Submitted on: 2025, 27th April; revised on: 2025, 19th October; accepted on: 2025, 30th October
Behavioral repertoire of the Madeiran wall lizard, Teira dugesii (Sauria: Lacertidae) in wildlife conditions
1 Universidade da Madeira, Faculdade de Ciências da Vida. 9000-082, Funchal, Ilha da Madeira, Portugal
2 Coleção de Insetos da Universidade da Madeira (UMACI), Portugal
3 Madeira Botanical Group (GBM), Universidade da Madeira, Faculdade de Ciências da Vida. 9000-082, Funchal, Ilha da Madeira, Portugal
*Corresponding author. Email: tomaschool@outlook.com
Abstract. The behavioral repertoire of the Madeiran wall lizard, Teira dugesii, was compiled from approximately 70 hours of direct field observations conducted between July 2022 and August 2024 across seven locations along the south coast of Madeira Island. A total of 73 distinct behaviors were recorded and categorized into five groups: maintenance, aggression and defense, feeding, mobility, and reproduction. Observations took place during the species’ daily activity period (09:00–15:00), primarily during the summer months. Maintenance behaviors accounted for the majority of records (86.89%), followed by mobility (6.95%), aggression and defense (3.92%), feeding (1.83%), and reproduction (0.41%). These findings provide new insights into the diverse and complex behavioral repertoire of T. dugesii under natural conditions.
Keywords. Behavior, Madeira Island, Lacertidae, ethogram, Macaronesia.
Index
Selection of observation sites and sampling periods
Aggressive and defensive behaviors
Mobility and feeding behaviors
Aggressive and defensive behaviors
Diurnal lizards serve as exceptional models for studying behavioral evolution due to the broad spectrum of traits exhibited across different taxa, populations, and species (Langkilde et al., 2003; Poma-Soto et al., 2021). In behavioral research, compiling ethograms is a crucial step in systematically documenting and classifying a species’ behavioral repertoire (Martin and Bateson, 2021). An ethogram is defined as a catalog of species-specific behaviors, encompassing descriptions, classifications, and habits that constitute the fundamental behavioral repertoire of a species (Martin and Bateson, 1994; Qi et al., 2011; Mather, 2025). Since behavioral patterns can vary significantly among individuals (Ruby and Niblick, 1994), constructing an ethogram is essential for identifying similarities and differences in behavior, thereby facilitating the formulation of novel research questions (Liu et al., 2009; Qi et al., 2011). Typically, ethograms provide a structured list of observed behaviors, each accompanied by a precise definition using descriptive terminology (Martin and Bateson, 2007). Ethograms offer valuable insights into a species’ life history and are particularly useful in studies focusing on courtship, intrasexual competition, and foraging behavior (Coelho et al., 2018). While behavioral research in captivity can yield important data, it often presents challenges due to the artificial nature of the environment, which may lead to stress-induced alterations in behavior (Carlstead, 1996). Whenever possible, observations should be conducted in natural settings to ensure ecological validity (Mather, 2025).
Teira dugesii (Milne-Edwards, 1829) is an endemic lacertid lizard of the Madeira Archipelago and Selvagens Islands (Jesus et al., 2009). Over time, it has been introduced to the Azores Archipelago (Ulfstrand, 1961), Gran Canaria Island (Santos et al., 2015), and in two cities of mainland Portugal, such as Lisbon (Sá-Sousa, 1995), and Porto (Ferreira et al., 2023). In its native distribution range, this species thrives in diverse habitats, from sea level up to elevations of 1,861 meters. It adapts to various substrates and degrees of sun exposure, although it is less common in shaded environments (Jesus et al., 2009). Currently, four subspecies are recognized: Teira dugesii dugesii (Madeira Island), Teira dugesii jogeri (Porto Santo Island), Teira dugesii selvagensis (Selvagens Islands), and Teira dugesii mauli (Desertas Islands) (Brehm et al., 2003; Jesus et al., 2009).
Although Teira dugesii is a well-known component of Madeira’s terrestrial fauna, behavioral studies on this species remain relatively scarce and fragmented. Nonetheless, some aspects of its behavior have been documented. These include arboreal activity (Petrovan, 2023), instances of predation on T. dugesii by other animals (Câmara and Jesus, 2025a), as well as predation by T. dugesii itself on various invertebrate and vertebrate prey (Matias et al., 2009). Reproductive behavior has also been addressed, although primarily under laboratory or captive conditions (Sleijpen, 1996; Galán, 2003).
Selection of observation sites and sampling periods
A total of approximately 70 hours of behavioral observations and video recordings of Teira dugesii were conducted between July 2022 and August 2024. Observations were distributed across four sampling periods and carried out under natural field conditions, without any experimental manipulation of temperature or other environmental variables. The first sampling period, conducted between July and September 2022, comprised approximately 36 hours of observations. Sessions were held three times per week and focused on the hours of peak activity, specifically between 10:00–12:00 and 13:00–15:00. A second period took place from 1 to 25 October 2022, during which around 10 hours of data were collected. Recordings occurred three times per week, exclusively on sunny days, between 10:00 and 12:00. Additional observations were carried out over seven days in early January 2024, yielding approximately 12 hours of data collected between 11:00 and 13:00. The final sampling period occurred between 27 July and 5 August 2024, during which a further 12 hours of observations were conducted between 11:00 and 12:30.
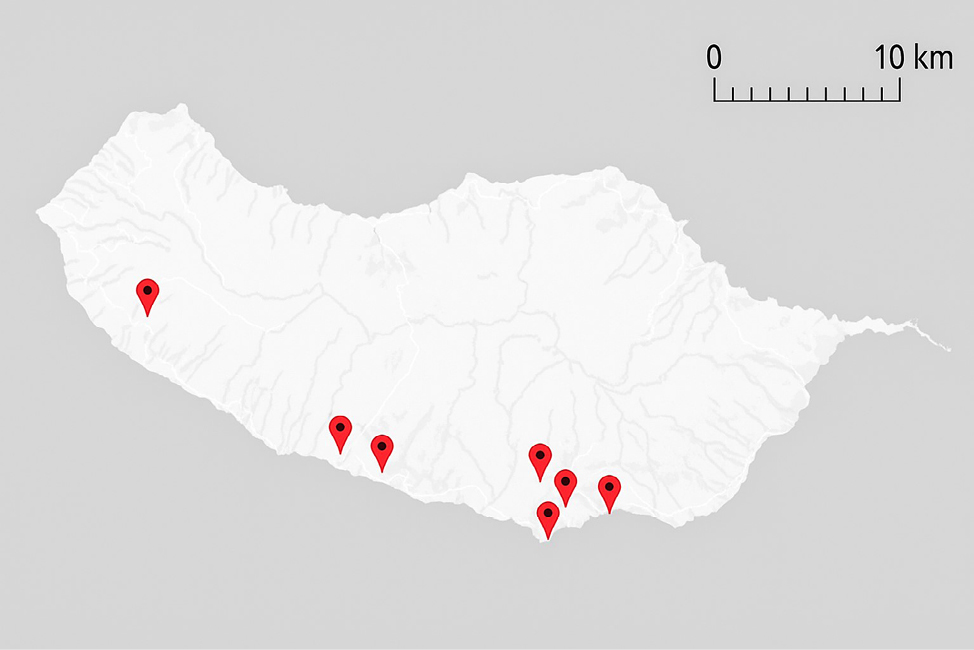
Behavioral observations were conducted at seven distinct locations (Table 1, Fig. 1) along the southern coast of Madeira, a region chosen for its warmer and more stable climatic conditions throughout the year, which allow for increased lizard activity and more frequent observations compared to cooler, wetter areas or dense laurel forests (Laurissilva) in the north, where Teira dugesii is rare or difficult to detect. In selecting these sites, we aimed to maintain a minimum distance of approximately 1 km between them to minimize the likelihood of repeatedly observing the same individuals and to increase spatial representativeness. Observations were primarily focused on substrates frequently used by T. dugesii, including stone walls, tree trunks, and rocky surfaces. Although no detailed studies currently exist on the species’ microhabitat preferences, this approach was based on direct field observations and aimed to ensure a broad and representative sampling of behavioral patterns across a variety of natural, semi-natural, and anthropogenic environments. Human presence at each site was qualitatively classified into three categories: “low”, “medium”, and “high”. Categorization was based on direct field observations during behavioral sessions, with “low” referring to areas with rare or occasional pedestrian activity, typically in quieter residential or rural zones; “medium” included locations with regular but non-disruptive human activity, such as university campuses or suburban viewpoints; “high” was assigned to sites frequently visited by tourists or the general public, such as botanical gardens and coastal recreational areas, where constant human movement was observed throughout the day.
| Location | Coordinates | Human Pressure |
|---|---|---|
| Rua Nova da Igreja, Funchal | 32°38′55″N, 16°53′4″W | Low |
| Lido, Funchal | 32°38′11″N, 16°56′4″W | High |
| Jardim Botânico, Funchal | 32°39′46″N, 16°53′42″W | High |
| Campanário, Ribeira Brava | Medium | |
| Miradouro D. Sebastião, Ribeira Brava | 32°40′12″N, 17°3′46″W | Low/Medium |
| Prazeres, Calheta | 32°45′15″N, 17°12′9″W | Low/Medium |
| Campus Penteada, Funchal | 32°39′33″N, 16°55′25″W | Medium |
A Nikon D3500 and a Nikon P1000 were used for video recordings and photography. Both devices allow simultaneous video and photo capture, enabling the collection of still images during active recording sessions. No tripod was used, which allowed the observer to follow rapid movements more effectively in the field. Most of the video material was recorded using the Nikon P1000, which provided higher optical zoom and flexibility in frame rate and resolution settings. The Nikon D3500 was used for only a small portion of recordings, representing approximately one hour of total footage. Recordings were conducted across all seasons, with the majority of observations and video sessions taking place during the summer months. In addition to video and photographic documentation, field notes and sketches from a notebook were also incorporated into the dataset.
