Submitted on: 2025, 2nd April; revised on: 2025, 14th June; accepted on: 2025, 29th June
First report of overwintering tadpoles in the endemic Italian agile frog Rana latastei
1 Department of Environmental Science and Policy, Università degli Studi di Milano, Via Celoria 10, 20133 Milan, Italy
2 Via Magni 41, 22044, Inverigo, Italy
*Corresponding author. Email: andrea.melotto@unimi.it
Abstract. Climate change is affecting organism seasonality patterns, and this can drive phenological shifts in key traits, including breeding activity. Here, we report the first record of overwintering tadpoles in the Italian agile frog (Rana latastei), a threatened endemic species of Northern Italy. This species usually breeds between late January and April, with tadpoles reaching metamorphosis by early summer. In February 2025, alongside the first freshly-laid egg-clutches of the usual breeding season, we observed several large-sized tadpoles at a reproductive site of R. latastei within Como province. A few days after hatching, six newly hatched tadpoles and six large-sized individuals at a later developmental stage were captured, photographed, and measured. Concurrently, a recently metamorphosed froglet was found at the same site. Morphological analyses revealed that individuals showed typical traits of R. latastei. Moreover, while newly hatched tadpoles were at Gosner’s development stages 25-26, large-sized individuals were visibly bigger and at a late developmental stage, which was incompatible with the classical late-winter breeding timing of this species. Our study provides the first evidence of overwintering tadpoles in R. latastei, suggesting that potential shifts in development timing (prolonged larval time) or altered breeding habits (autumnal reproduction) may be occurring. Although the mechanisms behind this unusual observation remain unclear, such a phenological shift may have been favoured by variation in climatic regime.
Keywords. Amphibians, anurans, breeding, climate change, global warming, froglet, phenology, reproduction.
Global warming is causing unprecedented quick phenological shifts in animal populations (Horton et al., 2020). These shifts can involve breeding activities, including both advances (Parmesan, 2007; Ficetola and Maiorano, 2016) and delays (Dalpasso et al., 2023) in the onset of breeding season after the winter. Additionally, due to the general increase in winter temperatures, the end of the activity season in autumn is expected to be postponed (Lang et al., 2025). This may influence voltinism, potentially favouring an increase in reproductive events within a year in some populations. However, this may also produce survival risks for early stages, when suitable conditions for development are not matched (Benard, 2015; Bison et al., 2021). For instance, in temperate regions several amphibian species have been shown to extend or shift their breeding season by days or weeks in the last decades (Todd et al., 2011), with some cases of amphibian and reptile species reproducing outside their typical breeding season reported (Graña and Martínez-Freiría, 2020; Rodriguez-Muñoz et al., 2020).
In Europe, most anurans breed in late winter-spring, with juveniles metamorphosing in late spring-summer. This timing allows them taking advantage of spring rain that fills temporary wetlands and exploiting the warm season for larval growth and development. However, breeding patterns can be different in some species in Southern Europe. For instance, in Sicily, the green toad (Bufo boulengeri Laurenti, 1768) can also breed in autumn and winter, with tadpoles from autumnal mating overwintering and metamorphosing in early spring (Sicilia et al., 2006). In this case, autumnal breeding has been interpreted as an adaptation to arid environments, where waterbodies filled by autumn rain are more likely to retain water long enough to enable toadlets attaining metamorphosis (Sicilia et al., 2006). Less evidence for autumnal breeding and tadpole overwintering is available for amphibians living in Northern Italy. Among them, the Italian agile frog, Rana latastei Boulenger, 1879, is endemic to the lowlands of Northern Italy and adjacent areas and breeds from late January to mid-April (Ambrogio and Mezzadri, 2018; Ficetola et al., 2025). In principle, changes in temperature and/or precipitation patterns related to climate change might affect breeding phenology of this species, but information on autumn breeding and/or overwintering tadpoles is so far lacking.
