys sulcata: Range extension to Algeria. Herpetol. Bull. 164: 44-45.
Boulenger, G.A. (1887): Remarks on Dr. A. Strauch’s catalogue of the geckos in the Zoological Museum of the Imperial Academy of St. Petersburg. Ann. Mag. Nat. Hist. 19: 383-388.
Boulenger, G.A. (1891): Catalogue of the reptiles and batrachians of Barbary (Morocco, Algeria, Tunisia), based chiefly upon the notes and collections made in 1880–1884 by M. Fernand Lataste. Trans. Zool. Soc. London 13: 93-164.
Boulenger, G.A. (1921): Monograph of the Lacertidae. Volume II. London, Adlard and son and West Newman.
Brito, J.C., Godinho, R., Martínez-Freiría, F., Pleguezuelos, J.M., Rebelo, H., Santos, X., Vale, C.G., Velo-Antón, G., Boratyński, Z., Carvalho, S.B., Ferreira, S., Gonçalves, D.V., Silva, T.L., Tarroso, P., Campos, J.C., Leite, J.V., Nogueira, J., Álvares, F., Sillero, N., Sow, A.S., Fahd, S., Crochet, P.-A., Carranza, S. (2014): Unravelling biodiversity, evolution and threats to conservation in the Sahara‐Sahel. Biol. Rev. 89: 215-231.
Burriel-Carranza, B., Tarroso, P., Els, J., Gardner, A., Soorae, P., Mohammed, A.A., Tubati, S.R.K., Eltayeb, M.M. Shah, J.N., Tejero-Cicuéndez, H., Simó-Riudalbas, M., Pleguezuelos, J.M., Fernández-Guiberteau, D., Šmíd, J., Carranza, S. (2019): An integrative assessment of the diversity, phylogeny, distribution, and conservation of the terrestrial reptiles (Sauropsida, Squamata) of the United Arab Emirates. PLoS ONE 14: e0216273.
Busack, S.D., McCoy, C.J. (1990): Distribution, variation and biology of Macroprotodon cucullatus (Reptilia, Colubridae, Boiginae). Ann. Carnegie Mus. 59: 261-285.
Caeiro-Dias, G., Luís, C., Pinho, C., Crochet, P-A., Sillero, N., Kaliontzopoulou, A. (2018): Lack of congruence of genetic and niche divergence in Podarcis hispanicus complex. J. Zool. Syst. Evol. Res. 56: 479-492.
Carranza, S., Arnold, E.N., Pleguezuelos, J.M. (2006): Phylogeny, biogeography, and evolution of two Mediterranean snakes, Malpolon monspessulanus and Hemorrhois hippocrepis (Squamata, Colubridae), using mtDNA sequences. Mol. Phylogenet. Evol. 40: 532-546.
Carranza, S., Arnold, E.N., Wade, E., Fahd, S. (2004): Phylogeography of the false smooth snakes, Macroprotodon (Serpentes, Colubridae): mitochondrial DNA sequences show European populations arrived recently from Northwest Africa. Mol. Phylogenet. Evol. 33: 523-532.
Chedad, A., Bouam, I., El Bouhissi, M., Dahmani, W., Ait Hammou, M., Mebarki, M.T., Mezzi, M., Sadine, S.E. (2024): Moorish Viper, Daboia mauritanica (Gray, 1849) (Squamata, Viperidae), in Algeria: new provincial records, range extension, and an update on its distribution. Check List 20: 536-543.
Chirio, L., Blanc, C.P. (1993): Existence in parapatry of two species of Ophisops in Algeria (Aures): zoogeographical implications. Amphib.-Reptil. 14: 341-347.
Chirio, L., Blanc, C.P. (1997a): Analyse biogéographique du peuplement reptilien de l’Aurès (Algérie). Biogeographica 73: 13-22.
Chirio, L., Blanc, C.P. (1997b): Statut et distribution des reptiles dans le massif de l’Aurès (Algérie). J. Afr. Zool. 111: 205-233.
Chowdhury, S., Ahmed, S., Alam, S., Callaghan, C.T., Das, P., Di Marco, M., Di Minin, E., Jarić, I., Labi, M.M., Rokonuzzaman, M., Roll, U., Sbragaglia, V., Siddika, A., Bonn, A. (2024): A protocol for harvesting biodiversity data from Facebook. Conserv. Biol. 38: e14257.