To document behavioral patterns, we employed two sampling methods: (1) ad libitum sampling, which allowed for the recording of spontaneous and infrequent behaviors, and (2) focal animal sampling, which provided a structured approach to observing specific individuals (Martin and Bateson, 1994). The duration of each focal session was variable, as observations continued either through recording or field notes until the focal individual left the area or was no longer visible. The combination of these methods ensured both broad behavioral coverage and detailed individual-level data.
To prevent behavioral alterations, a minimum observation distance of approximately 5 meters was maintained during all sessions. Preliminary trials revealed that closer proximity (less than 5 meters) consistently caused Teira dugesii individuals to either flee or adopt a state of alert immobility, thus compromising the reliability of behavioral recordings. It is important to note, however, that in areas with high human presence, such as the Madeira Botanical Garden and coastal lidos, individuals exhibited significantly reduced wariness. In these settings, lizards often continued their natural movements without displaying typical alert or escape behaviors in response to human observers, likely due to habituation. A similar distance was used by Molina-Borja (1987) in a behavioral study of Gallotia galloti.
Sex differentiation followed Jesus et al. (2009), who described adult males as generally larger, with a broader head and tail base. In contrast, females and young males exhibit similar external features, making them difficult to distinguish in the field. Additionally, individuals show variable color patterns (Báez, 1990). Dorsal coloration may be uniform or divided by longitudinal stripes, with background hues ranging from light brown to black. On Madeira Island, females, juveniles, and subadults typically exhibit light longitudinal stripes, whereas adult males tend to display greater variation in dorsal patterns. Although generally more uniform, females also present some degree of variation. Ventral coloration may be uniform or mottled with spots of various colors, with background tones ranging from cream, yellowish-green, blue, orange, and brown to black (Báez, 1990).
The behaviors of Teira dugesii were categorized into five main groups based on Molina Borja (1981, 1987). (1) Maintenance: elementary postures and movements, as well as different types of orientation behaviors related to body positioning and environmental awareness; (2) Mobility: locomotor patterns exhibited by individuals, including walking, running, climbing, and other forms of displacement; (3) Feeding: all actions related to foraging, prey capture, and food manipulation; (4) Aggression and defense: various orientations, such as direct confrontations and visual displays, as well as complex postures and movements, which may involve gular inflation, tail displays, or other intimidation tactics used in intra- or interspecific interactions; (5) Reproduction: courtship displays, mating postures, and any observed interactions between males and females related to reproduction.
Video footage was systematically reviewed after fieldwork, with behavioral events manually identified and categorized. The data were then summarized as absolute counts and/or percentages of total recorded events. All video footage was reviewed and classified by a single observer (J.C.), which ensured internal consistency in behavioral identification and reduced the risk of inter-observer variability. In cases of uncertainty, video segments were reviewed multiple times and cross-referenced with field notes for clarification. No automated software or external observers were involved in the identification process, allowing for a flexible, case-by-case approach to ambiguous behaviors. Field notes were used to complement the classification process, ensuring accurate interpretation of behaviors. Each behavioral event was assigned to a single category, such as foraging, basking, locomotion, or social interaction. Although the categories were generally mutually exclusive, more complex behaviors occasionally involved overlapping or sequential components. In such cases, a hierarchical rule was applied to assign the event to the most dominant or functionally relevant category. For example, if an individual was moving while visually scanning the ground, the behavior was only classified as foraging if active prey-searching was clearly observed, and not simultaneously as both locomotion and foraging. This approach ensured that no behaviors were double-counted. Once assigned, behaviors were excluded from any other potentially overlapping categories.
The values presented in the results were obtained by counting the number of occurrences of each behavior throughout the analyzed footage. In some cases, the duration of behaviors was also recorded (e.g., time spent basking).
The site with the highest number of observation hours was Rua Nova da Igreja, a quiet residential area with minimal pedestrian traffic during observation periods. The main microhabitat consisted of a stone wall approximately two meters high and several meters long, characterized by high sun exposure and the presence of ornamental potted plants. In contrast, sites such as botanical gardens experienced occasional tourist presence, and observations there were mostly restricted to vertical structures such as stone walls or tree trunks. A minimum of approximately 100 distinct individuals is estimated to have been observed throughout the study. Observed behaviors are illustrated in Table 2, and their frequencies are in Table 3.
| Behavior category | Behaviors | Description |
|---|---|---|
| Maintenance |  Head-Down Head-Down |
Head resting on the substrate. |
 Head-Up Head-Up |
Lower surface of the head detached from the substrate. | |
 Body-Up Body-Up |
The front and back ends elevate the animal’s body to different degrees. | |
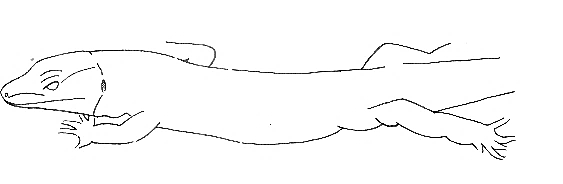 Body-Down |
Ventral region supported by substrates. | |
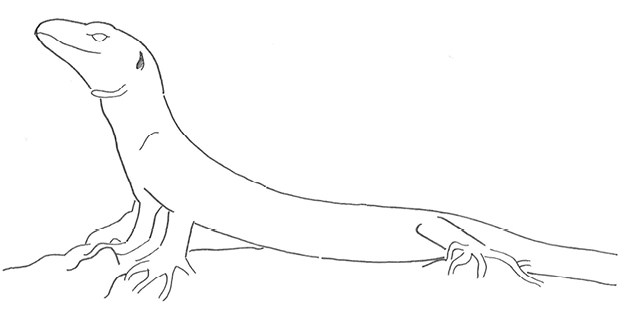 Anterior Body-Up |
Posterior ends resting on the substrate while the anterior ends raise the anterior part of the animal’s body in different degrees. | |
 Tail-Low Tail-Low |
Tail resting, in all its extension, on the substrate. | |
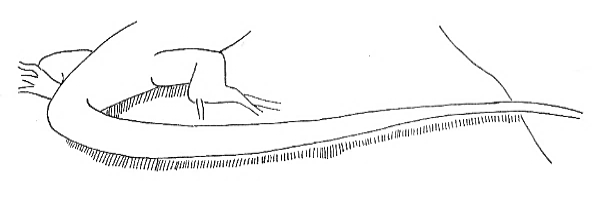 Tail-Up |
