Submitted on: 2025, 9th June; revised on: 2025, 4th November; accepted on: 2025, 5th November
Metazoan endoparasites of Erythrolamprus poecilogyrus caesius (Cope, 1862) (Serpentes: Dipsadidae) in South American Chaco: Structural attributes at different organization levels
1 Laboratorio de Biología y Ecología de Helmintos Parásitos. Centro de Ecología Aplicada del Litoral (CECOAL) CONICET-UNNE. Ruta Provincial 5, km 2,5 (3400) - Corrientes, Argentina
2 Laboratorio de Biodiversidad y Conservación de Tetrápodos. Instituto Nacional de Limnología (INALI). CONICET. Ruta Nacional 168 km 0, Paraje “El Pozo” (3000) - Santa Fe, Argentina
3 Laboratorio de Biología de Vectores y Parásitos, Instituto de Zoología y Ecología Tropical (IZET), Facultad de Ciencias. Universidad Central de Venezuela (UCV). P.O. Box 47058, 1041A - Caracas, Venezuela
4 Facultad de Humanidades y Ciencias, Universidad Nacional del Litoral (UNL). Ruta Nacional 168 km 0, Paraje “El Pozo” (3000) - Santa Fe, Argentina
*Corresponding author. Email: cynthyaelizabethgonzalez@gmail.com
Abstract. In this study, the structure of metazoan endoparasites infracommunities and component community in Erythrolamprus poecilogyrus caesius (Cope, 1862) (Serpentes: Dipsadidae) is presented. From 21 specimens (15 females; 6 males) collected between February 2017 and March 2022 at Ingeniero Juárez, Formosa, Argentina, 90.5% of individuals surveyed (n= 19) were infected with at least one species of metazoan endoparasite. Nine hundred sixty specimens were collected (117 adults; 843 larvae), from 11 taxa. Kalicephalus appendiculatus (Nematoda), Raillietiella furcocerca (Pentastomida), Ophiotaenia joanae, Catadiscus uruguayensis, Opisthogonimus lecithonotus (Platyhelminthes) were found as adults. Larvae identified were Oligacanthorhynchus sp. (Acanthocephala), Physaloptera sp. (Nematoda), Strigea spp. and Alaria sp. (Digenea). Digenea was the richest group (S= 6). Adults were collected in intestines, trachea and esophagus, while larvae were found in mesenteries and body cavity. Ophiotaenia joanae was the most prevalent species (38.1%) and K. appendiculatus had the highest mean abundance (3.7). Considering larvae, Strigea spp. were dominant and Oligacanthorhynchus sp. was subdominant. Between pairs of metazoans, positive and negative covariations were obtained. In general, intensity of metazoan taxa had positive correlation to host weight and body condition but had negative relationships with snout-vent length. Mean infracommunity richness (including larvae and adults) was 4.35 ± 1.27 taxa/host. Richness and diversity were higher in larval infracommunities than those composed by adults. This is the first ecological study of endoparasites in snakes from Argentina. New reports to parasite biodiversity in Dry Chaco were added, also, information about habitat use, diet, and position in trophic webs of host was generated.
Keywords. Community, endoparasitism, Erythrolamprus poecilogyrus caesius, metazoan.
Index
Site of study, host collection, and taxonomical ME identification
Description of component community
Description of component community
Description of component community
Parasitism is one of the most diverse life forms on Earth (Poulin and Morand, 2000). Among parasitic organisms, wildlife metazoan endoparasites, and helminths in particular, are often considered a ‘hidden’ and ‘cryptic’ biodiversity by several authors. ‘Hidden’, considering their life histories and the difficulty of note their presence related to their endoparasite living form, despite their important ecological, frequently undetected (Wood and Johnson, 2015). ‘Cryptic’ refers to the challenges in distinguishing them morphologically from other similar taxa within the same taxonomical group, or the necessity of using molecular biology techniques to identified them due to their high mutational rates and genetic diversity (Pérez-Ponce de León and Poulin, 2016; 2018).
Helminths parasites, as one of the most numerous and frequent components of this cryptic biodiversity, are far from being fully understood in certain vertebrate classes, including reptiles (Carlson et al., 2020 a). The decline of their populations, caused by anthropogenic interventions and their environmental consequences, could be one of the major obstacles preventing us from achieving this goal (Carlson et al., 2020 b).
In South America, ‘Gran Chaco’ region represents the second most extensive forest area following the Amazon rainforest (Eva et al., 2004). Sixty-two percent of this region is located in Argentina, representing 23% of the total extension of the country (TNC, 2005). Dry Chaco ecoregion comprise 489,000 km2 of pastures and semi-deciduous forests that includes Chaco and Formosa provinces, two of the provinces with highest snake species richness (De Marzo et al., 2022; Williams and Vera, 2023). Nevertheless, only 9.1% of this surface is under a protected area regime (Nori et al., 2016).
Seasonal precipitation, vegetation, and soil structure of Dry Chaco landscapes allow the formation of temporal, semi-permanent and permanent ponds environments and microhabitats where several vertebrates species, especially herpetofauna, coexists (Lescano et al., 2015). Nevertheless, these landscapes and their endemic fauna are constantly threatened by intentional fires, deforestation, extensive pasture for cattle, and land uses changes. As a result of these activities, the loss of vegetation cover in the Dry Chaco has reached 200,000 hectares per year, highlighting the urgent need to establish conservation zones for snakes, which are highly threatened by these environmental changes and by climate change. A future biotic homogenization is predicted for this region (Andrade-Diaz et al., 2019; Cabral et al., 2024).