Here, we report the first observation of overwintering tadpoles in the Italian agile frog. The reproductive site is a small artificial permanent pond (roughly 2 × 1 m; 0.3 m depth) located in a small wooded area in the foothills of the Como district (Inverigo, Lombardy, Italy). The site is located approximately 350 m a.s.l. and the area hosts numerous water bodies supporting a diverse amphibian community, including three urodele and six anuran species. This area is part of a long-term amphibian monitoring program covering the past two decades (Ficetola et al., 2009; Falaschi et al., 2021), which revealed the presence of a stable meta-population for R. latastei across interconnected breeding sites (Manenti et al., 2020). The permanent pond where overwintering tadpoles were found has been consistently surveyed multiple times during spring since 2010 and is a stable breeding site of R. latastei, where no other amphibian, except the fire salamander (Salamandra salamandra Linnaeus, 1758), has been reported reproducing. On 27 January 2025, ~5 days after the first mid-winter rainfalls that usually trigger amphibian breeding in the area, the site was monitored along with other ones in the surroundings. This survey revealed the presence of a single freshly laid egg-clutch of R. latastei (the first clutch of the breeding season found in the area). The site was then monitored on January 30 (one new clutch), February 6 (no new clutches), February 11 (one new clutch), and on February 26 (four new clutches). The first egg hatch was observed on February 11. On February 6, the presence of three large-sized tadpoles showing R. latastei traits was observed. On February 26, we collected six large-sized tadpoles and six small-sized tadpoles (Gosner’s stage >25) for measuring (Fig. 1). Concurrently, a newly metamorphosed froglet was found (Fig. 2). Individuals were captured by gently netting the pool, then briefly kept in a small plastic tank, and photographed on graph paper using a 100 mm macro lens to minimize distortion. Pictures were taken by a single operator using a standardized photographic setting. After the photoshoot, the individuals were immediately released (permits listed in acknowledgements). Froglet and tadpoles were measured from scaled pictures using ImageJ software (Schneider et al., 2012) by a single operator following standard procedures (Relyea, 2001; Melotto et al., 2020, 2021). Tadpole developmental stages were determined based on Gosner’s tables (Gosner, 1960). Although never observed breeding in the study site, another Rana Linnaeus, 1758 species with similar ecology, the agile frog (Rana dalmatina Fitzinger in Bonaparte, 1838), is present in the nearby woods, and tadpoles of this frog sometimes show phenotypes difficult to tease apart from R. latastei (Barbieri et al., 2000). Hereafter, along with morphometric and stage comparison between tadpole cohorts, key phenotypic traits differing between the two frog species are highlighted (Lanza et al., 2009; Ambrogio and Mezzadri, 2014).


We obtained tadpole’s measurements from both dorsal and side pictures (Table 1A, Fig. 1A-D) and included total length (Totl), body length (BL), body height (BH), body width (BW), tail length (TL), tail height (TH), tail muscle height (TMH), and tail muscle width (TMW). Moreover, we used dorsal pictures to calculate minimum eye distance (ED) and minimum nostril distance (ND). ND was available for large-sized tadpoles only, as nostril position was not clearly identifiable in newly hatched tadpoles. All tadpole measurements, except for ND, were included in a principal component analysis to ascertain the existence of distinct size classes among tadpoles from different cohorts and if they matched the eye-based and Gosner’s stage identification. Moreover, we calculated eye-distance nostril-distance ratio (ENDr) from ED and ND measurements for the perspective overwintering tadpoles (large-sized individuals). This ratio is one of the key traits that differ between tadpoles of R. dalmatina and R. latastei, being around 2 for the first one and rarely >1.5 for R. latastei (Lanza et al., 2009). Finally, we used ventral pictures of the froglet to take some morphological measurements (Table 1B): total length (TL), body width (BW), jaw width (JW), and few left hindlimb lengths, including proximal hindlimb (LPHL), distal hindlimb (LDHL), tarsus (LTL), and foot (LFL). Additionally, froglet ED and ND were obtained from dorsal picture (Table 1B).