Tail raised in varying degrees from the substrate. | |
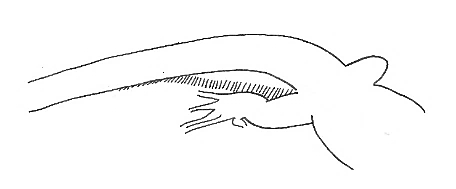 Tail-Up-At-Base Tail-Up-At-Base |
Tail raised at the base while the distal part remains on the substrate. The tail in this position forms an arch. | |
 Tail-Up-At-Tip Tail-Up-At-Tip |
Tail raised at the distal part, while de base part remains on the substrate. | |
 S-Tail S-Tail |
Tail on the ground, bent in a S shape. | |
 C-Tail C-Tail |
Tail on the ground, bent in a C shape. | |
 Foot-Up Foot-Up |
One or more feet rise above the substrate or all four of them. | |
 Toes-Up Toes-Up |
The toes of one or more feet rise above the substrate. | |
 Mouth-Open Mouth-Open |
Opening in varying degrees of the mouth. | |
 Lie-on Lie-on |
A lizard rests part of its body on another animal and maintains that position. | |
 Quiet/rest Quiet/rest |
Body extended against substrate; head slightly elevated. | |
 Eyes closed Eyes closed |
The eyelids close over the eyes. | |
 Sleep Sleep |
Eyes closed, body in all its extension resting on the substratum, head down. | |
 Oblique-Orientation |
Oblique position of the lizard through the support of its forelimbs on a protruding part of the substrate. | |
 Extreme Oblique Orientation Extreme Oblique Orientation |
The lizard clings to irregularities in the substrate, remaining almost vertical. | |
 Adpress Adpress |
One or more limbs are raised off the substrate and held against the lateral side of the body. | |
 Bask Bask |
The body is flattened onto the substrate and oriented at right angles to a heat source. | |
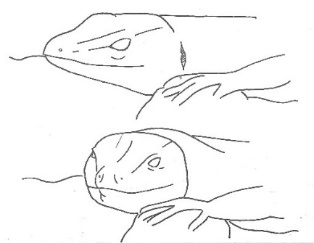 Head-to-side Head-to-side |
The lizard keeps its head slightly raised at an angle of 90º to its body, keeping it resting on the substrate for its entire length. The head can stand still pointing either to the left or to the right. | |
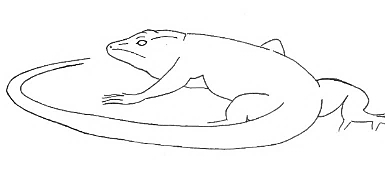 C-Body C-Body |
The lizard stands still in a C-shape. | |
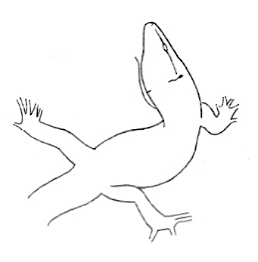 S-Body S-Body |
The lizard stands still in an S-shape due to the irregularities of the substrate. | |
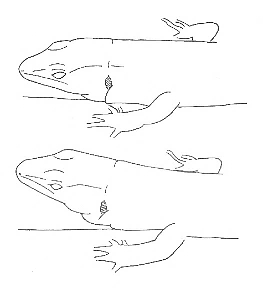 Head Up-Down Head Up-Down |
The lizard moves its head up and down along a vertical axis. | |
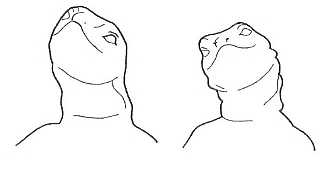 Head rotation Head rotation |
Movement in which one side of the head is oriented upwards and the other downwards. | |
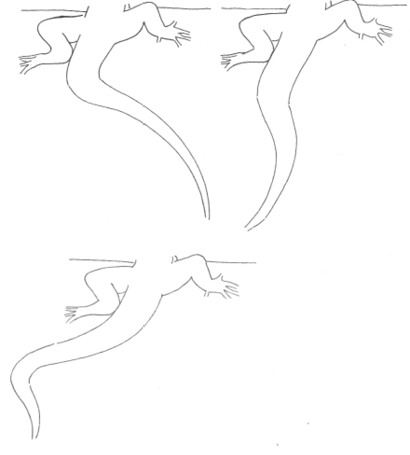 Tail-Lash Tail-Lash |
Side-to-side undulation of the tail. | |
 Tail-Twitch Tail-Twitch |
Rapid movement of the distal part of the tail. | |
 Contortion of the body Contortion of the body |
Undulating movement of the body. | |
 Mouth-Scrape |
The lizard scratches one side of its mouth on the substrate, and slightly lifts its body on the opposite side of the scratched part. | |
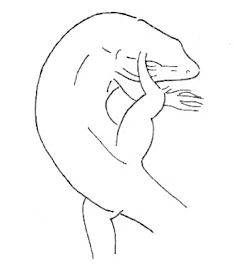 Scratch Scratch |
The Body is scratched by one of the hind limbs. | |
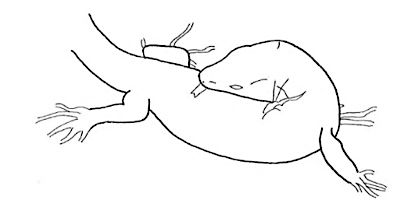 Groom Groom |
A lizard bit or licks its own body. | |
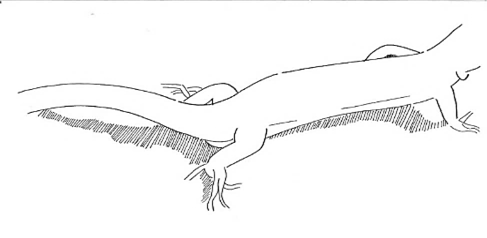
 Digging Digging |
The lizard moves its head towards the substrate and starts moving its front legs from front to back, in a repetitive movement. | |
 Cloaca-Drag Cloaca-Drag |
With the body and tail - slightly Up, the lizard drags the cloaca over the substrate. | |
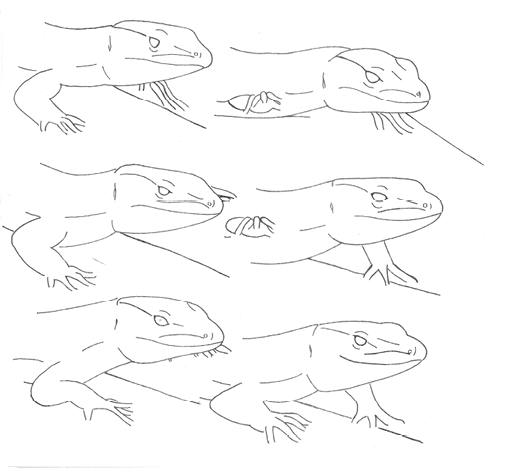 “Palateo” “Palateo” |
The lizard in “head up” position strikes the substratum with one (or both) front paws in a repetitive, short and fast movement. | |
| Mobility behaviors | Jumping | The lizard jumps from one place to another, using its rear legs to propel its body forward. |
| Walking | Body slightly raised, tail down, forward movements. During the forward movement, one of the forelegs and the contralateral rear leg are moved simultaneously, followed by the corresponding symmetrical legs. As a result, the trunk of the animal shows an undulating movement (contortion of the body motion). | |
| Run | Fast forward movement, with body and tail raised also with contortion of the body motion. | |
| Forage | Foraging behaviour was distinguished from general locomotion based on consistent behavioural cues. Lizards identified as foraging paused frequently, often in vegetated or debris-rich areas, and exhibited repeated tongue-flicking, as well as marked lateral or vertical head movements indicative of active environmental scanning. In contrast, individuals engaged in simple locomotion typically moved in a more continuous manner, with little or no tongue-flicking and without pronounced head movement. These criteria allowed us to differentiate purposeful foraging from mere traversal of the habitat. | |
| Move-over | One lizard moves over the top of another. | |
| Arboreal | The arboreal movement refers to the displacement of animals through trees. | |
| Feeding behavior | 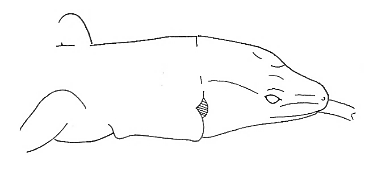 Tongue Touch Tongue Touch |
The tongue briefly touches the substrate, water, or other animal. |
 Food-Licking Food-Licking |
Repeated projections of a lizard’s tongue towards food. | |
 Lick Lick |
Repeated projections of a lizard’s tongue. | |
 Mandible lick Mandible lick |
Displacement of the animal’s tongue over its mandible. | |
 Food-Bite Food-Bite |
The lizard opens its mouth and grabs food with its mandible. | |
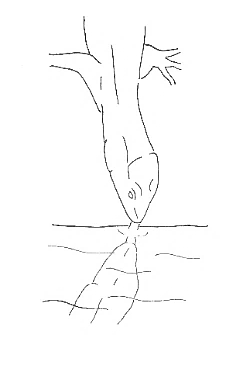 Drinking Drinking |
Repetitive projection and retraction movements of the tongue over or in a liquid. | |
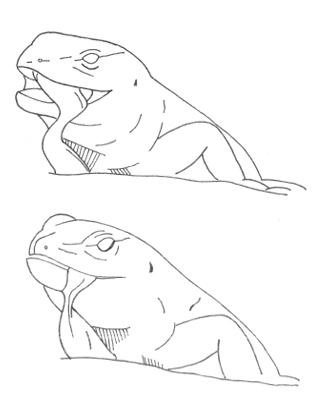 Chew Chew |
The lizard repeatedly moves the mandible up and down, mandibulation. | |
 Food-shaking Food-shaking |
A food item is shaken by side-to-side motion of the head. | |
 Defecation Defecation |
The lizard assumes the Tail-Up-At-Base posture, progressively arching the tail until expelling the faecal material and producing the Cloaca-Drag movement. After a few steps, the lizard lowers its tail. | |
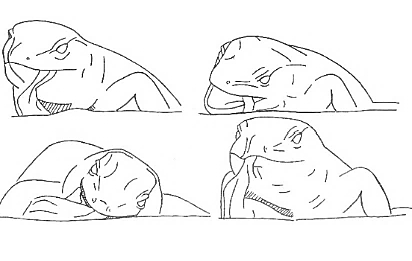
| Aggression and defence behaviors |  Threat position Threat position |
The lizard assumes the Body-Up posture, bends its head downwards, arches its back, and produces the Gular movement. |
 Gular movement Gular movement |
Expansion of the throat region. | |
 Lateral Threat Orientation |
Lateral orientation of one lizard relative to another during aggressive encounters. | |
 Perpendicular Threat Orientation Perpendicular Threat Orientation |
Perpendicular orientation of one lizard relative to another during aggressive encounters. | |