Erythrolamprus poecilogyrus (Wied-Neuwied, 1825) (Serpentes: Dipsadidae) is a species distributed in Argentina, Brazil, Bolivia, Paraguay, Peru, Uruguay, and Venezuela (Williams et al., 2021; Williams and Vera, 2023). Erythrolamprus poecilogyrus caesius (Cope, 1862) is one of the three subspecies present in Argentina with a wide distribution in the Argentine Chaco including provinces of Chaco, Corrientes, Formosa, Salta, Santa Fe, Santiago del Estero, and Tucumán (Williams et al., 2021; Williams and Vera, 2023). Quantitative studies on the diet of E. poecilogyrus in Argentina and Paraguay indicate that it is a specialist species with a predominantly batrachophagous diet, although it also consumes reptiles in a smaller proportion (Prieto et al., 2012; Bellini et al., 2015). In Brazil, some dietary analyses have identified additional prey items, including fish and mammals (Corrêa et al., 2016).
Erythrolamprus poecilogyrus caesius inhabits near to cricks, temporal ponds or humid pastures, where it could be seeing aggregated in groups until 10 individuals in different developmental stages together in a same pond, giving it reproductive advantages and protection against predators (Williams et al., 2021; Cabral et al., 2022; Williams and Vera, 2023).
At specific level, E. poecilogyrus is considered as of ‘least concern’ by Nogueira et al. (2020); at subspecific level, E. p. caesius is considered as ‘not threatened’ by Giraudo et al. (2012).
To date, three reports of digeneans and one of pentastomid were made for this species in three Argentinean provinces: Opisthogonimus artigasi Ruiz et Leão 1942 in Misiones (Caubisens Poumarau, 1968), Travtrema stenocotyle (Cohn, 1902) Goodman, 1951 and Catadiscus uruguayensis Freitas et Lent, 1939 both in Buenos Aires (Ostrowski de Núñez, 1979; Lunaschi and Drago, 2002), and Kiricephalus cf. coarctatus in Formosa (Montes et al., 2025).
On the other hand, comprehensive ecological studies in parasite fauna associated to reptiles, and specifically snakes, are non-existent in Argentina (Caraballo et al., 2024).
The aim of this study was to determine, for the first time, the metazoan endoparasites (from now referred as ME) community structure in E. p. caesius across its entire distribution range, based on specimens collected in Formosa province, located in the Argentine Dry Chaco. Additionally, the study explores possible relationships between the ME taxa reported and host biological characteristics.
Site of study, host collection, and taxonomical ME identification
Ingeniero Juárez (23°54′S, 61°51′W) is a locality in Matacos Department of Formosa province, Argentina. It is located in the Dry Chaco ecoregion. In this area the forest is adapted to dry conditions (xerophytic deciduous forest), with predominance of small leaved deciduous and thorny species adapted to fluctuations in water availability, as well as to seasonal thermal variations. Woody vegetation (e.g., Aspidosperma quebracho-blanco, Prosopis ruscifolia and Schinopsis lorentzii) is sparse, and the herbaceous species are predominantly grasses (Gouinia sp., Setaria argentina and Trichloris sp.). The landscape is flat and the climate is featured by low rainfall (mean annual about <700 mm), and high temperatures, at times exceeding 47°C since this area comprises part of the South American Heat Pole (Maldonado et al., 2006). Twenty-one E. p. caesius specimens (15 females; 6 males) (Fig. 1) were collected by direct observation in microhabitats (temporal ponds at both sides of rural ways, surrounded by pastures and native forest patches) between February 2017 and March 2022. Hosts were euthanized using an intracoelomic injection of commercial anesthesia according to size of host (carticaine clorhydrate, 4%; L-adrenaline 1:100.000 units). Bioethical considerations and guidelines for host catching and euthanasia follow Beaupre et al. (2004). Each snake was sexed by direct observation of gonads or eggs in uterus. Specimens were weighted (in g), using an electronic scale, and the snout-vent length (SVL) was measured (in mm). Differences in mean weight and SVL between sexes were evaluated using a Mann-Whitney U test, and normality was assessed with an Anderson-Darling test. Snakes systematics, synonyms and distribution follow Williams et al. (2023). A snout-cloaca vent incision and body organs were isolated and surveyed (trachea, heart, lungs, esophagus, stomach, small and large intestines, kidneys, mesenteries, and coelomic cavity). All ME were fixed in hot formaldehyde 10% v/v and studied with standard techniques for helminths and pentastomids described in Amato and Amato (2010) and Barton and Riley (2004), respectively. Taxonomic identification followed Amin (1987, 2013) for acanthocephalans, Christoffersen and De Assis (2013) for pentastomids, Anderson et al. (2009) for nematodes, Khalil et al. (1994) for cestodes, Jones et al. (2005), Bray et al. (2008), and Fernandes and Kohn (2014) for digeneans. For larval stages of Platyhelminthes, identification followed Hamann et al. (2019, 2022 a). Voucher material from sampled hosts (12 specimens, 10 females, 2 males) were deposited in Colección Herpetológica of Instituto Nacional de Limnología (INALI) (accession numbers: INALI 7391, 7397, 7398, 7402, 7404, 7405, 7406, 7407, 7408, 7413, 7539, 7540). ME were deposited in the Helminthological Collection of Reference of Centro de Ecología Aplicada del Litoral (CECOAL-CONICET); accession numbers are provided in Supplementary Materials, Table S1.