| A) | ID | size class | Gosner | BH | TH | TMH | BL | TL | TotL | BW | TMW | ED | ND |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| GIR1 | large | 37-39 | 0.884 | 1.042 | 0.510 | 1.858 | 3.149 | 4.525 | 1.089 | 0.349 | 0.341 | 0.244 | |
| GIR2 | large | 40 | 0.912 | 0.810 | 0.405 | 1.563 | 2.320 | 3.489 | 1.049 | 0.301 | 0.267 | 0.232 | |
| GIR3 | large | 40 | 0.811 | 0.858 | 0.403 | 1.539 | 2.450 | 3.626 | 1.027 | 0.289 | 0.263 | 0.220 | |
| GIR4 | large | 40 | 0.847 | 0.826 | 0.375 | 1.550 | 2.347 | 3.467 | 0.917 | 0.363 | 0.274 | 0.192 | |
| GIR5 | large | 40 | 0.857 | 0.983 | 0.423 | 1.563 | 2.461 | 3.698 | 1.076 | 0.312 | 0.323 | 0.247 | |
| GIR6 | large | 37-39 | 0.992 | 1.069 | 0.606 | 1.850 | 3.483 | 4.841 | 1.136 | 0.379 | 0.332 | 0.259 | |
| GIR7 | small | 25-26 | 0.374 | 0.423 | 0.178 | 0.633 | 1.285 | 1.772 | 0.395 | 0.128 | 0.119 | - | |
| GIR8 | small | 25-26 | 0.370 | 0.403 | 0.172 | 0.624 | 1.194 | 1.685 | 0.404 | 0.091 | 0.138 | - | |
| GIR9 | small | 25-26 | 0.347 | 0.387 | 0.168 | 0.662 | 1.206 | 1.740 | 0.455 | 0.095 | 0.157 | - | |
| GIR10 | small | 25-26 | 0.362 | 0.404 | 0.167 | 0.683 | 1.258 | 1.696 | 0.431 | 0.091 | 0.146 | - | |
| GIR11 | small | 25-26 | 0.297 | 0.344 | 0.166 | 0.582 | 1.201 | 1.585 | 0.367 | 0.091 | 0.128 | - | |
| GIR12 | small | 25-26 | 0.361 | 0.404 | 0.171 | 0.700 | 1.299 | 1.781 | 0.404 | 0.082 | 0.126 | - | |
| B) | ID | TL | BW | JW | LDHL | LPHL | LTL | LFL | ED | ND | ENDr | ||
| froglet | 1.867 | 0.888 | 0.819 | 0.968 | 0.952 | 0.480 | 0.899 | 0.437 | 0.195 | 2.241 |
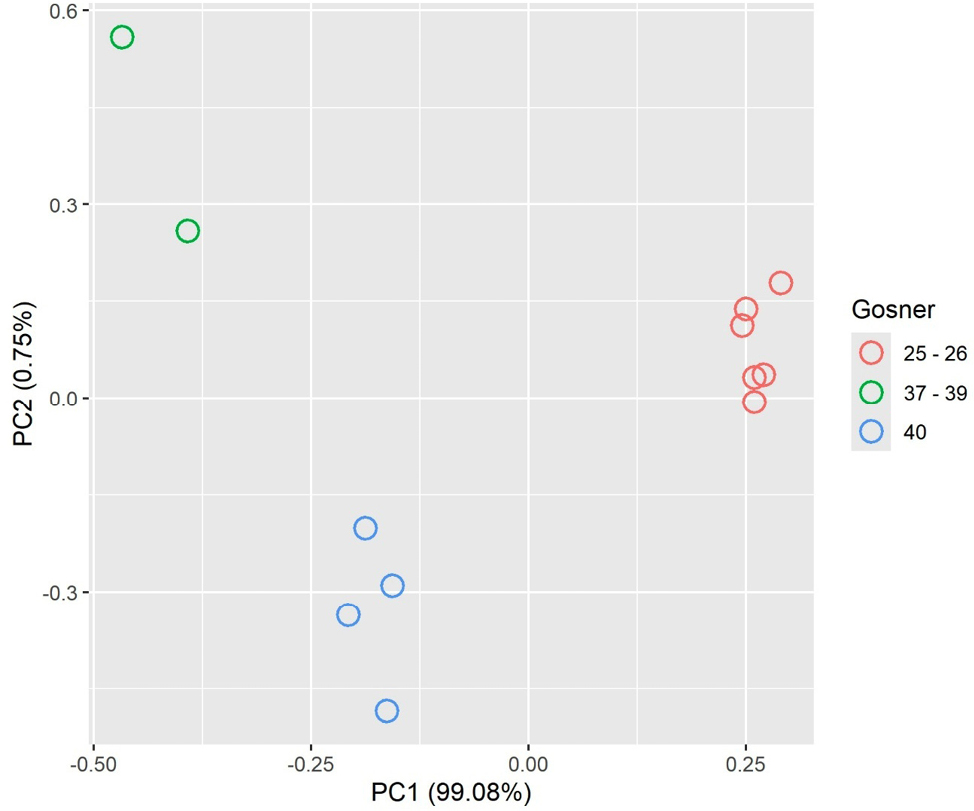
Overall, all tadpoles showed characters matching the typical features of R. latastei [absent, poor, or incomplete ventral colouration and visible guts (Fig. 1E); EDNr of large-sized tadpoles ranged from 1.15 to 1.43 (average: 1.29) (Lanza et al., 2009; Ambrogio and Mezzadri, 2014)]. All the small-sized individuals were between Gosner’s stage 25 and 26, showing functional oral canal but no trace of hindlimb formation. Instead, large-sized tadpoles were at the Gosner’s stage 40 (four individuals; hindlimb with clearly differentiated toes and tubercles, cloacal tail piece visible), or between 37 and 39 (evident