 Circling Circling |
Circular movement of two lizards in lateral threat orientation, with heads facing opposite sides. | |
 Lunge Lunge |
Rapid jumping movement by one lizard toward another. | |
 Stalk Stalk |
A slow approach to a stimulus. | |
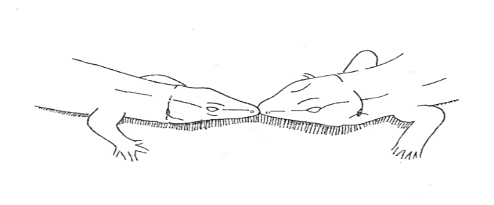 Head-Touch Head-Touch |
Two lizards touch snouts facing each other. | |
 Bite Bite |
The lizard opens its mouth and grabs another animal with its mandible. | |
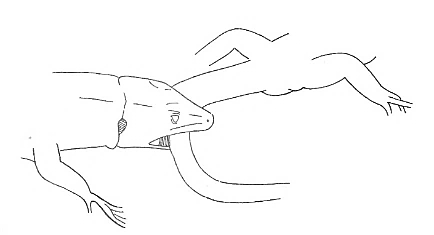 NIP NIP |
One lizard grasp another with its jaws and then releases immediately (less than one second later). | |
 Tongue Flick Tongue Flick |
Rapid movement of projection and retraction of the tongue, however, this behavior is also used for reconnaissance. | |
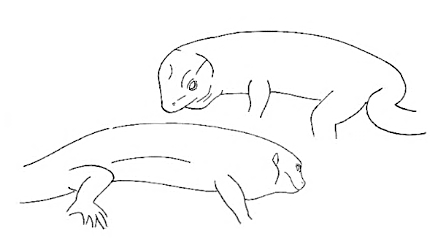 Face-OFF Face-OFF |
Two male lizards stare at each other. | |
 Roll-up Roll-up |
The predated lizard wraps its entire body, including its tail, around the predatory lizard. | |
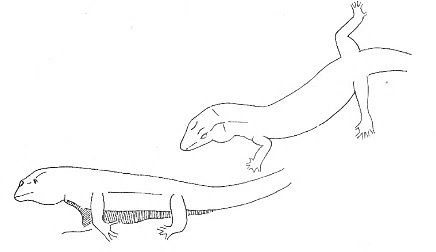 Retreat Retreat |
Move rapidly towards the opposite direction of the opponent. | |
| Reproductive behaviors | Pre copulation | The male observes the female and slowly approaches her. Once relatively close, he pauses for a few seconds before suddenly lunging toward her. |
| Copulatory bite | The male bites the female’s lower trunk region from the side. | |
| Female copulatory bite | The female bites the male’s head or trunk region during copulation. | |
| Copulation | Following the bite, the male curves his body into a C-shape, aligning his cloaca with that of the female, and wraps his tail around her. He then inserts one of his hemipenes into the female’s cloaca. | |
| Release bite | The male bites the female’s cloacal region during copulation. | |
| Terminating copulation | Copulation ends with the separation of the male’s genitalia from the female’s cloaca, as the male straightens his body. | |
| Post copulatory | After withdrawal, the male raises his head and neck, keeping the anterior part of his body elevated. He rests his abdomen on the ground, lifts his cloacal region and the base of the tail off the ground, while the tip of the tail remains in contact with the substrate. The hemipenes are still externally visible. |
| Behavioral Category | Behavior | Counts | Percentage (%) |
|---|---|---|---|
| Maintenance | Head-Down | 465 | 13.62 |
| Head-Up | 767 | 22.47 | |
| Body-Up | 78 | 2.28 | |
| Body-Down | 636 | 18.63 | |
| Anterior Body-Up | 95 | 2.78 | |
| Tail-Low | 178 | 5.21 | |
| Tail-Up | 6 | 0.18 | |
| Tail-Up-At-Base | 3 | 0.09 | |
| Tail-Up-At-Tip | 2 | 0.06 | |
| S-Tail | 6 | 0.18 | |
| C-Tail | 33 | 0.97 | |
| Foot-Up | 221 | 6.47 | |
| Toes-Up | 111 | 3.25 | |
| Mouth-Open | 19 | 0.56 | |
| Lie-on | 6 | 0.18 | |
| Quiet/rest | 26 | 0.76 | |
| Eyes closed | 24 | 0.70 | |
| Sleep | 2 | 0.06 | |
| Oblique-Orientation | 15 | 0.44 | |
| Extreme Oblique Orientation | 22 | 0.64 | |
| Adpress | 76 | 2.23 | |
| Bask | 446 | 13.06 | |
| Head-to-side | 33 | 0.97 | |
| C-Body | 37 | 1.08 | |
| S-Body | 3 | 0.09 | |
| Head Up-Down | 3 | 0.09 | |
| Head rotation | 3 | 0.09 | |
| Tail-Lash | 19 | 0.56 | |
| Tail-Twitch | 5 | 0.15 | |
| Contortion of the body | 40 | 1.17 | |
| Mouth-Scrape | 3 | 0.09 | |
| Scratch | 3 | 0.09 | |
| Groom | 4 | 0.12 | |
| Digging | 1 | 0.03 | |
| Cloaca-Drag | 5 | 0.15 | |
| “Palateo” | 18 | 0.53 | |
| Total | 3414 | 100 | |
| Mobility | Jumping | 20 | 7.33 |
| Walking | 152 | 55.68 | |
| Run | 65 | 23.81 | |
| Forage | 15 | 5.49 | |
| Move-over | 2 | 0.73 | |
| Arboreal | 19 | 6.96 | |
| Total | 273 | 100 | |
| Feeding | Tongue Touch | 27 | 37.50 |
| Food-Licking | 7 | 9.72 | |
| Lick | 16 | 22.22 | |
| Mandible lick | 7 | 9.72 | |
| Food-Bite | 2 | 2.78 | |
| Drinking | 7 | 9.72 | |
| Chew | 2 | 2.78 | |
| Food-shaking | 2 | 2.78 | |
| Defecation | 2 | 2.78 | |
| Total | 72 | 100 | |
| Aggression/Defence | Alert | 34 | 22.08 |
| Threat position | 5 | 3.25 | |
| Lateral Threat Orientation | 6 | 3.90 | |
| Perpendicular Threat Orientation | 3 | 1.95 | |
| Gular movement | 5 | 3.25 | |
| Circling | 5 | 3.25 | |
| Lunge | 3 | 1.95 | |
| Bite | 3 | 1.95 | |
| Stalk | 17 | 11.04 | |
| Head-Touch | 3 | 1.95 | |
| NIP | 6 | 3.90 | |
| Tongue Flick | 47 | 30.52 | |
| Face-OFF | 1 | 0.65 | |
| Roll-up | 2 | 1.30 | |
| Retreat | 14 | 9.09 | |
| Total | 154 | 100 | |
| Reproductive | Pre copulation | 7 | 43.75 |
| Copulatory bite | 3 | 18.75 | |
| Female copulatory bite | 2 | 12.50 | |
| Copulation | 1 | 6.25 | |
| Release bite | 1 | 6.25 | |
| Terminating copulation | 1 | 6.25 | |
| Post copulatory | 1 | 6.25 | |
| Total | 16 | 100 |
Maintenance behaviors represented 86.81% of all recorded observations (3414 of 3929), highlighting their central role in the daily activity budget of Teira dugesii. The most frequently recorded postures included head-up (767 observations), body-down (636), head-down (465), and body-up (113). These behaviors were evenly distributed throughout the observation periods, with no marked concentration at specific times of day, suggesting a balanced expression of maintenance activities over the diurnal cycle.
In addition to postural behaviors, basking (516 observations, in which the lizard flattens its body against the substrate), was also commonly observed. Other thermoregulatory postures included foot-up (221), toes-up (111), and mouth-open (19), which may facilitate heat absorption and exchange. These behaviors occurred most frequently on sunlit stone walls, tree trunks, and rocky surfaces.
Self-directed maintenance behaviors, although less frequent, were also recorded. These included grooming (4 observations), scratching (3), body contortion (40), mouth scraping (3), and palateo (18) (Table 3) – a tongue flick behavior potentially linked to chemoreception or hydration.
Overall, maintenance behaviors overwhelmingly dominated the activity budget of T. dugesii, contrasting with the markedly lower frequencies of mobility (6.95%), feeding (1.83%), aggression and defense (3.92%), and reproductive behaviors (0.41%).
Mobility behaviors represented 6.95% of all recorded observations (273 of 3929). The most frequent behavior within this category was walking, with 152 records (55.68%), followed by running (65; 23.81%), jumping (20; 7.33%), and foraging (15; 5.49%). Less frequent actions included arboreal locomotion (19; 6.96%) and move-over (2; 0.73%) (Table 3).