Cogălniceanu, D., Castilla, A.M., Valdeón, A., Gosá, A., Al-Jaidah, N., Alkuwary, A., Saifelnasr, E.O.H., Mas-Peinado, P., Richer, R., Mohd Al-Hemaidi, A.A. (2014): A preliminary report on the distribution of lizards in Qatar. Zookeys 373: 67-91.
Colley, M., Lougheed, S.C., Otterbein, K., Litzgus, J.D. (2017): Mitigation reduces road mortality of a threatened rattlesnake. Wildlife Res. 44: 48-59.
Cox, N., Chanson, J., Stuart, S. (2006): The status and distribution of reptiles and amphibians of the Mediterranean Basin. Gland, Switzerland and Cambridge, UK, IUCN.
Daget, P. (1977): Le bioclimat Méditerranéen: Analyse des formes climatiques par le système d’Emberger. Vegetatio 34: 87-103.
Denzer, W. (2021): The correct name for the North African rock lizard is Agama bibronii A. Duméril in Duméril & Duméril, 1851, not Agama impalearis Boettger, 1874 (Reptilia, Squamata). Bionomina 25: 76-80.
Dufresnes, C., Mazepa, G., Jablonski, D., Oliveira, R.C., Wenseleers, T., Shabanov, D.A., Auer, M., Ernst, R. Koch, C., Ramírez-Chaves, H.E., Mulder, KP., Simonov, E., Tiutenko, a., Kryvokhyzha, D., Wennekes, P.L., Zinenko, O.T., Korshunov, O.V., Al-Johany, A.M., Peregontsev, E.A., Masroor, R., Betto-Colliard, C., Denoël, M., Borkin, L.J., Skorinov, D.V., Pasynkova, R.A., Mazanaeva, L.F., Rosanov, J.M., Dubey, S., Litvinchuk, S. (2019): Fifteen shades of green: The evolution of Bufotes toads revisited. Mol. Phylogenet. Evol. 141: 106615.
Dufresnes, C., Monod-Broca, B., Bellati, A., Canestrelli, D., Ambu, J., Wielstra, B., Dubey, S., Crochet, P-A., Denoël, M., Jablonski, D. (2024): Piecing the barcoding puzzle of Palearctic water frogs (Pelophylax) sheds light on amphibian biogeography and global invasions. Glob. Change Biol. 30: e17180.
El Maalouf, A. (1985): معجم الحيوان [The Animal Lexicon]. Beirut, Dar Erraid El Arabi.
Escoriza, D., Ben Hassine, J. (2019): Amphibians of North Africa. Academic Press.
Escoriza, D., Díaz-Paniagua, C., Andreu, A., Ben Hassine, J. (2022): Testudo graeca Linnaeus 1758 (Western Subspecies Clade: Testudo g. graeca, T. g. cyrenaica, T. g. marokkensis, T. g. nabeulensis, T. g. whitei) – Mediterranean spur-thighed tortoise, Moorish tortoise, Libyan tortoise, Moroccan tortoise, Tunisian tortoise, Souss Valley tortoise. In: Conservation biology of freshwater turtles and tortoises: A compilation project of the IUCN/SSC Tortoise and Freshwater Turtle Specialist Group. Chelon. Res. Monogr. 5: 117, pp. 1-18. Rhodin, A.G.J., Iverson, J.B., van Dijk, P.P., Stanford, C.B., Goode, E.V., Buhlmann, K.A., Mittermeier, R.A. Eds, Arlington, Chelonian Research Foundation and Turtle Conservancy.
European Space Agency (2024): Copernicus global digital elevation model. Distributed by OpenTopography.
Faria, J.F., Harris, D.J. (2020): Phylogeography of Psammodromus algirus (Linnaeus, 1758) in Morocco. Herpetol. Notes 13: 1055-1061.
Fonseca, M.M., Brito, J.C., Paulo, O.S., Carretero, M.A., Harris, D.J. (2009): Systematic and phylogeographical assessment of the Acanthodactylus erythrurus group (Reptilia: Lacertidae) based on phylogenetic analyses of mitochondrial and nuclear DNA. Mol. Phylogenet. Evol. 51: 131-142.
Fonseca, M.M., Brito, J.C., Rebelo, H., Kalboussi, M., Larbes, S., Carretero, M.A., Harris, D.J. (2008): Genetic variation among spiny-footed lizards in the Acanthodactylus pardalis group from North Africa. Afr. Zool. 43: 8-15.