Description of component community
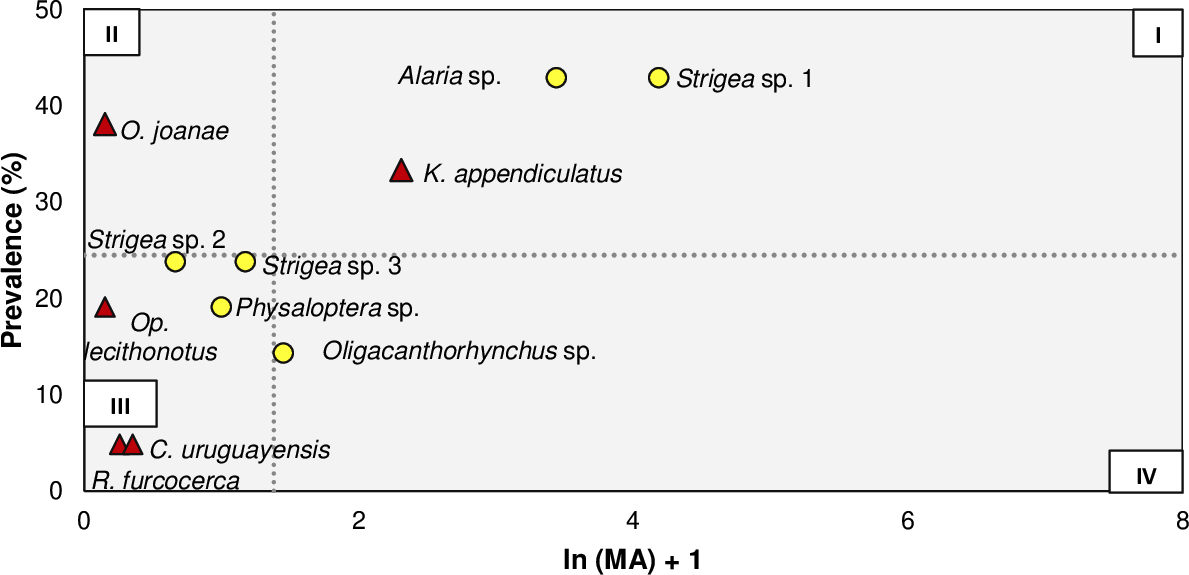
ME taxa were classified in function of their life cycle in geohelminths or biohelminths by consulting specialized literature for each genus (Table S1, Supplementary Materials). Prevalence (P), mean abundance (MA), and mean intensity (MI) were calculated following Bush et al. (1997). Dispersion index was calculated for each taxon through the variance/mean ratio in order to assign every taxon to their correspondent distribution (σ²/X << 1 when a ME taxon followed a normal distribution; σ²/X = 1 when a ME taxon followed a Poisson’s distribution; σ²/X >> 1 when a ME taxon followed a negative binomial distribution). We avoid to presenting standard deviations for ME taxa with negative binomial distribution; following Reiczigel et al. (2024) considerations (presenting standard deviations for mean values is not congruent with non-symmetrical distributions as negative binomial distribution). On the other hand, Bush and Holmes (1986) criterium (B&H), based on prevalence for the dominance of ME (>40% for core species, 10-20% for secondary species, and <10% for satellite species), were compared with the ecological roles assigned to each metazoan taxon. These roles were obtained by three different indices, which are defined and described as follows: (a) Infection index (ii= MA x P/100) with three ecological roles (dominants, subdominants, and accessories) based on logarithmic differences among values obtained by the index for each taxon [Janion (1967) apud Pence and Eason (1980) modified by Guerrero (1996)]; (b) Specific importance index [(SII= P + (MA x 100)] where no categories of importance are established, but, arithmetic differences between scores obtained by each taxon could be compared. We assign the following categories: high, medium and lesser importance (Bursey et al., 2001); (c) Thul’s index (TI) with four categories (dominants, when TI ≥ 1.0; codominants, when 0.01 ≤ TI < 1.0; subordinates, when 0 < I < 0.01, and unsuccessful pioneer, when: TI = 0) (Thul et al., 1985). Ecological roles were represented graphically using Olmstead-Tukey diagrams. These representations are generated using means of P and ln (MA) + 1 of all taxa of ME of the component community. Representation of these in a cartesian plane allows the obtention of four quadrants, with a previous assignation of roles: I for dominant species; II for subdominants or constant species; III for accessory or rare species, and IV for indicator species.
Interspecific relations by site of infection, influence of host biological characteristics and structural attributes
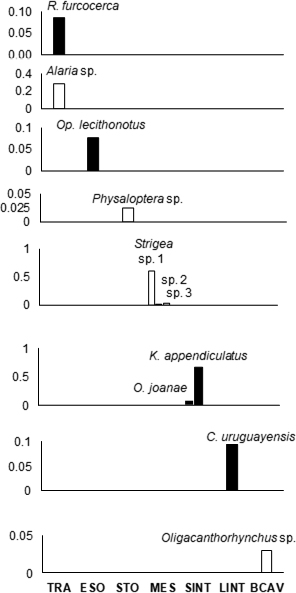
Interspecific relations between pairs of ME taxa or the influence of host biological characteristics on the intensity of each taxon was analyzed in two levels: infrapopulations and component populations. An infrapopulation is a group of ME of the same taxon in an individual host. On the other hand, component population includes all stages of ME from the same species in a host population (Bush et al., 1997). Frequency (F) of each ME taxon component population by site of infection was calculated following Morales and Pino (1987) and plotted by bars graphs. In each pair of taxa collected in the same organ, a Spearman’s rank correlation coefficient was obtained in order to analyze the covariation of their infrapopulation intensities and identify possible interactions among them. The influence of weight and SVL on the intensity of each ME infrapopulations was assessed using Spearman’s rank correlation coefficient (p) between these variables. Hosts body condition was measured through Quetelet’s index of body mass (defined as weight/SVL2 and expressed in kg/m2). Quetelet’s index has been proven on other snakes’ species successfully (Falk et al., 2017). The approach to structural attributes was performed in two levels, infracommunities and component community. An infracommunity includes all the infrapopulations of ME in an individual host. Consequently, component community includes all the component populations of ME in the host population (Bush et al., 1997). Infracommunity mean richness was estimated counting the number of ME taxa in each host (±1 standard deviation). Shannon-Weaver’s diversity index (H’), and Simpson’s dominance index (λ) were obtained at infracommunity and component community levels and compared with the ranges proposed by Margalef (1972) and Magurran (1988): (a) high dominance/low diversity (0 < H’ < 1.35 / 0.01 < λ < 0.33); (b) medium dominance/medium diversity (1.36< H’ < 3.5 / 0.34 < λ < 0.66) or (c) low dominance/high diversity (H’ ≥ 3.6 / λ ≥ 0.67). For measuring the effect of the sample size in the estimation of component community richness and its precision, an accumulation species curve based in Chao-2 index was constructed using the “Mao’s tau” method described in Colwell et al. (2004). All statistical analysis was performed at 95% confidence level using Paleontological Statistics (Past) in its version 4.16c (Hammer et al., 2001).