toes with no tubercles). The principal component analysis revealed that the first axis explained 99% of variation, identifying three distinct size classes corresponding to the three Gosner’s stages identified, clearly distinguishing as different classes large-sized individuals and newly hatched ones (Fig. 3), also evident from picture comparison (Fig. 1E).
The newly metamorphosed individual measured 1.87 cm (Table 1B) and presented the typical traits of R. latastei (Lanza et al., 2009): the light stripe running above upper lip suddenly stops under the eye (while it is generally prolonged in R. dalmatina), and nostril distance was shorter than eye distance (Fig. 2A-B, Table 1B); moreover, chest and throat looked diffusely spotted and a “T” shaped stripe is noticeable between the throat and the forelimb junction (Fig. 2C), while ventral colouration is generally homogeneously pale and unspotted in R. dalmatina.
Taken together, our observations confirm that all individuals belong to R. latastei. At the same time, the size and late stage of the large tadpoles along with the presence of a froglet and the comparison with newly hatched tadpoles, suggest that large-sized individuals originated from reproductive events occurred in the previous year and underwent overwintering at larval stage. In absence of a direct observation of autumnal reproductive events, only speculations can be made concerning the breeding period. Overwintering tadpoles are not rare in anurans (McDiarmid and Altig, 1999) and can be occasionally observed in other Northern-Italian species [e.g., green frogs, Pelophylax synkl. esculentus (Linnaeus, 1758)] and in some Rana species in other areas (Walsh et al., 2008; Lanza et al., 2009). However, this is generally associated with anurans facing shorter growing seasons, such as late-breeding species, or species and populations from high latitude or elevation (McDiarmid and Altig, 1999), as overwintering can extend the growth period and allow individuals attaining larger size at metamorphosis (Walsh et al., 2008; Iwai, 2024). This is not the case for R. latastei, which is an early breeder, whose tadpoles typically reach metamorphosis in June-July (Lanza et al., 2009).