Frost, D.R. (2024): Amphibian species of the world: an online reference. Version 6.2. Electronic Database accessible at https://amphibiansoftheworld.amnh.org/index.php. New York, USA, American Museum of Natural History.
Gauthier, H. (1932): Remarques sur la faune aquatique de la «Fontaine-Chaude» au nord de Batna, en Algérie. Bull. Soc. Hist. Natur. Afr. Nord 23: 105-108.
GBIF.org (06 January 2025): GBIF occurrence download. https://doi.org/10.15468/dl.b2t77j
Geniez, P. (2015): Serpents d’Europe, d’Afrique du Nord et du Moyen–Orient. Paris, Delachaux et Niestlé.
Ghaleb, E. (1986): Encyclopedia of natural sciences. Beirut, Dar El-Machreq.
Gherbi, N., Tiar-Saadi, M., Boucheker, A., Široký, P., Mezghiche, C., Draidi, K., Bouslama, Z., Tiar, G. (2023): Distribution and conservation status of European pond turtles Emys orbicularis (L., 1758) in Algeria. Diversity 15: 993.
Gonçalves, D.V., Martínez-Freiría, F., Crochet, P-A., Geniez, P., Carranza, S., Brito, J.C. (2018): The role of climatic cycles and trans-Saharan migration corridors in species diversification: Biogeography of Psammophis schokari group in North Africa. Mol. Phylogenet. Evol. 118: 64-74.
Harris, D.J., Perera, A. (2009): Phylogeography and genetic relationships of North African Bufo mauritanicus Schlegel, 1841 estimated from mitochondrial DNA sequences. Biologia 64: 356-360.
Harris, D.J., Sergiadou, D., Faria, J.F. (2024): New data on the diversity and distribution of lineages of the Acanthodactylus erythrurus species complex in North Africa derived from mitochondrial DNA markers. Herpetozoa 37: 281-286.
Hortal, J., de Bello, F., Diniz-Filho, J.A.F., Lewinsohn, T.M., Lobo, J.M., Ladle, R.J. (2015): Seven shortfalls that beset large-scale knowledge of biodiversity. Annu. Rev. Ecol. Evol. S. 46: 523-549.
Husemann, M., Schmitt, T., Zachos, F.E., Ulrich, W., Habel, J.C. (2014): Palaearctic biogeography revisited: evidence for the existence of a North African refugium for Western Palaearctic biota. J. Biogeogr. 41: 81-94.
Ineich, I., Martelli, J.L., Clary, J. (2003): Catalogue des collections de reptiles du Muséum d’Histoire naturelle de Lyon. Cinquième note: amphisbéniens et sauriens (première partie). Cah. Sci. 2003: 53-86.
Ineich, I., Martelli, J.L., Clary, J. (2005): Catalogue des collections de reptiles du Muséum, Lyon. Sixième note: sauriens (deuxième partie) et rhynchocéphales. Cah. Sci. 8: 33-66.
IUCN (2024): The IUCN Red List of Threatened Species. Version 2024-2. https://www.iucnredlist.org
Joger, U., Bischoff, W. (1989): Erste Ergebnisse einer herpetologischen Forschungsreise nach Nordwest-Afrika. Tier und Museum 1: 99-106.
Joger, U., Slimani, T., El Mouden, H., Martínez-Solano, I., Geniez, P. (2009): Psammodromus blanci. The IUCN Red List of Threatened Species 2009: e.T61559A12491750.
Kaliontzopoulou, A., Carretero, M.A., Llorente, G.A. (2012): Morphology of the Podarcis wall lizards (Squamata: Lacertidae) from the Iberian Peninsula and North Africa: patterns of variation in a putative cryptic species complex. Zool. J. Linn. Soc. 164: 173-193.
Kaliontzopoulou, A., Pinho, C., Harris, D.J., Carretero, M.A. (2011): When cryptic diversity blurs the picture: a cautionary tale from Iberian and North African Podarcis wall lizards. Biol. J. Linn. Soc. 103: 779-800.
Kapli, P., Lymberakis, P., Crochet, P-A., Geniez, P., Brito, J.C., Almutairi, M., Ahmadzadeh, F., Schmitz, A., Wilms, T., Pouyani, N.R., Poulakakis, N. (2015): Historical biogeography of the lacertid lizard Mesalina in North Africa and the Middle East. J. Biogeogr. 42: 267-279.