Description of component community
Eleven ME taxa from three phyla were identified: excluding Kalicephalus appendiculatus, all taxa identified were biohelminths. Erythrolamprus poecilogyrus caesius ME were generalist, parasitizing in larval stages other amphibians and reptiles or for their predators in the adult stage, excepting Kalicephalus and Opisthogonimus genera, which have strict parasitic specificity for snakes as definitive hosts. Detailed information about taxonomical classification of each ME and their intermediate, paratenic or definitive hosts is provided in Supplementary Material (Table S1).
Nine hundred sixty ME specimens were collected; 843 of them were found in larval stages and 117 were in adult stage. Of all host examined, 90.5% (n= 19) were infected at least by one ME. The most prevalent ME species in adult stage was Ophiotaenia joanae, meanwhile K. appendiculatus showed the highest mean abundance (Table 1). In larval stages, Strigea sp. 1 metacercariae and Alaria sp. mesocercariae showed the highest prevalence and mean abundance. With the exception of O. joanae, all ME component populations showed an aggregation pattern and overdispersion in the host population, proper of the negative binomial distribution (Table 1).
| ME taxa | Site of infection | Life cycle/Transmission of helminth | N | P | MA | MI | min-max | σ²/X | Distribution |
|---|---|---|---|---|---|---|---|---|---|
| Adult stage | |||||||||
| Nematoda | |||||||||
| Kalicephalus appendiculatus | SINT | G | 78 | 33.3 | 3.71 | 4.11 | 1–37 | 19.8 | BN |
| Platyhelminthes | |||||||||
| Ophiotaenia joanae | SINT | B | 9 | 38.1 | 0.43 | 0.47 | 1 – 2 | 0.83 | N |
| Catadiscus uruguayensis | LINT | B | 11 | 4.8 | 0.52 | 0.58 | 1 –11 | 11 | BN |
| Opisthogonimus lecithonotus | ESO | B | 9 | 19.1 | 0.43 | 0.47 | 1 – 4 | 3.8 | BN |
| Pentastomida | |||||||||
| Raillietiella furcocerca | TRA | B | 10 | 4.8 | 0.48 | 0.53 | 1 –10 | 10 | BN |
| Larval stage | |||||||||
| Acanthocephala | |||||||||
| Oligacanthorhynchus sp. | BCAV | B | 21 | 23.8 | 1.19 | 1.32 | 2 – 9 | 4.5 | BN |
| Nematoda | |||||||||
| Physaloptera sp. | STO | B | 25 | 19.1 | 1.00 | 1.11 | 1 – 15 | 13.6 | BN |
| Platyhelminthes | |||||||||
| Alaria sp. | BCAV, TRA | B | 241 | 42.9 | 11.48 | 12.68 | 1 – 116 | 64.2 | BN |
| Strigea sp. 1 | MES | B | 508 | 42.9 | 24.19 | 26.74 | 1 – 216 | 164.7 | BN |
| Strigea sp. 2 | MES | B | 15 | 14.3 | 0.71 | 0.79 | 1 – 9 | 6.7 | BN |
| Strigea sp. 3 | MES | B | 33 | 23.8 | 1.57 | 1.74 | 1 – 16 | 12.8 | BN |
Considering only adult ME taxa, K. appendiculatus and O. joanae were not classified in the same ecologic role by the four indices applied. These species presented dominant and subdominant ecologic roles for ii, respectively, but were considered as secondary species by Bush and Holmes (1986) criterium, of medium and lesser importance by the SII and, codominant and subordinate species by TI. Opisthogonimus lecithonotus, C. uruguayensis, and R. furcocerca were considered as less importance ME by all indices compared (Table 2).
| ME taxa | ii Janion (1967) apud Pence and Eason (1980) |
B&H Bush and Holmes (1986) |
SII Bursey et al. (2001) |
TI Thul et al. (1985) |
|---|---|---|---|---|
| Adult stage | ||||
| Nematoda | ||||
| K. appendiculatus | 1.24 (D) | Secondary | 404.73 (M) | 0.07 (C) |
| Platyhelminthes | ||||
| O. joanae | 0.16 (SD) | Secondary | 80.96 (L) | 0.009 (S) |
| C. uruguayensis | 0.03 (A) | Satellite | 52.42 (L) | 0.001 (S) |
| Op. Lecithonotus | 0.07 (A) | Secondary | 42.11 (L) | 0.004 (S) |
| Pentastomida | ||||
| R. furcocerca | 0.02 (A) | Satellite | 52.42 (L) | 0.001 (S) |
| Larval stage | ||||
| Acanthocephala | ||||
| Oligacanthorhynchus sp. | 0.28 (SD) | Secondary | 142.85 (M) | 0 (UP) |
| Nematoda | ||||
| Physaloptera sp. | 0.19 (SD) | Secondary | 119.10 (M) | 0 (UP) |
| Platyhelminthes | ||||
| Alaria sp. | 4.92 (D) | Core | 1190.52 (H) | 0 (UP) |
| Strigea sp. 1 | 10.38 (D) | Core | 2461.95 (H) | 0 (UP) |
| Strigea sp. 2 | 0.17 (SD) | Secondary | 95.23 (L) | 0 (UP) |
| Strigea sp. 3 | 0.22 (SD) | Secondary | 171.44 (M) | 0 (UP) |
According to the TI index, all the larvae were classified as unsuccessful pioneers; no taxa were identified as dominant. All indices excepting TI consider Alaria sp. and Strigea sp. 1 as core/dominant/high importance taxa. Plotting all identified taxa without considering their developmental stage in an Olmstead-Tukey diagram, 3 taxa (1 adult, 2 larval) were dominant, one taxon (O. joanae) was subdominant, 6 taxa (3 adults, 3 larval) were accessory, and Oligacanthorhynchus sp. was an indicator taxon (Fig. 2).