Feeding behaviors represented 1.83% of all recorded observations. The most frequently observed actions were tongue touch (27; 37.50%), followed by lick (16; 22.22%), food-licking (7; 9.72%), mandible lick (7; 9.72%), and drinking (7; 9.72%). Less frequent behaviors included food-bite, chew, food-shaking, and defecation, each recorded twice (2.78%) (Table 3). Feeding events involved the capture of arthropods and the consumption of Hibiscus rosa-sinensis flower petals, as previously documented (Câmara and Jesus, 2025b). Additionally, opportunistic feeding on anthropogenic food sources, such as fruit and biscuit remains, was observed. Drinking behavior was recorded at small artificial ponds and from water droplets accumulated on leaves following irrigation events.
Aggressive and defensive behaviors
Aggressive and defensive behaviors represented 3.92% of all recorded observations, occurring primarily during the summer season, coinciding with periods of heightened social interaction and territoriality. A total of 154 events were assigned to this category, with alert posture (34 observations; 22.08%) being the most frequently recorded behavior. This response was typically triggered by the sudden appearance of tourists or the proximity of other conspecifics, particularly adult males and females.
Although six complete fights were documented (four between adults and juveniles and two between adult males), at least nine additional aggressive interactions were interrupted before escalating. Commonly observed behaviors included threat position (5 observations; 3.25%), gular movement (5; 3.25%), stalk (17; 11.04%), lateral threat orientation (6; 3.90%), NIP (6; 3.90%), and retreat (14; 9.09%) (Table 3).
Gular movements were exclusively observed during threat position displays and were used by both juveniles and adult males. In male-male encounters, biting (3 observations; 1.95%) occasionally led to individuals falling to the ground, often resulting in the involuntary release of the opponent and subsequent escape.
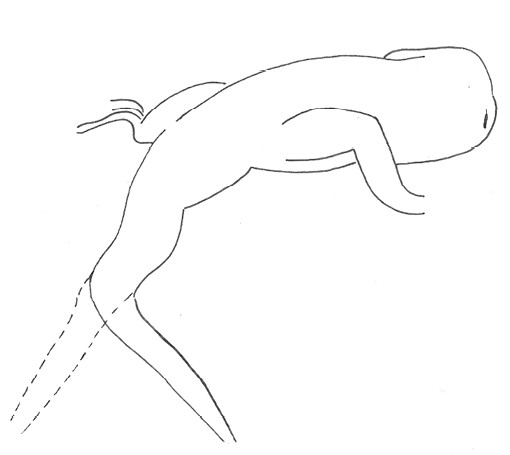
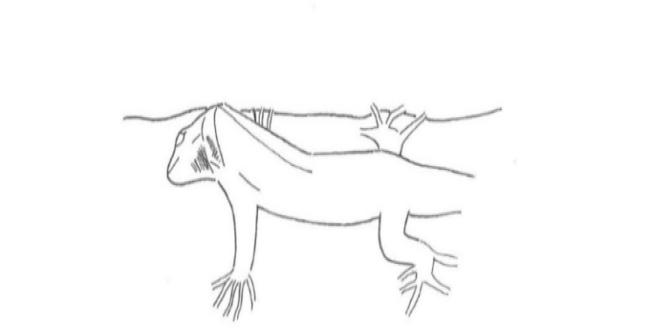
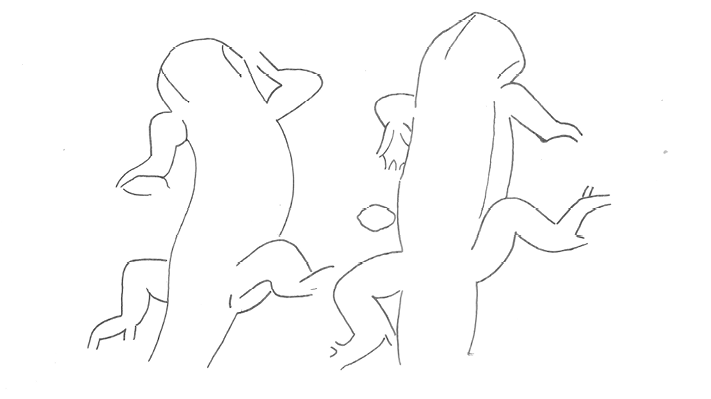
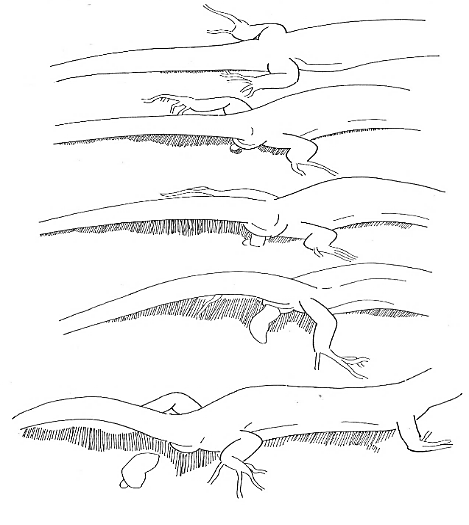
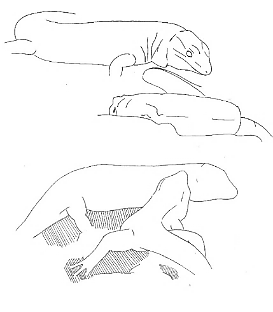
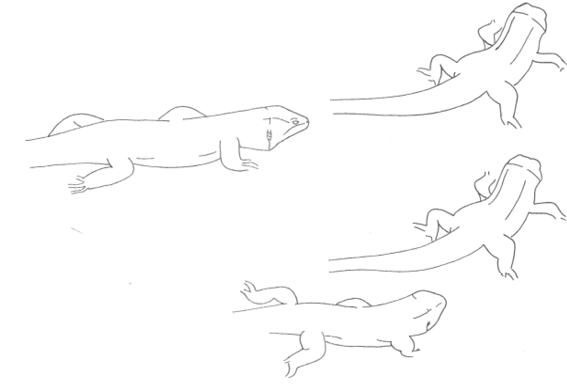
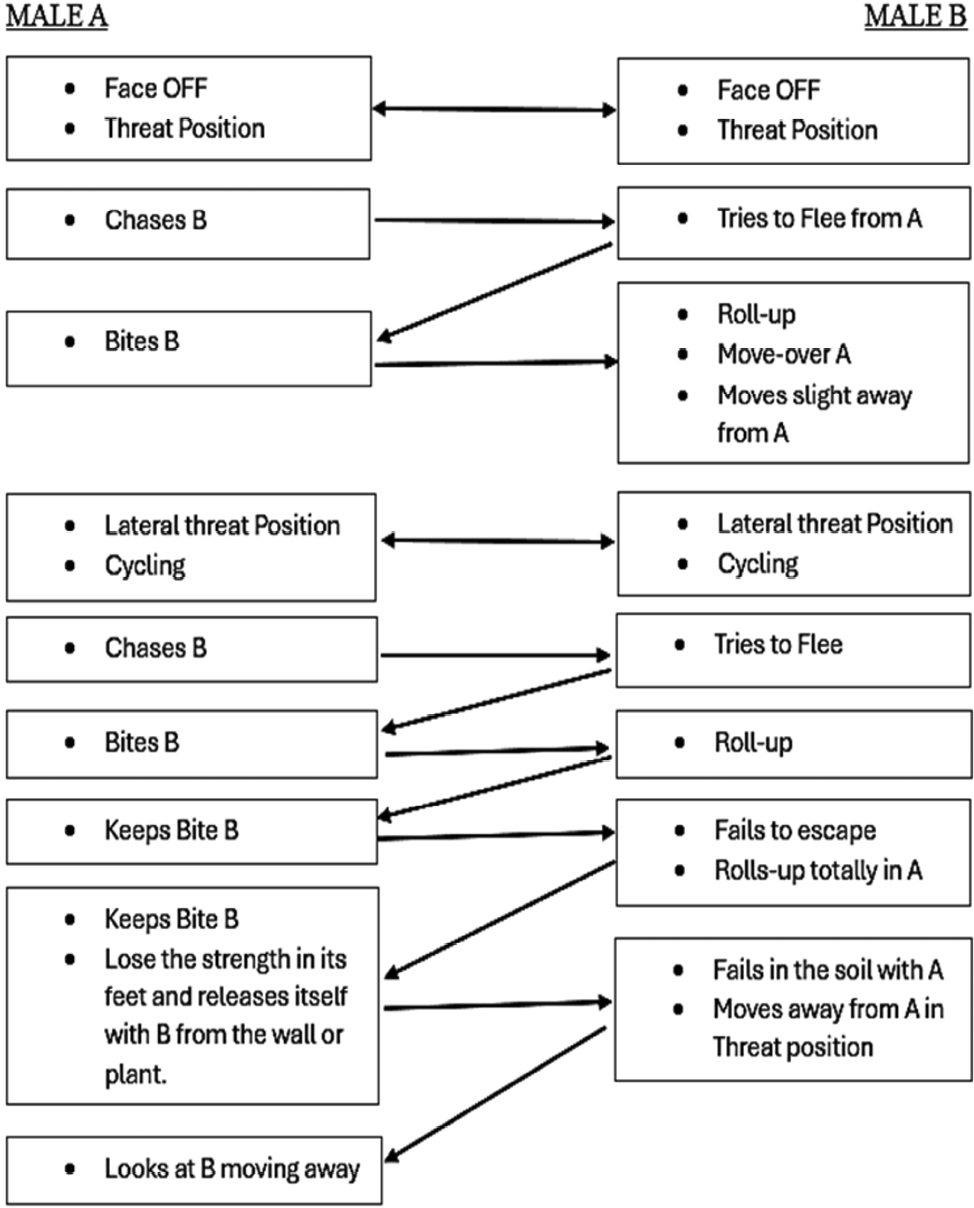
The aggressive repertoire observed in Teira dugesii comprised threat displays, orientation adjustments (lateral and perpendicular threat orientation), body or head movements (circling, lunge, head-touch, face-off), and escape behaviors such as retreat and roll-up. Although diverse, these behaviors were relatively infrequent in the total activity budget. A full male-male confrontation sequence is in Fig. 2 and 3, illustrating the escalation from initial threat to resolution.