Kapli, P., Lymberakis, P., Poulakakis, N., Mantziou, G., Parmakelis, A., Mylonas, M. (2008): Molecular phylogeny of three Mesalina (Reptilia: Lacertidae) species (M. guttulata, M. brevirostris and M. bahaeldini) from North Africa and the Middle East: Another case of paraphyly? Mol. Phylogenet. Evol. 49: 102-110.
Khelfaoui, F., Bouam, I., Saoudi, M., Saadi, O., Tahar-Chaouch, L. (2023): Non-synergistic sexual and geographical morphological variation in the lacertid lizard Psammodromus algirus (Linnaeus, 1758) from North Africa. Herpetol. Notes 16: 889-895.
Lataste, F. (1880a): Diagnoses de reptiles nouveaux d’Algérie. Le Naturaliste 2: 299.
Lataste, F. (1880b): Diagnoses de reptiles nouveaux d’Algérie. Le Naturaliste 2: 306-307.
Lataste, F. (1881): Diagnoses de reptiles nouveaux d’Algérie. Le Naturaliste 3: 357-359.
Lataste, F. (1885): Les acanthodactyles de Barbarie et les autres espèces du genre. Description d´une nouvelle espèce, du pays des çomalis (Acanthodactylus Vaillanti). Ann. Mus. Civ. Stor. Nat. Genova 2: 476-516.
Lima, A., Pinho, C., Larbes, S., Carretero, M.A., Brito, J.C., Harris, D.J. (2009): Relationships of Podarcis wall lizards from Algeria based on mtDNA data. Amphibia-Reptilia 30: 483-492.
Liz, A.V., Licata, F., Santos, B., Gonçalves, D.V., Lakušić, M., Roumelioti, M., Serén, N., Tarroso, P., Abdulkareem, A., László, P., Brito, J.C. (2025): Biogeographic implications of biodiversity shortfalls in a mid-altitude desert ecotone of the Arabian Peninsula. Divers. Distrib. 31: e70016.
Liz, A.V., Mochales-Riaño, G., Velo-Antón, G., García-Cardenete, L., Brito, J.C., Carranza, S., Martínez-Freiría, F. (2024): Historical biogeography of the Saharan horned viper enlightens past dynamics of hyperarid desert habitats. J. Arid Environ. 222: 105171.
Liz, A.V., Rödder, D., Gonçalves, D.V., Velo-Antón, G., Fonseca, M.M., Geniez, P., Crochet, P.-A., Brito, J.C. (2021): The role of Sahara highlands in the diversification and desert colonization of the Bosc’s fringe-toed lizard. J. Biogeogr. 48: 2891-2906.
Loveridge, A. (1947): Revision of the African lizards of the family Gekkonidae. Bull. Mus. Comp. Zool. 98: 1-469.
Loveridge, A., Williams, E.E. (1957): Revision of the African tortoises and turtles of the suborder Cryptodira. Bull. Mus. Comp. Zool. 115: 163-557.
Martínez-Freiría, F., Crochet, P-A., Fahd, S., Geniez, P., Brito, J.C., Velo-Antón, G. (2017): Integrative phylogeographical and ecological analysis reveals multiple Pleistocene refugia for Mediterranean Daboia vipers in north-west Africa. Biol. J. Linn. Soc. 122: 366-384.
Martínez-Freiría, F., Fahd, S., Crochet, P-A., Beddek, M. (2024): Vipera monticola. The IUCN Red List of Threatened Species 2024: e.T221202628A3105206.
Martínez-Freiría, F., Freitas, I., Velo-Antón, G., Lucchini, N., Fahd, S., Larbes, S., Pleguezuelos, J.M., Santos, X., Brito, J.C. (2021): Integrative taxonomy reveals two species and intraspecific differentiation in the Vipera latastei–monticola complex. J. Zool. Syst. Evol. Res. 59: 2278-2306.
McDiarmid, R.W., Foster, M.S., Guyer, C., Gibbons, J.W., Chernoff, N. (2012): Reptile biodiversity: Standard methods for inventory and monitoring. Los Angeles, University of California Press.
Mebarki, A. (2007): The hydrological basins of eastern Algeria: water resources, planning and environment. Houille Blanche 93: 112-115.
Mendes, J., Harris, D.J., Carranza, S., Salvi, D. (2017): Biogeographical crossroad across the Pillars of Hercules: Evolutionary history of Psammodromus lizards in space and time. J. Biogeogr. 44: 2877-2890.