Interspecific relations by site of infection, influence of host biological characteristics and structural attributes
Erythrolamprus poecilogyrus caesius ME were site-specific. Most of the adult ME (4 taxa) were found at higher frequencies in the small intestine, while larval stages (6 taxa) were predominantly found in the mesenteries or free in the body cavity, most frequently associated with the trachea, as Alaria sp. (Fig. 3).
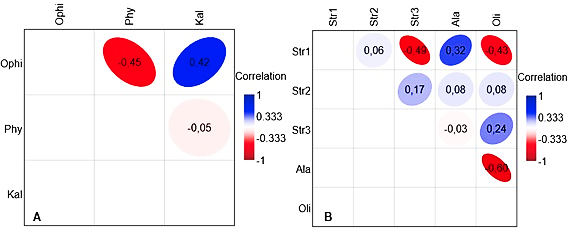
Covariation between abundances of ME parasitizing the same organ did not show a clear pattern, and the correlation coefficients were generally weak and not statistically significant (Fig. 4). Two pairs of larval mesenterial ME (Oligacanthorhynchus sp. – Alaria sp., Strigea sp. 1 – Strigea sp. 3), and two pairs of ME from the digestive system (O. joanae – Physaloptera sp., K. appendiculatus – Physaloptera sp.) had negative correlation coefficients. Contrastingly, in three pairs of larval mesenterial ME (Oligacanthorhynchus sp. – Strigea sp. 3, Strigea sp. 2 – Strigea sp. 3, Alaria sp. – Strigea sp. 1), and one pair of ME from the digestive system (K. appendiculatus – O. joanae), the correlation coefficients were positive.
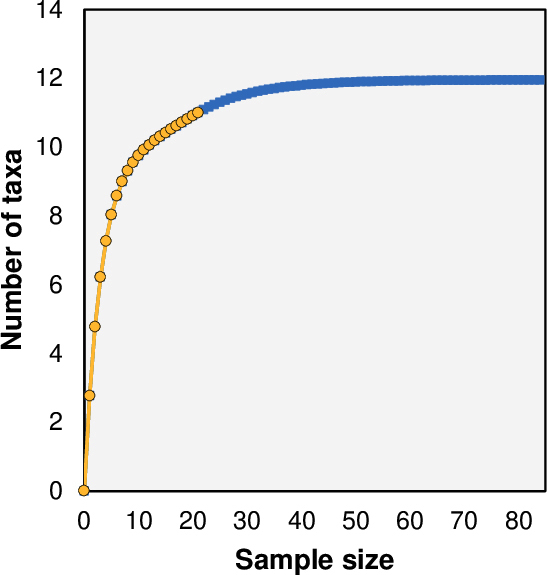
The estimated number of taxa for our sample size was 11 taxa, with an expected richness of 11.95 taxa based on the Chao-2 index (Fig. 5). This estimate reached 92.1% of the potential taxa that could be detected with a large sample size.
The infracommunity mean richness of snakes parasitized by both adults and larvae ME simultaneously was of 4.35 ± 1.27 taxa per host; in contrast, mean richness was higher in snakes infected solely by larval taxa compared to those infected only by adult taxa (Table 3).
| Stage | Infracommunity level | Community level | ||||
|---|---|---|---|---|---|---|
| MS ± SD | H’ ± SD | λ ± SD | S | H’ | λ | |
| Only adults | 1.47 ± 0.83 | 0.15 ± 0.31 | 0.88 ± 0.21 | 5 | 1.10 | 0.45 |
| Only larvae | 2.92 ± 1.26 | 0.43 ± 0.31 | 0.75 ± 0.19 | 6 | 1.06 | 0.47 |
| Adults + larvae | 4.35 ± 1.27 | 0.61 ± 0.41 | 0.66 ± 0.24 | 11 | 1.43 | 0.35 |
Shannon-Weaver’s diversity and Simpson’s dominance indices showed that, their associated ME at infracommunity and component community levels are generally dominated by few species (K. appendiculatus, Alaria sp., and Strigea spp.), resulting in low diversity at this level. Diversity increased slightly and lower dominance were observed at infracommunities and component communities composed by adults and larvae at same time (Table 3).
Snakes showed slight variations in their weight (26.6±11.2) and SVL (43.8±6.9); SVL did not show statistically significant differences among sexes (U= 26.5; p = 0.16). On the other hand, weights show statistically significant differences among sexes (U= 6; p = 0.027). However, given the unbalanced sample – with more females (15) than males (6) – sex was not considered in subsequent analyses. All variables followed a normal distribution (Aweights = 0.55; ASVL = 0.23; pweights = 0.14; pSVL = 0.77).