We suggest these tadpoles originated from one or more breeding events that occurred in late summer or autumn 2024. Tadpoles exposed to low temperatures typically incur in metabolic depression, which slows down growth and development (McDiarmid and Altig, 1999; Enriquez‐Urzelai et al., 2022). However, autumn and winter temperatures of 2024-2025 have been among the mildest in recent decades and 2024 has been the warmest year since consistent temperature monitoring began (https://climate.copernicus.eu/). A similar increase in temperature was recorded at the study site, where precipitation showed large variation with peaks of autumn rainfall in the last two years (see Supplementary material: Table S1; Fig. S1). This could have allowed larval development during the winter months, similarly to what happens in anurans breeding in warmer regions (Sicilia et al., 2006). Additionally, previous records of R. latastei males calling in autumn have been reported for nearby areas (Grossenbacher et al., 2000). While the presence of calling males does not guarantee that breeding activities are occurring, milder autumn and winter temperatures may favour egg deposition and allow tadpole development during this time.
An alternative hypothesis might be that large-sized tadpoles originated from a typical late-winter deposition event in spring 2024. The monitored breeding site is partially shaded by canopy cover, and 20 egg-clutches were laid by February 2024 (the last observed deposition). We cannot exclude that tadpole density and cold water temperatures might have induced delayed development in some individuals at the breeding site. However, tadpole density was comparable to other nearby sites monitored where R. latastei metamorphosed as usual in early summer. Additionally, June–September temperatures of 2024 (mean air temperature ± SD: 29.7 ± 5.1 °C) were markedly higher than those experienced over the typical growing season (March–June temperatures: 21.4 ± 6.1 °C), making it extremely unlikely that overwintering individuals originated from a late-winter 2024 deposition that experienced developmental delays due to low temperatures.
Whatever the period of deposition, the present report reveals an unusual case of shift in breeding phenology for R. latastei, which calls for further investigations assessing the frequency of these events and unravelling drivers and potential implications of such a shift. Indeed, variation in reproductive timing can result in crucial consequences for individual life-history and survival (Bison et al., 2021; Enriquez‐Urzelai et al., 2022). For instance, amphibians developing during winter may face mass mortality risks due to freezing or desynchronization with trophic resources and increase competition (Li et al. 2013); moreover, in explosive breeders, such as wood frogs, these shifts can also trigger cascading effects on amphibian populations and freshwater community dynamics, with complex and rarely predictable outcomes (Todd et al., 2011). This first record of overwintering tadpoles in the Italian agile frog may represent an anomalous or isolated event, but correlation between increasing temperatures, altered precipitation patterns, and shifts in amphibian breeding period have been observed in multiple species as a response to global warming (Todd et al., 2011; Ficetola and Maiorano, 2016). Under the ongoing climate change scenarios, amphibian phenology is expected to be considerably impacted worldwide, and variations in their reproductive activity should deserve particular attention. Further research should aim at unravelling physiological mechanisms regulating shifts in breeding phenology and their implications on amphibian life-history traits and fitness. In light of the present case, autumnal monitoring of the study site and surrounding areas will be planned during the incoming years to ascertain the occurrence of R. latastei depositions outside the typical reproductive season. This study case also stresses the importance of constant monitoring of amphibian activity and phenology in a climate change scenario, calling for increased surveillance to implement appropriate conservation strategies.
Acknowledgements and authorizations:
We are grateful to Arturo Binda for his longstanding help in the field surveys. We also wish to thank Caspani family and friends for welcoming us in their properties during field surveys. The research was conducted under permits released by Italian Ministry of Environment (MiTE 0032672, released on 15th March 2022).
Supplementary material associated with this article can be found at: https://oaj.fupress.net/index.php/ah/article/view/17665/14575.