Reproductive behaviors were the least frequently observed, comprising only 0.41% of all recorded behaviors. A total of 16 reproductive events were documented, including one complete mating sequence.
Pre-copulatory behaviors were the most common within this category, typically involving the male slowly approaching the female before performing a sudden lunge (Pre-Copulation, 43.75%). During copulation, males exhibited Copulatory Bite (18.75%), a typical behavior in lacertid lizards used to maintain grip on the female, while Female Copulatory Bite (12.5%) was occasionally observed as a response. Only one instance of Copulation itself was recorded (6.25%). Additional behaviors included Release Bite, Terminating Copulation, and Post-Copulatory actions, each observed once (6.25% each) (Table 3).
Maintenance behaviors were by far the most frequently observed, comprising nearly 87% of all recorded behaviors. This pattern aligns with previous studies on lacertid lizards, such as Gallotia galloti (Molina-Borja, 1981), where maintenance behaviors also predominated. Their high frequency likely reflects their essential role in survival, requiring relatively low energetic investment when compared to more demanding behaviors such as aggression, reproduction, or locomotion (Martin and Bateson, 2021).
As ectotherms, lizards such as Teira dugesii do not produce sufficient internal heat through cellular metabolism to regulate their body temperature (Huey, 1982). As a result, they rely heavily on behavioral strategies to gain or dissipate heat, making thermoregulation a central aspect of their daily activity (Huey, 1982). This explains the high frequency of thermoregulatory behavior observed during our study. To maintain their physiological processes within optimal thermal ranges, individuals must adopt postures that either facilitate heat gain or prevent overheating, depending on environmental conditions.
In our study, thermoregulatory postures were classified into two functional categories, following the framework proposed by Greenberg (1977): basking and heat-relief (thermal comfort). Postures associated with basking, such as Body-Down, Head-Down, and Tail-Low; or Body-Down combined with Head-Up and Tail-Low, were frequently observed and serve to maximize solar exposure and heat absorption (Greenberg, 1977; Molina-Borja, 1981). The second category, postures associated with heat stress relief, included Toes-Up, Foot-Up, and occasionally Tail-Up-at-Tip. Greenberg (1977), in his study of Sceloporus cyanogenys, observed similar behaviors at substrate temperatures between 45–50 °C. Carpenter (1961) suggested these postures minimize contact between highly vascularized regions (e.g., digits) and hot surfaces, potentially preventing overheating. In T. dugesii, these postures were often recorded during the warmer hours of the day and were sometimes accompanied by Mouth-Open, which likely aids evaporative cooling. However, Mouth-Open appears to be multifunctional. These findings align with previous reports for other lacertid lizards, such as Gallotia galloti (Molina-Borja, 1981), and underscore the central role of postural and thermoregulatory behaviors in the behavioral ecology of insular reptiles. Brattstrom (1971) distinguished four main contexts in which lizards open their mouth: drinking/eating, yawning, thermoregulation, and aggression. In T. dugesii, this behavior was also observed outside thermal contexts, suggesting similar functional diversity. Self-maintenance behaviors such as Groom, Scratch, Contortion of the body, and Palateo were less frequent but noteworthy.
In addition to postural and maintenance behaviors, we also recorded elementary locomotor movements, particularly in juveniles. Among these, tail movements provided intriguing insights. A lateral tail-waving motion was consistently observed prior to short displacements. Juveniles moved only a few centimeters at a time and frequently displayed this tail movement before proceeding. While the function remains uncertain, it may serve as a risk-assessment behavior or a subtle anti-predator strategy.
In sum, Teira dugesii appears to adopt a predominantly energy-conserving behavioral strategy, heavily focused on maintenance activities. This is consistent with the behavior of other insular lacertids and reflects ecological adaptations to relatively stable environments with predictable thermal conditions.
Mobility and feeding behaviors
Mobility behaviors represented 6.95% of all recorded actions and played a secondary role in the species’ activity budget. These patterns reflect a strategy of cautious movement, likely aimed at minimizing energy expenditure while allowing efficient exploration of the environment.
Walking was predominantly observed on open ground, stone walls, and flat rocky surfaces, suggesting a calm exploratory mode used during thermoregulation or when foraging. In contrast, running and jumping were more frequent in response to disturbances or during social interactions, such as fights or courtship chases. Arboreal movements, often leading to nectar sources in plants like Hibiscus rosa-sinensis, underline the behavioral plasticity of Teira dugesii, which may take advantage of vertical structures and vegetation, particularly in anthropogenic habitats.
Jumping, though less frequent, was particularly notable in lizards navigating between branches or elevated surfaces, indicating a capacity for precise locomotion in three-dimensional space. This flexibility may be especially advantageous in the Madeiran landscape, where vegetation and human structures offer complex microhabitats.
The relative rarity of high-speed or intense locomotor activity supports the interpretation that T. dugesii follows an energy-conserving behavioral strategy, with bursts of mobility mostly linked to specific ecological triggers. These observations are consistent with those in other island lacertids, such as Gallotia galloti, where walking dominates under normal activity conditions and running is usually associated with escape or confrontation (Molina-Borja, 1981). Overall, mobility behaviors in Teira dugesii appear to balance caution, energy efficiency, and ecological opportunity, contributing to its adaptability across diverse habitats.
Many studies have examined the predation of vertebrates and invertebrates by Teira dugesii (Sadek, 1981). In this study, we observed its feeding behavior, water intake, and defecation. Regarding feeding, we recorded the lizard hunting twice and also licking nectar from flowers. During hunting, Teira dugesii moved rapidly toward its prey, minimizing the chance of escape. Once captured, the prey was held in the lizard’s mouth while it performed food-shaking movements. This was followed by chewing before the lizard fled, likely due to the approach of other Teira dugesii individuals, as seen in our recordings.
The second feeding behavior recorded was nectar licking. This behavior has been documented in previous studies (Câmara and Jesus, 2025c) not only in Teira dugesii but also in other lizard species that rely on nectar as part of their diet (Romero-Egea et al., 2023). The lizard engaged in arboreal movement to reach the flower, and upon locating the nectar source, it began licking the nectar.
Regarding water intake, the Madeiran wall lizard uses a tongue-touch movement to drink. An interesting behavior was observed at a pond in the Madeira Botanical Garden, where the lizards approached the water one at a time to drink.
For defecation, Teira dugesii exhibits a behavior that may play a role in communication among individuals. After defecation, the lizards drag their cloaca and pelvic area over the substrate, potentially leaving an odor produced by the femoral pores.
Aggressive and defensive behaviors
The Madeiran wall lizard is known for being highly tolerant to humans (Sarmento, 1948). This is because many island-dwelling lizards have evolved in environments with few predators, leading to longer reaction times (Cooper Jr. et al., 2014). While escape is a common response to threats among lizards, species inhabiting islands often exhibit reduced evasive behavior due to the decreased predation pressure over generations (Goode and Pasachnik, 2018). However, when approached, the lizard often adopts an alert posture in response to stimuli such as noise or sudden movements. In these situations, head movements, including head rotation, were also observed. According to Molina-Borja (1981), alert postures in lizards may serve the function of gathering information from their immediate surroundings. Similarly, Carpenter (1967) described an alert posture in the context of challenge and fight situations.
Throughout the observations and recordings, we documented agonistic behaviors between male-male and male-juvenile interactions. This type of behavior was exclusively recorded during the summer, when temperatures were at their highest. However, as the majority of observations and recordings were conducted in summer, there was a higher likelihood of capturing such interactions. One possible explanation for the reduced frequency of aggressive interactions in Teira dugesii is its high population density. Increased population density often leads to greater competition for resources, which in turn shapes social behavior (MacGregor et al., 2017). In some lacertid species, such as Podarcis hispanica, higher densities have been associated with a greater reliance on non-aggressive dominance displays, minimizing energy expenditure and reducing the risk of injury (López and Martín, 2001).