In general, host weight and the intensity of each ME infrapopulation showed weak positive correlations, with the exception of Oligacanthorhynchus sp., when SVL and intensity of infrapopulations were considered, only O. joanae, and Physaloptera sp. showed negative and weak correlation coefficients. Regarding of Quetelet’s index of body mass vs. intensity, O. joanae and Oligacanthorhynchus sp. showed negative correlations and Oligacanthorhynchus sp. showed statistical significance, while the correlations coefficients for the other taxa were weak (Table 4).
| ME taxa | Weight vs ME intensity (ρ) |
SVL vs ME intensity (ρ) |
Quetelet’s index of body mass vs ME intensity (ρ) |
|---|---|---|---|
| Adult stage | |||
| Nematoda | |||
| K. appendiculatus | 0.48 | -0.18 | 0.41 |
| Platyhelminthes | |||
| O. joanae | -0.32 | -0.24 | -0.25 |
| Larval stage | |||
| Acanthocephala | |||
| Oligacanthorhynchus sp. | -0.51 | -0.45 | -0.78 |
| Nematoda | |||
| Physaloptera sp. | -0.71 | -0.37 | 0.00 |
| Platyhelminthes | |||
| Alaria sp. | 0.45 | 0.048 | 0.62 |
| Strigea sp. 1 | 0.25 | -0.098 | 0.12 |
| Strigea sp. 2 | 0.50 | 1.00 | 0.50 |
| Strigea sp. 3 | 0.45 | -0.22 | 0.45 |
Studies on the ecology of helminths in snakes are extremely scarce in South American portion of Neotropics (Ávila et al., 2013; Mati et al., 2015; Carvalho et al., 2018; Matias et al., 2018, Oliveira et al., 2024). This study represents the first contribution to the knowledge of the ME ecology in snakes in Argentina, focusing on the genus Erythrolamprus.
Despite the lack of ecological studies in Argentina, several taxonomic studies, species distribution reports, new host records, and species listings have been conducted (see Caraballo et al., 2024). A compilation of parasitological studies from Argentina, Brazil, and Uruguay in Erythrolamprus species and subspecies reveal high parasite richness. Notably, acanthocephalans are the only helminth group with reported larval stages (Table S2, Supplementary Materials). With the exception of C. uruguayensis, all ME reported herein are being documented for first time in E. p. caesius as host in Argentina.
Description of component community
The taxonomic composition of ME component community in E. p. caesius could be indicative of semi-aquatic habitat uses of this subspecies, as well as, the amplitude of its diet. Erythrolamprus poecilogyrus has been described as a dietary generalist, preying on a broad range of taxa such as arthropods, fishes, anurans, amphisbaenas, lizards, birds, and rodents (Amaral, 1978; Corrêa et al., 2016; Di Pietro et al., 2020). Nevertheless, studies from Argentina and Paraguay suggest a degree of specialization, with a diet composed mainly by tadpoles, frogs, and toads (Prieto et al., 2012; Bellini et al., 2015; Cabral et al., 2017). In general, published data on trophic ecology studies in E. poecilogyrus did not present identification of snakes surveyed at subspecific level. In this sense, plausible differences in diet among subspecies in its distribution have to be considered, as well as its influence in catalogue them as generalist or specialist.
Despite the developmental stage of the ME reported herein, ecological conditions affect the spatial and temporal overlap of the host and parasite, while species-specific behaviors characteristics can modify these interactions. Adaptations that increase encounter with potential hosts will likely lead to higher infection probabilities (Combes, 2005; Bolek et al., 2024). Several potential routes for host infection were identified, including or not intermediate or paratenic hosts.
In regard of adult ME, Kalicephalus spp. could be considered as a geohelminth. Infections may occur when the host explores soil, introducing infective stages with tongue movement (Schad, 1956; Anderson, 2000). Values of parasitological descriptors of K. appendiculatus could represent parasitological evidence of the terrestrial habitat use in this snake. Nevertheless, the other four taxa were biohelminths with freshwater-associated intermediate hosts, together supporting the semi-aquatic habits of this subspecies.
The life cycle in Ophiotaenia species is not completely elucidated and studies about this topic are scarce (Scholz and De Chambrier, 2003). Two transmission pathways had been suggested: a three-host cycle including copepods as first intermediate hosts, fishes or amphibians as secondary intermediate hosts, and reptiles as definitive hosts, or a two-host cycle with a tissue phase of development in the final host, followed by migration to the intestine, without intermediate or paratenic vertebrate host (Biserkov and Kostadinova, 1997; De Chambrier et al., 2021). The first transmission route is considered more likely for O. joanae, given the semi-aquatic habits of E. p. caesius and its most common prey items (tadpoles and adult anurans).
For Catadiscus and Opisthogonimus, Planorbidae snails are obligatory intermediate hosts in their life cycle. Catadiscus cercariae encyst in vegetation, where it can be ingested by herbivorous tadpoles or during predation activity (Yamaguti, 1975; Hamann and Kehr, 2003). Metacercariae of Opisthogonimus have been found parasitizing the body cavity, muscle, pharyngeal region, and tegument of tadpoles (Hamann and González, 2009), and adult anurans (Hamann et al., 2009). Both amphibians life stages are plausible prey for E. p. caesius. Seasonal conditions in the Dry Chaco may limit water availability and shorter pond duration, limiting resources for anurans and explaining the low prevalence and mean abundances of these digeneans in adult stage.
Raillietiella furcocerca commonly infects lizards, where both nymphs and adults occur, using insect prey as intermediate hosts (Fain, 1966; Melhorn, 2004 apud Fredes and Raffo, 2005). Lizards are occasional prey for E. p. caesius (Andrade et al., 2020) and the low prevalence of this ME suggests they are minor component of its diet.
Parasite ecology studies in snakes from Nearctic and Neotropical do not report frequently the presence of larval helminths (see Detterline et al., 1984; Fontenot and Font, 1994; Jiménez-Ruiz et al., 2002; Ávila et al., 2013; Mati et al., 2015; Carvalho et al., 2018; Matias et al., 2018; Oliveira et al., 2024; Silva-Trejo et al., 2025). Interestingly, 81.87% of ME collected in our study were larval stages of biohelminths.