Ambrogio, A., Mezzadri, S. (2014): Girini d’Italia - tadpoles of Italy. Piacenza, Gavia Edizioni.
Ambrogio, A., Mezzadri, S. (2018): Brown frogs of Italy. Piacenza, Gavia Edizioni.
Barbieri, F., Bernini, F., Vercesi, A., Lavizzari, G. (2000): Caratteri meristici e morfologici del disco orale negli stadi larvali di Rana latastei e Rana dalmatina. Riv. Idrobiol. 38: 19.
Benard, M.F. (2015): Warmer winters reduce frog fecundity and shift breeding phenology, which consequently alters larval development and metamorphic timing. Glob. Change Biol. 21: 1058-1065.
Bison, M., Yoccoz, N.G., Carlson, B.Z., Klein, G., Laigle, I., Van Reeth, C., Delestrade, A. (2021): Earlier snowmelt advances breeding phenology of the common frog (Rana temporaria) but increases the risk of frost exposure and wetland drying. Front. Ecol. Evol. 9: 645585.
Bonaparte, C. L. J. L. (1838): Iconographia della fauna italica per le quattro classi degli animali vertebrati. Tomo II. Amphibi. Fascicolo 22. Rome: Salviucci.
Boulenger, G. A. (1879): Étude sur les grenouilles rousses, Ranae temporariae et description d’espèces nouvelles ou méconnues. Bull. Soc. Zool. Fr. 4: 158-193.
Dalpasso, A., Seglie, D., Eusebio Bergò, P., Ciracì, A., Compostella, M., Laddaga, L., Manica, M., Marino, G., Pandolfo, I., Soldato, G. (2023): Effects of temperature and precipitation changes on shifts in breeding phenology of an endangered toad. Sci. Rep. 13: 14573.
Enriquez‐Urzelai, U., Nicieza, A.G., Montori, A., Llorente, G.A., Urrutia, M.B. (2022): Physiology and acclimation potential are tuned with phenology in larvae of a prolonged breeder amphibian. Oikos 2022: e08566.
Falaschi, M., Giachello, S., Lo Parrino, E., Muraro, M., Manenti, R., Ficetola, G.F. (2021): Long‐term drivers of persistence and colonization dynamics in spatially structured amphibian populations. Conserv. Biol. 35: 1530-1539.
Ficetola, G.F., Maiorano, L. (2016): Contrasting effects of temperature and precipitation change on amphibian phenology, abundance and performance. Oecologia 181: 683-693.
Ficetola, G.F., Novarini, N., Seglie, D. (2025): Rana latastei Boulenger, 1879. In: Atlante degli Anfibi e Rettili d’Italia - Atlas of Italian Amphibians and Reptiles, pp. 204-207. Razzetti, E., Bruni, G., Di Tizio, L., Liuzzi, C., Sindaco, R., Eds, Latina, Societas Herpetologica Italica, Edizioni Belvedere.
Ficetola, G.F., Padoa‐Schioppa, E., De Bernardi, F. (2009): Influence of landscape elements in riparian buffers on the conservation of semiaquatic amphibians. Conserv. Biol. 23: 114-123.
Gosner, K.L. (1960): A simplified table for staging anuran embryos and larvae with notes on identification. Herpetologica 16: 183-190.
Graña, R.V., Martínez-Freiría, F. (2020): First record of autumnal mating in the Iberian adder, Vipera seoanei. Boletín de la Asociación Herpetológica Española 32: 31-35.
Grossenbacher, K., Lippuner, M., Zumbach, S., Borgula, A., Lüscher, B. (2000): Phenology and reproduction of the 3 brown frog species Rana latastei, R. dalmatina, R. temporaria; development and status of the R. latastei populations in Mendrisiotto, Southern Ticino, Switzerland. In: Atti del terzo Convegno “Salvaguardia Anfibi”, Lugano, 23-24 giugno 2000, pp. 99-100. Ferri V., Ed, Penne, Cogecstre Edizioni.