Male-juvenile fights were recorded on four occasions. Two distinct types of interactions were observed. In the first case, the juvenile performs the gular movement to appear bigger. However, upon the sudden approach of the adult male, the juvenile move-away, avoiding the confrontation. In the second type of interaction, recorded three times, the juvenile was caught by surprise by the adult male. The male bit the juvenile on the dorsal region, prompting the juvenile to struggle by biting back and contorting its body in an attempt to escape. On one occasion, after managing to free itself from the initial bite, the juvenile was immediately grabbed again by the tail, with the adult male holding its weight solely through the strength of its bite. In response, the juvenile curled its body into a C-shape to aid in escaping. In both cases, the juveniles ultimately managed to break free. At times, juveniles are not as fortunate, as they may be killed or have their tails forcibly detached. Instances of adult Teira dugesii preying on juveniles have already been documented (Gloor and Benito, 2023).
Regarding male-male fights, we were able to record an entire combat sequence lasting seven minutes. Other fight initiations consisted only of gular display movements, followed by a chase until the losing male moved away. Another factor contributing to the quick resolution of fights was their occurrence on vertical surfaces. The face-off and threat position may serve as a way for males to display and communicate their dominance or fighting ability by utilizing various physical traits such as body length (Alberts et al., 1992; Rodda, 1992), head shape variation, dewlap color, and throat size. Previous studies with other species have indicated that color variation among males can signal fighting ability as well as aggressiveness (Molnár et al., 2016). The face-off can be of great importance, as the withdrawal of one of the males can prevent direct confrontation, allowing the lizards to conserve energy and avoid injuries (Briffa and Sneddon, 2006). This behavior may serve as a distraction mechanism, diverting the opponent’s attention to the tail rather than more vulnerable body areas. The escalation of these male-male conflicts in Teira dugesii was linked to competition for a high-visibility perch – a prime location frequently occupied by a small number of lizards. In the study by Peters et al. (2016) on Phrynocephalus vlangalii, the intensity of the signal relative to other movements, as well as the observation that males signal more rapidly in the presence of another male, support the hypothesis that tail-lashing represents heightened aggression by the resident and that its use depends on the characteristics of the defended resource.
Both complete and incomplete agonistic sequences occurred in the same locations, which may suggest several ecological and behavioral patterns. One possibility is that these sites represent key resources for Teira dugesii, such as thermoregulation spots, feeding areas, or strategic vantage points for territory surveillance. The recurrence of aggressive interactions in these areas may indicate territorial behavior, where individuals, particularly males, compete for dominance over high-value locations (Cooper and Vitt, 1993; Stamps, 1994). Another potential explanation is the presence of a social hierarchy, where dominant individuals claim and defend prime areas while challenging intruders (Olsson and Madsen, 1995).
In Teira dugesii, several lizards were observed Tongue Flicking their surroundings, and Tongue Touch the substrate. These behaviors related to chemical senses are important in different contexts: foraging (prey discrimination), predator avoidance and social relationships. This last component includes discrimination between familiar and unfamiliar conspecific and self-recognition (Cooper, 1994, Cooper, 1996, Aragón et al., 2001).
The mating behavior of lizards has been studied in various species, including Eremias argus, Podarcis liffordi, Takydromus septentrionalis, Lacerta kulzeri, and Lacerta agilis (Galán, 1997; In den Bosch and Zandee, 2001; Du and Yao, 2007). In the case of Teira dugesii, Richter (1986), Galán and Vicente (2003) and Sleijpen (1996) described several reproductive traits, including clutch size in captivity. However, they did not provide detailed accounts of mating behavior, except for Sleijpen (1996), who noted that males bite the female’s flank during copulation. According to Richter (1986), Madeiran wall lizards typically mate in early April, about 3–4 weeks after emerging from hibernation. This species also exhibits a relatively long reproductive season compared to other lacertids, which usually complete egg-laying by July. Galán (2003) observed that females lay eggs from May to August, at intervals of 5 to 8 weeks. Our observations, however, suggest that mating is not confined to these months. Small juveniles were observed throughout the year, including in January, suggesting that hatching may occur continuously rather than within a narrow seasonal window. Nevertheless, two distinct peaks of reproductive activity were detected during the main breeding season, between May and August.
We observed a complete mating event only once, which lasted approximately two minutes, the same duration reported by Sleijpen (1996) in captivity. On another occasion, copulation was avoided as the female managed to escape. Although these behaviors appeared rare, their true frequency is likely underestimated. In several instances, individuals were observed engaging in courtship or copulation-like behavior behind rocks or flowerpots, partially obstructing the observer’s view. This suggests that some reproductive interactions may have gone undocumented due to limited visibility in dense or structurally complex microhabitats.
Males attempted to initiate copulation multiple times, but the female always managed to escape. Occasionally, the male succeeded in biting her, yet she still fled. Males usually tried to catch the female off guard, taking advantage of moments when she was not alert. Female rejection of mating attempts can be influenced by several factors. One possible explanation is the costly nature of copulation, where a female may gain indirect genetic benefits by rejecting all but the most persistent males (Chapman et al., 2003). Additionally, mate rejection may occur when a female is no longer in her receptive ovulation period or immediately after copulating with another male. Empirical evidence suggests that females may actively resist mating attempts, particularly after having already copulated with a previous mate. For example, in Enyalius perditus, Lima and Sousa (2006) documented a case in which, after courtship, a female rejected a second male following prior copulation. In response, the male attempted to force copulation by biting the female’s nape to restrain her. Despite three persistent attempts, the female exhibited strong resistance by fleeing and counterattacking, biting the male’s mouth. In some cases, copulation efforts can lead to significant harm or even mortality. A notable example is reported in Ctenophorus maculosus, where excessive force during mating resulted in severe injury, as the male’s teeth punctured the female’s spine (Olsson, 1995).
The copulation behavior was the rarest of all, making it extremely difficult to observe. In the study of Galan (1997) with Podarcis bocagei copulation behaviors were also very rare. In the pre copulation behaviors, Teira dugesii, the male observes the female and slowly approaches her. This may allow the male to confirm whether the individual is indeed female, using both visual and chemical cues, as observed by Carpenter and Ferguson (1977). Once he is relatively close, he pauses for a few seconds before suddenly rushing toward her.
Regarding the reproductive behavior of Teira dugesii, it was observed that the male bites the female’s lower body to restrain her, a behavior also seen in the lacertid Eremias argus, before initiating copulation. At the beginning of copulation, females may bite the male in response. After the initial bite, the male grips the female just below her front limbs and then curves his body into a C-shape, aligning his cloaca with hers while wrapping his tail around her. He then inserts his hemipenes into the female’s cloaca. During copulation, the male raises his head and neck, keeping his front end upright while resting his abdomen on the ground. After copulation the male lifts his cloaca and the front part of his tail off the ground, while the back end of the tail remains in contact with the substrate. The hemipenis remains visible outside the cloaca. In some lacertid lizards, females exhibit mate choice based on male morphological traits or behavioral displays (Andersson, 1994). Although T. dugesii males initiate copulation by directly approaching and restraining the female, it remains unclear whether female choice plays a role in mating success. Further studies examining female responses to different male traits could clarify the influence of sexual selection in this species.
This study represents the most detailed behavioral documentation of Teira dugesii to date, offering a broad spectrum of behavioral data that can serve as a baseline for future ecological and ethological studies. Understanding natural behavioral patterns is essential for assessing the impact of habitat modifications, climate change, and human interactions on this endemic species.
Alberts, A.C., Pratt, N.C., Phillips, J.A. (1992): Seasonal productivity of lizard femoral glands: Relationship to social dominance and androgen levels. Physiol. Behav. 51: 729-733.
Andersson, M. (1994): Sexual selection. Princeton, Princeton University Press.
Aragón, P., López, P., Martín, J. (2001): Chemosensory discrimination of familiar and unfamiliar conspecifics by lizards: Implications of field spatial relationships between males. Behav. Ecol. Sociobiol. 50: 128-133.
Báez, M. (1990): Observaciones sobre colorido y diseño de Podarcis dugesii en la isla de Madeira. Vieraea 18: 197-203.
Bateson, M., Martin, P. (2021): Measuring behaviour: An introductory guide. 4th ed. Cambridge, Cambridge University Press.
Bateson, M., Martin, P. (1994): Part 6: Recording methods. In: Measuring behaviour: An introductory guide, pp. 84-100. Cambridge, Cambridge University Press.
Berry, K.H. (1974): Ecology and social behavior of the chuckwalla, Sauromalus obesus obesus Baird. University of California Publications in Zoology, volume 101. University of California Press, Berkeley, California.
Brattstrom, B.H. (1971): Social and thermoregulatory behavior of the Bearded Dragon, Amphibolurus barbatus. Copeia 1971: 484-497.