Metallinou, M., Arnold, E.N., Crochet, P-A., Geniez, P., Brito, J.C., Lymberakis, P., Baha El Din, S., Sindaco, R., Robinson, M., Carranza, S. (2012): Conquering the Sahara and Arabian deserts: systematics and biogeography of Stenodactylus geckos (Reptilia: Gekkonidae). BMC Evol. Biol. 12: 258.
Metallinou, M., Červenka, J., Crochet, P-A., Kratochvíl, L., Wilms, T., Geniez, P., Shobrak, M.Y., Brito, J.C., Carranza, S. (2015): Species on the rocks: Systematics and biogeography of the rock-dwelling Ptyodactylus geckos (Squamata: Phyllodactylidae) in North Africa and Arabia. Mol. Phylogenet. Evol. 85: 208-220.
Miralles, A., Geniez, P., Beddek, M., Aranda, D.M., Brito, J.C., Leblois, R., Crochet, P-A. (2020): Morphology and multilocus phylogeny of the Spiny-footed Lizard (Acanthodactylus erythrurus) complex reveal two new mountain species from the Moroccan Atlas. Zootaxa 4747: 302-326.
Mouane, A., Bourougaa, D., Hamdi, M., Boudjerada, K., Harrouchi, A., Ghennoum, I., Sekour, M., Chenchouni, H. (2021): The Rough Bent-toed Gecko Cyrtopodion scabrum (Heyden, 1827) (Squamata: Gekkonidae): First records in Algeria and NW Africa with morphometric and meristic description of population. Afr. J. Ecol. 59: 312-319.
Mouane, A., Harrouchi, A., Ghennoum, I., Sekour, M., Chenchouni, H. (2024): Amphibian and reptile diversity in natural landscapes and human-modified habitats of the Sahara Desert of Algeria: A better understanding of biodiversity to improve conservation. Elem. Sci. Anth. 12: 00106.
Nijman, V., Bergin, D. (2017): Trade in spur-thighed tortoises Testudo graeca in Morocco: volumes, value and variation between markets. Amphib.-Reptil. 38: 275-287.
Nouira, S., Blanc, C.P., Crochet, P-A., Frétey, T., Geniez, P., Ineich, I., De Massary, J.C., Ohler, A., Tlili, W., Lescure, J. (2022): Nouvelle liste taxinomique de l’herpétofaune de Tunisie. Bull. Soc. Herpétol. Fr. 180: 5-26.
Nouira, M.S., Joger, U. (2006): Acanthodactylus blanci. The IUCN Red List of Threatened Species 2006: e.T61455A12488766.
Olivier, E. (1894): Herpétologie algérienne ou catalogue raisonné des reptiles et des batraciens observés jusqu’à ce jour en Algérie. Mém. Soc. Zool. Fr. 7: 98-131.
Olson, D.M., Dinerstein, E., Wikramanayake, E.D., Burgess, N.D., Powell, G.V.N., Underwood, E.C., D’amico, J.A., Itoua, I., Strand, H.E., Morrison, J.C., Loucks, C.J., Allnutt, T.F., Ricketts, T.H., Kura Y., Lamoreux, J.F., Wettengel, W.W., Hedao, P., Kassem, K.R. (2001): Terrestrial ecoregions of the world: A New map of life on Earth: A new global map of terrestrial ecoregions provides an innovative tool for conserving biodiversity. BioScience 51: 933-938.
Perera, A., Harris, D.J. (2010): Genetic variability within the Oudri’s fan-footed gecko Ptyodactylus oudrii in North Africa assessed using mitochondrial and nuclear DNA sequences. Mol. Phylogenet. Evol. 54: 634-639.
Pizzigalli, C., Crochet, P-A., Geniez, P., Martínez-Freiría, F., Velo-Antón, G., Brito, J.C. (2021): Phylogeographic diversification of the Mesalina olivieri species complex (Squamata: Lacertidae) with the description of a new species and a new subspecies endemic from North West Africa. J. Zool. Syst. Evol. Res. 59: 2321-2349.
Rato, C., Brito, J.C., Carretero, M.A., Larbes, S., Shacham, B., Harris, D.J. (2007): Phylogeography and genetic diversity of Psammophis schokari (Serpentes) in North Africa based on mitochondrial DNA sequences. Afr. Zool. 42: 112-117.