Oligacanthorhynchus sp. and Physaloptera sp. are biohelminths of broad host range that include arthropods (myriapods and several insect orders) as intermediate hosts. Their ocurrence in E. p. caesius suggests a paratenic role and potential predator-prey interactions with birds or mammals, as possible definitive hosts (Cawthorn and Anderson, 1976; Anderson, 2000; Kennedy, 2006; Richardson, 2006).
Pioneer studies in Argentine snakes reported high number of larval digeneans (mesocercariae and metacercariae) in mesenteries and epiploic folds (Caubisens Poumarau, 1968); in this particular case, from 960 digeneans collected, 797 were larval, indicating high abundance.
Alaria and Strigea include one obligatory intermediate host (Planorbidae), with snakes as secondary intermediate hosts, and reach adult stages in carnivorous mammals or birds of Accipritidae, Strigidae, and Falconidae families (Niewiadomska and Pojmańska, 2011). At Ingeniero Juárez and other localities from Dry Chaco, metacercariae of Strigea species and mesocercariae of Alariinae were found parasitizing anuran in previous research. These were reported as dominant and more abundant taxa in community of Lepidobatrachus llanensis Reig et Cei, 1963 (Hamann et al., 2019, 2022 a, b). We confirm the presence of larval stage of Digenea with this identity in the material collected from this snake species.
Considering the dynamics of microhabitats in Dry Chaco, where host as resource for ME are neither permanent nor abundant, and environmental conditions fluctuate drastically, ME as Strigea and Alaria may maximize their prevalence by enlarging their host range in secondary intermediate or paratenic roles in function of their temporal availability. The accumulation of stages in other suitable hosts, such an anurans or snakes, allows that predation could facilitate potential transmission to definitive hosts (birds for Strigea spp. or mammals for Alaria spp.).
Several Strigea species had been recorded in Formosa province, Argentina, parasitizing Rupornis magnirostris (Gmelin, 1788) (= Buteo magnirostris), Buteogallus meridionalis (Latham, 1790) (Accipritidae), and Caracara plancus (Miller, 1777) (Falconidae) (Lunaschi et al., 2007; Drago and Lunaschi, 2015). In Argentina and Brazil, predation on Erythrolamprus species and other dipsadids by birds of these and other families (de Souza et al., 2021; Fernández-Reinoso et al., 2022) and carnivores as Procyon cancrivorus (Cuvier, 1978) and Cerdocyon thous (Linnaeus, 1766) are reported (Quintela et al., 2014; Castellari-González et al., 2016). Another transmission route is the consumption of rodents that had been reported as prey as well as a paratenic host for Strigea and Alaria species in Holartic (Möhl et al., 2009; Batista et al., 2019). These could support acquisition routes of infective larval stages of these taxa by this snake and reaching maturity in predators in this site of study.
Species serving as intermediate hosts tend to lie in different trophic levels in food web to that for definitive host species. In these positions, they tend to have many different predators, mediating many food chains, acting as focal points of predation interactions involving species across various trophic levels, from higher to lower. Those mechanisms could promote frequent and stable predator-prey interactions, which are essential for the successful completion of parasite life cycles (Chen et al., 2008).
In regard of parasitological descriptors, in the limited studies on ME ecology in semi-aquatic to aquatic snakes in Neotropics and Holartic, it is observed that a single taxon or a few from one phylum often show high prevalence and mean abundances, while other taxa are less abundant or prevalent. In this sense, ME component populations prevalence in studies from Neotropical snakes, prevalence did not exceed 63% (see Jiménez-Ruiz et al., 2002; Ávila et al., 2013; Mati et al., 2015; Carvalho et al., 2018; Matias et al., 2018; Oliveira et al., 2024; Silva-Trejo et al., 2025), while in Holarctic snakes can reach 100% (see Fontenot and Font, 1994; Santoro et al., 2013). In both realms, mean abundances were between 0.02 and 262 parasites/host. Our results are similar to those obtained in other realms in terms of prevalence and mean abundance. Regarding ME proportion of phyla in taxonomic structure, in Neotropical studies, Nematoda taxa show higher richness, prevalence, and mean abundance, while other as Acanthocephala, Pentastomida, and Platyhelminthes (Digenea and Cestoda) appears occasionally with lower richness, prevalence, and mean abundance (Ávila et al., 2013; Mati et al., 2015; Carvalho et al., 2018; Matias et al., 2018; Oliveira et al., 2024). In contrast, North American studies on Akgistrodon Palisot de Beauvois, 1799, Nerodia Baird et Girard, 1853 and Thamnophis Fitzinger, 1843 showed high variability in the number of taxa and parasitological descriptors (Fontenot and Font, 1994; Jiménez-Ruiz et al., 2002; Silva-Trejo et al., 2025). Our results resemble with studies on colubrid snakes from Nearctic and Palearctic realms (Fontenot and Font, 1996; Santoro et al., 2013), where digeneans and larval helminths dominate the communities. However, contrast with finding in Neotropical Dipsadidae were geohelminths and Nematoda are often the most diverse (Table S3, Supplementary Materials)). These differences may reflect spatial and host-related variation in ME prevalence and mean abundances.
Despite their relevance, importance and dispersion indices are rarely included in Nearctic and Neotropical studies on snake parasite ecology (Fontenot and Font, 1994; Jiménez-Ruiz et al., 2002; Ávila et al., 2013; Mati et al., 2015; Carvalho et al., 2018; Matias et al., 2018). Their use can yield key insights into community structure, parasite transmission dynamics, and the selection of appropriate biomarkers for characterizing parasite assemblages (Poulin, 1993; 1999; McVinish and Lester, 2020; Lester and Blomberg, 2021).