Briggs, V. (2012): Agonistic behavior of the Mediterranean gecko Hemidactylus turcicus. Zool. Stud. 51: 956-965.
Câmara, J., Jesus, J. (2025a): Teira dugesii (Squamata: Lacertidae) visiting flowers: New observations and a review. Phyllomedusa 24: 105-115.
Câmara, J., Jesus, J. (2025b): A review of the predators of the Madeiran wall lizard (Teira dugesii) with new data. Capt. and Field Herpetol. 7: in press.
Câmara, J., Jesus, J. (2025c): Florivory. Herpetol. Rev. 56: 35-36.
Carlstead, K. (1996): Effects of captivity on the behavior of wild mammals. In: Wild Mammals in Captivity: Principles and Techniques for Zoo Management, pp. 317–333. Kleiman, D.G., Allen, M.E., Thompson, K.V., Lumpkin, S., Eds, Chicago, University of Chicago Press.
Carpenter, C.C. (1961): Temperature relationships of two Oklahoma lizards. Proc. Okla. Acad. Sci. 41: 72-77.
Carpenter, C.C. (1967): Display patterns of the Mexican iguanid lizards of the genus Uma. Herpetologica 23: 285-293.
Carpenter, C.C., Ferguson, G.W. (1977): Variation and evolution of stereotyped behavior in reptiles. In: Biology of the Reptilia, pp. 335-554. Gans, C., Tinkle, D.W., Eds, New York, Academic Press.
Chapman, T., Arnqvist, G., Bangham, J., Rowe, L. (2003): Sexual conflict. Trends Ecol. Evol. 18: 41-47.
Coelho, F.E.A., Bruinj, A.C., Costa, G.C. (2018): Ethogram with the description of a new behavioral display for the striped lava lizard, Tropidurus semitaeniatus. South Am. J. Herpetol. 13: 96-101.
Cooper, W.E., Vitt, L.J. (1993): Female mate choice of large male broad-headed skinks. Anim. Behav. 45: 683-693.
Cooper, W.E. (1994): Chemical discrimination by tongue-flicking in lizards: A review with hypotheses on its origin and its ecological and phylogenetic relationships. J. Chem. Ecol. 20: 439-437.
Cooper, W.E. (1996): Chemosensory recognition of familiar and unfamiliar conspecifics by the scincid lizard Eumeces caticeps. Ethology 102: 454-464.
Cooper, W.E. Jr., Pyron, R.A., Garland, T. Jr. (2014): Island tameness: Living on islands reduces flight initiation distance. Proc. R. Soc. B 281: 20133019.
Den Bosch, H.A.J., Zandee, M. (2001): Courtship behaviour in lacertid lizards: Phylogenetic interpretations of the Lacerta kulzeri complex (Reptilia: Lacertidae). Neth. J. Zool. 51: 263-284.
Du, W.G., Yao, Z.J. (2007): Mating behavior of the northern grass lizard, Takydromus septentrionalis. Chin. J. Zool. 42: 7-12.
Ferreira, A.I., Vasconcelos, D.S., Harris, D.J. (2023): Origins of an introduced Teira dugesii (Squamata: Lacertidae) population in Porto, Portugal. Herpetol. Notes 16: 9-11.
Galán, P. (1997): Reproductive ecology of the lacertid lizard Podarcis bocagei. Ecography 20: 197-209.
Galán, P., Vicente, L. (2003): Reproductive characteristics of the insular lacertid Teira dugesii. Herpetol. J. 13: 149-154.
Gloor, R., Benito, M. (2023): First cases of cannibalism in the Madeiran Wall Lizard, Teira dugesii (Milne-Edwards, 1829). Herpetol. Notes 16: 427-428.
Goode, A.B.C., Pasachnik, S.A. (2018): Flight response and human habituation in an insular endemic lizard. Herpetol. Rev. 49: 215-218.
Greenberg, N. (1977): An ethogram of the blue spiny lizard, Sceloporus cyanogenys (Reptilia, Lacertilia, Iguanidae). J. Herpetol. 11: 177-195.
Huey, R.B. (1982): Temperature, physiology, and the ecology of reptiles. In: Biology of the Reptilia, Vol. 12. Physiology C: Physiological Ecology, pp. 25-91. Gans, C., Pough, F.H., Eds, London, Academic Press.
Jesus, J., Teixeira, S., Teixeira, D., Freitas, T., Russo, D. (2009): Vertebrados terrestres autóctones dos Arquipélagos da Madeira e Selvagens. Funchal, Direcção Regional de Ambiente.
Langkilde, T., Schwarzkopf, L., Alford, R. (2003): An ethogram for adult male rainbow skinks, Carlia jarnoldae. Herpetol. J. 13: 141-148.
Lima, A.F.B., Sousa, B.M. (2006): Court and copulation behaviors of Enyalius perditus Jackson, 1978 (Squamata, Leiosauridae) in captivity conditions. Rev. Bras. Zoociências 8: 193-197.
Liu, Y.X., Wang, J., Shi, H.T., Murphy, R.W., Hong, M.L., He, B., Fong, J.J., Wang, J., Fu, L.R. (2009): Ethogram of Sacalia quadriocellata (Reptilia: Testudines: Geoemydidae) in captivity. J. Herpetol. 43: 318-325.
López, P., Martín, J. (2001): Fighting rules and rival recognition reduce costs of aggression in male lizards, Podarcis hispanica. Behav. Ecol. Sociobiol. 49: 111–116.
MacGregor, H.E., Lewandowsky, R.A., d’Ettorre, P., Leroy, C., Davies, N.W., While, G.M., Uller, T. (2017): Chemical communication, sexual selection, and introgression in wall lizards. Evolution 71: 2327-2343.
Mather, J. (2025): Making an ethogram for octopuses: A personal story. Int. J. Comp. Psychol. 38: 1-8.
Matias, R., Rebelo, R., Granadeiro, J.P., Catry, P. (2009): Predation by Madeiran wall lizards Teira dugesii on Cory’s shearwater Calonectris diomedea hatchlings at Selvagem Grande, North Atlantic. Waterbirds 32: 600-603.
Molina-Borja, M. (1981): Etograma del lagarto de Tenerife, Gallotia galloti galloti (Sauria-Lacertidae). Doñana 8: 43-78.
Molnár, O., Bajer, K., Szövényi, G., Török, J., Herczeg, G. (2016): Space use strategies and nuptial color in European green lizards. Herpetologica 72: 40-46.
Olsson, M. (1995): Territoriality in Lake Eyre dragons Ctenophorus maculosus: Are males “superterritorial”? Ethology 101: 222-227.
Olsson, M., Madsen, T. (1995): Female choice on male quantitative traits in lizards – Why is it so rare? Behav. Ecol. Sociobiol. 36: 179-184.
Petrovan, S. (2023): The wall (and tree) lizard: Surveys and citizen science to improve understanding of arboreal behaviour of Madeiran Wall Lizard Teira dugesii. Herpetol. Notes 16: 791-709.
Poma-Soto, F., Narváez, A.E., Romero-Carvajal, A. (2021): Visual signaling in the semi-fossorial lizard Pholidobolus montium (Gymnophthalmidae). Animals 11: 3022.
Qi, Y., Li, S., Suo, L., Li, H., Wang, Y. (2011): An ethogram of the toad-headed lizard Phrynocephalus vlangalii during the breeding season. Asian Herpetol. Res. 2: 110-116.
Richter, K. (1986): Podarcis dugesii (Milne-Edwards, 1829) - Madeira Mauereidechse. In: Handbuch der Reptilien und Amphibien Europas, Bd. 2, pp. 388-398. Böhme, W., Ed., Wiesbaden, Aula-Verlag.
Rodda, G.H. (1992): The mating behavior of Iguana iguana. Smithson. Contrib. Zool. 534: 1-40.
Romero-Egea, V., Robles, C., Traveset, A., Del Rio, L., Hervías-Parejo, S. (2023): Assessing the role of lizards as potential pollinators of an insular plant community and its intraspecific variation. Animals 13: 1122.
Ruby, D.E., Niblick, H.A. (1994): A behavioral inventory of the desert tortoise: Development of an ethogram. Herpetol. Monogr. 8: 88-102.
Sá-Sousa, P. (1995): The introduced Madeiran lizard, Lacerta (Teira) dugesii in Lisbon. Amphib.-Reptil. 16: 211-214.
Sadek, R.A. (1981): The diet of the Madeiran lizard Lacerta dugesii. Zool. J. Linn. Soc. 73: 313-341.
Santos, L.D., López-Jurado, L.F., Hernández-Peñate, A., Mateo, J.A. (2013): Una nueva población de lagartija (Teira dugesii) en Las Palmas de Gran Canaria. Bol. Asoc. Herpetol. Esp. 24: 102-103.
Sleijpen, F. (1996): Ervaringen met de Madeira-muurhagedis (Podarcis dugesii). Lacerta 54: 179-186.
Stamps, J.A. (1994): Territorial behavior: Testing the assumptions. Adv. Study Behav. 23: 173-232.