Rato, C., Carranza, S., Harris, D.J. (2012): Evolutionary history of the genus Tarentola (Gekkota: Phyllodactylidae) from the Mediterranean Basin, estimated using multilocus sequence data. BMC Evol. Biol. 12: 14.
Ribeiro-Júnior, M.A., Koch, C., Flecks, M., Calvo, M., Meiri, S. (2022): Dwarves in a big world: Two new species of Tropiocolotes (Squamata: Gekkonidae) from the Sahara Desert, with the first detailed skull description of the genus. J. Herpetol. 56: 396-421.
Robertson, M.P., Cumming, G.S., Erasmus, B.F.N. (2010): Getting the most out of atlas data. Divers. Distrib. 16: 363-375.
Roshnath, R., Divakar, N. (2019): Solving species quandary: Why awareness programs are pivotal in snake conservation. Herpetol. J. 29: 214-218.
Rouag, R., Benyacoub, S. (2006): Inventaire et écologie des reptiles du Parc national d’El Kala (Algérie). Bull. Soc. Herpétol. Fr. 117: 25-40.
Rouag, R., Dahel, R., Rahmouni, S., Benkacimi, S., Ziane, N. (2016): First records of the rainbow mabuya Trachylepis quinquetaeniata (Lichtenstein, 1823) (Squamata: Scincidae) in Algeria. Herpetol. Notes 9: 167-169.
Rouag, R., Ziane, N., De Sousa, M. (2024): A tentative list of reptilian fauna of Algeria and their conservation status. Biodivers. Data J. 12: e120471.
Sabaj, M.H. (2020): Codes for natural history collections in ichthyology and herpetology. Copeia 108: 593-669.
Salvador, A. (1982): A revision of the lizards of the genus Acanthodactylus (Sauria: Lacertidae). Bonn. Zool. Monogr. 16: 1-167.
Saoudi, M., Necer, A., Bensaci, M., Bouam, I. (2017): Distribution extension of Agama impalearis (Boettger, 1874) (Reptilia: Agamidae), with a new record from Batna province, north-eastern Algeria. Herpetol. Notes 10: 261-262.
Salvi, D., Perera, A., Sampaio, F.L., Carranza, S., Harris, D.J. (2018): Underground cryptic speciation within the Maghreb: Multilocus phylogeography sheds light on the diversification of the checkerboard worm lizard Trogonophis wiegmanni. Mol. Phylogenet. Evol. 120: 118-128.
Schleich, H.H., Kästle, W., Kabisch, K. (1996): Amphibians and reptiles of North Africa. Königstein, Koeltz Scientific Books.
Sillero, N., Celaya, L., Martín-Alfageme, S. (2005): Using Geographical Information Systems (GIS) to make an atlas: a proposal to collect, store, map and analyse chorological data for herpetofauna. Rev. Esp. Herp. 19: 87-101.
Sindaco, R., Jeremčenko, V.K. (2008): The reptiles of the Western Palearctic. 1. Annotated checklist and distributional atlas of the turtles, crocodiles, amphisbaenians and lizards of Europe, North Africa, Middle East and Central Asia. Latina, Edizioni Belvedere.
Sindaco, R., Simó-Riudalbas, M., Sacchi, R., Carranza, S. (2018): Systematics of the Mesalina guttulata species complex (Squamata: Lacertidae) from Arabia with the description of two new species. Zootaxa 4429: 513-547.
Strauch, A. (1862): Essai d’une Erpétologie de l’Algérie. Mem. Acad. Sci. St. Petersb. 4: 1-85.
Strauch, A. (1882): Bemerkungen über die Eidechsenfamilie der Amphisbaeniden. Bull. Scient. Acad. Imp. Sci. St. Petersb. 28: 45-132.
Strauch, A. (1887): Bemerkungen über die Geckoniden-Sammlung im Zoologischen Museum der Kaiserlichen Akademie der Wissenschaften zu St. Petersburg. Mem. Acad. Sci. St. Petersb. 35: 1-72.
Tamar, K., Carranza, S., Sindaco, R., Moravec, J., Trape, J-F., Meiri, S. (2016): Out of Africa: Phylogeny and biogeography of the widespread genus Acanthodactylus (Reptilia: Lacertidae). Mol. Phylogenet. Evol. 103: 6-18.