Horton, K.G., La Sorte, F.A., Sheldon, D., Lin, T.-Y., Winner, K., Bernstein, G., Maji, S., Hochachka, W.M., Farnsworth, A. (2020): Phenology of nocturnal avian migration has shifted at the continental scale. Nat. Clim. Change 10: 63-68.
Iwai, N. (2024): Effects of growing season and individual growth rates on the occurrence of larval overwintering in Otton frog tadpoles. J. Zool. 323: 284-291.
Lang, W., Zhang, Y., Li, X., Meng, F., Liu, Q., Wang, K., Xu, H., Chen, A., Peñuelas, J., Janssens, I. A. (2025): Phenological divergence between plants and animals under climate change. Nat. Ecol. Evol. 9: 261-272.
Lanza, B., Nistri, A., Vanni, S. (2009): Anfibi d’Italia. Ministero dell’Ambiente e della Tutela del Territorio e del Mare/Istituto Superiore per la protezione la ricerca ambientale. Modena, Grandi & Grandi Editori.
Laurenti, J. N. (1768): Specimen medicum, exhibens synopsin reptilium emendatum cum experimentis circa venena et antidota reptilium austriacorum. Vienna, Johann Thomas von Trattner.
Linnaeus, C. (1758): Systema Naturae, per Regna Tria Naturae, secundum Classes, Ordines, Genera, Species, cum Characteribus, Differentiis, Synonymis, Locis, tenth ed., Tomus I. Stockholm, Laurentii Salvii.
Li, Y., Cohen, J.M., Rohr, J.R. (2013): Review and synthesis of the effects of climate change on amphibians. Integr. Zool. 8: 145-161.
Manenti, R., Falaschi, M., Delle Monache, D., Marta, S., Ficetola, G.F. (2020): Network-scale effects of invasive species on spatially-structured amphibian populations. Ecography 43: 119-127.
McDiarmid, R.W., Altig, R. (1999): Tadpoles: The biology of anuran larvae. Chicago, University of Chicago Press.
Melotto, A., Ficetola, G.F., Pennati, R., Ancona, N., Manenti, R. (2021): Raised by aliens: Constant exposure to an invasive predator triggers morphological but not behavioural plasticity in a threatened species tadpoles. Biol. Invasions 23: 3777-3793.
Melotto, A., Manenti, R., Ficetola, G.F. (2020): Rapid adaptation to invasive predators overwhelms natural gradients of intraspecific variation. Nat. Comm. 11: 3608.
Parmesan, C. (2007): Influences of species, latitudes and methodologies on estimates of phenological response to global warming. Glob. Change Biol. 13: 1860-1872.
Relyea, R.A. (2001): Morphological and behavioral plasticity of larval anurans in response to different predators. Ecology 82: 523-540.
Rodriguez-Muñoz, M.J., Galdeano, A.P., Martínez, T.A., Acosta, R., Acosta, J.C., Blanco, G. (2020): First report of overwintering in tadpoles of Odontophrynus occidentalis (Anura: Odontophrynidae) from Argentina. Phyllomedusa 19: 117-120.
Schneider, C.A., Rasband, W.S., Eliceiri, K.W. (2012): NIH image to ImageJ: 25 years of image analysis. Nat. Methods 9: 671-675.
Sicilia, A., Bernini, F., Zava, B., Lillo, F. (2006): Breeding phenology of Bufo viridis Laurenti, 1768 in Sicily. Acta Herpetol. 1: 107-117.
Todd, B.D., Scott, D.E., Pechmann, J.H., Gibbons, J.W. (2011): Climate change correlates with rapid delays and advancements in reproductive timing in an amphibian community. P. Roy. Soc. B-Bio. Sci. 278: 2191-2197.
Walsh, P.T., Downie, J., Monaghan, P. (2008): Larval over‐wintering: Plasticity in the timing of life‐history events in the common frog. J. Zool. 276: 394-401.