Tamar, K., Metallinou, M., Wilms, T., Schmitz, A., Crochet, P-A., Geniez, P., Carranza, S. (2018): Evolutionary history of spiny-tailed lizards (Agamidae: Uromastyx) from the Saharo-Arabian region. Zool. Scr. 47: 159-173.
Title, P.O., Bemmels, J.B. (2018): ENVIREM: an expanded set of bioclimatic and topographic variables increases flexibility and improves performance of ecological niche modelling. Ecography 41: 291-307.
Titley, M.A., Snaddon, J.L., Turner, E.C. (2017): Scientific research on animal biodiversity is systematically biased towards vertebrates and temperate regions. PLoS ONE 12: e0189577.
Tolley, K.A., Alexander, G.J., Branch, W.R., Bowles, P., Maritz, B. (2016): Conservation status and threats for African reptiles. Biol. Conserv. 204: 63-71.
Trape, J-F. (2023): Guide des serpents d’Afrique occidentale, centrale et d’Afrique du Nord. Marseille, IRD Éditions.
Uetz, P., Freed, P., Aguilar, R., Reyes, F., Kudera, J., Hošek, J. (2024): The Reptile Database. http://www.reptile-database.org
UNESCO (2015): Twenty new sites added to UNESCO’s World Network of Biosphere Reserves. https://www.unesco.org/en/articles/twenty-new-sites-added-unescos-world-network-biosphere-reserves
Velo-Antón, G., Henrique, M., Liz, A.M., Martínez-Freiría, F., Pleguezuelos, J.M., Geniez, P., Crochet, P.-A., Brito, J.C. (2022): DNA barcode reference library for the West Sahara-Sahel reptiles. Sci. Data 9: 459.
Verdú-Ricoy, J., Carranza, S., Salvador, A., Busack, S., Díaz, J. (2010): Phylogeography of Psammodromus algirus (Lacertidae) revisited: systematic implications. Amphibia-Reptilia 31: 576-582.
Wade, E. (1988): Intraspecific variation in the colubrid snake genus Macroprotodon. Herpetol. J. 1: 237-245.
Wade, E. (2001): Review of the False Smooth snake genus Macroprotodon (Serpentes, Colubridae) in Algeria with a description of a new species. Bull. Br. Mus. Nat. Hist. Zool. 67: 85-107.
Wagner, P., Melville, J., Wilms, T.M., Schmitz, A. (2011): Opening a box of cryptic taxa - the first review of the North African desert lizards in the Trapelus mutabilis Merrem, 1820 complex (Squamata: Agamidae) with descriptions of new taxa. Zool. J. Linn. Soc. 163: 884-912.
Wagner, P., Wilms, T. (2013): Trapelus boehmei. The IUCN Red List of Threatened Species 2013: e.T203803A2771535.
Wagner, P., Wilms, T., Niagate, B. (2021): Trapelus mutabilis. The IUCN Red List of Threatened Species 2021: e.T198525A2529839.
Waldron, A., Mooers, A.O., Miller, D.C., Nibbelink, N., Redding, D., Kuhn, T.S., Roberts, J.T., Gittleman, J.L. (2013): Targeting global conservation funding to limit immediate biodiversity declines. Proc. Natl. Acad. Sci. USA 110: 12144-12148.
Weiss, D.J., Nelson, A., Gibson, H.S., Temperley, W., Peedell, S., Lieber, A., Hancher. M., Poyart, E., Belchior, S., Fullman, N., Mappin, B., Dalrymple, U., Rozier, J., Lucas, T.C.D., Howes, R.E., Tusting, L.S., Kang, S.Y., Cameron, E., Bisanzio, D., Battle, K.E., Bhatt, S., Gething, P.W. (2018): A global map of travel time to cities to assess inequalities in accessibility in 2015. Nature 553: 333-336.
Werner, F. (1892): Ausbeute einer herpetologischen Excursion nach Ost-Algerien. Verh. Zool. Bot. Ges. Wien 42: 350-355.
Werner, F. (1893): Herpetologische Nova (2. Teil). Zool. Anz. 16: 359-363.
Werner, F. (1894): Zweiter Beitrag zur Herpetologie von Ost-Algerien. Verh. Zool. Bot. Ges. Wien 44: 75-87.
Yadollahvandmiandoab, R., Koroiva, R., Bashirichelkasari, N., Mesquita, D.O. (2023): Phylogenetic relationships and divergence times of the poorly known genus Spalerosophis (Serpentes: Colubridae). Org. Divers. Evol. 23: 415-423.