Generally, parasite populations follow a negative–binomial distribution which reflects the common pattern of parasite dynamics in natural hosts’ populations (many hosts with few parasites and few hosts with many parasites) (Reiczigel et al., 2024). According to metapopulations theory, this distribution support transmission dynamics by ensuring uninfected hosts serve as available ‘patches’ for parasites recruitment. In our results, only O. joanae showed a regular distribution, likely due to its constant abundance at infrapopulation level, making the mean and variance ratio close to 1.
In our study, the high abundance of larval forms, compared to adult, makes traditional importance indices less effective in assessing the true ecological role of these species. These indices tended to assign similar roles to species that are equally abundant and prevalent, or, in some cases, classify them into categories that exclude them from being considered ‘important’. Therefore, it is crucial to analyze the helminth community considering both mature and immature stages, both simultaneously and separately. Additionally, it is necessary to employ other qualitative and quantitative techniques to prevent the exclusion of species categorized as ‘unsuccessful pioneers’, a label based solely on their maturation stage. This approach is essential, as it considers for the role of paratenic hosts in maintaining the life history of parasites and recognizes the potential of parasites as indicators of possible predators.
Interspecific relations by site of infection, influence of host biological characteristics and structural attributes
ME in adult stage exhibited high specificity for the site of infection. However, a high number of larval taxa, which lower specificity and wide host range, may influence interspecific interactions, aiding their survival until they reach full development in suitable hosts.
Some ME pairs of taxa showed positive correlation. Aho (1990) suggested that helminth communities in amphibians and reptiles tend to be isolationist and non-interactive. Nevertheless, recent studies on fish and mammal parasite communities supports that facilitation among parasite species is more common than negative interactions. Positive interactions contribute more significantly to increasing communities’ richness (Dallas et al., 2019; Rodgers and Bolnick, 2024). The mechanisms are unclear, but may involve a pioneer species weakening the host’s immune system, allowing secondary species to colonize. However, considering the indirect approach in testing these associations (Poulin, 2001), we suggest that a larger sample size could be needed to provide more statistically robust results.
Parasite community studies in aquatic snake in Nearctic and Palearctic region reported 11 to 13 taxa (Fontenot and Font, 1994; Santoro et al., 2013), similar to our finding, though, only Santoro et al. (2013) included larval stages. In contrast, the richness reported for Neotropical snake appears lower, which may be related to the absence or lack of reporting of larval stages.
Excluding larval stages underestimates infracommunity richness. Infracommunities that include larval stages, either exclusively or combined with adult, show greater richness than those composed only of adult. Larval and larval-adult communities show slightly higher diversity and dominance than adult-only communities. A similar pattern was observed by Santoro et al. (2013) in the habitat generalist snake Hierophis viridiflavus carbonarius (Bonaparte, 1833) (Colubridae) in southern Italy. The low diversity and dominance indices observed in our results are consistent with research on snakes with different habitat uses (Fontenot and Font, 1994; Jiménez-Ruiz et al., 2002; Santoro et al., 2013).
Weight and SVL are variables linked to host characteristics as body condition and age (Poulin, 1997). The most accepted hypothesis suggests that larger hosts may harbor more parasites due to their greater size which provides more resources and space. Additionally, age may influence parasite richness or abundance due to longer exposure to infections or changes in feeding preferences (Poulin, 1997). Studies on host size or mass as predictors of parasite richness or abundance in snakes are scarce (Lettoof et al., 2019; Oliveira et al., 2024), and no general pattern has been established. For example, while SVL predicts higher richness in Neotropical amphibians (Campião et al., 2015), no such relationship was found in lizards (Leung and Koprivnikar, 2019).
In snakes, a particularly relevant issue is the possibility that food availability early in life can have a disproportionate impact on later growth patterns and, ultimately, on maximum adult body size. Such an effect could generate substantial size variation among cohorts, even in the absence of genetic differences (Madsen and Shine, 2000; Brown et al., 2017).
Minor SVL and weight variation among specimens suggest they belong to the same cohort, consistent with Cabral et al. (2022), who observed newborn groups for this subspecies. These individuals were likely born in the pond and stayed nearby, using its water, shelter, and amphibian prey as a food source. Juveniles exhibit opportunistic feeding behavior – although not yet specifically reported for this subspecies – that could be associated with site fidelity, efficient use of resources, and the absence of alternative water bodies in the area (Cabral et al., 2017, 2022).
This may explain the observed pattern: snakes with greater body mass showed higher ME intensities, whereas those with longer SVL had lower ones. In this sense, we suggest that relationships between body weight or SVL in with snakes’ age, as well as, associations of these host features with the intensity of some ME cannot be analyzed isolated, or as response of a univariate phenomenon.
In addition, reptiles have a strong innate immune response, a moderate adaptative component with a slower activation, no lymphatic nodes, and seasonal variations in T and B cell activity influenced by temperature (Rios and Zimmerman, 2015). We propose that unconsidered effects on the innate and adaptive immune system during host development may differentially affect the recruitment of ME taxa and the increase of its intensity over time.
Our results suggest that some in hosts with reduced body conditions (and possibly depressed immune system) could facilitate the colonization of some ME species could be facilitated over time. This pattern could not be necessarily only related to host age but could also occur before mating and reproduction seasons, hydric stress, prey availability and host characteristics do not hinder successful ME recruitment.
A non-significant positive correlation between larval ME intensity and host condition suggests that certain snake sizes may be more vulnerable to predation Predators could take advance of the temporal aggregation behavior reported in this species as prey, and consequently, this could increase the likelihood of ME reaching their definitive hosts.
Oligacanthorhynchus sp., was the only larval species showing a significant negative correlation, both with host condition and with other helminths sharing the mesenterial–body cavity site. In this sense, both experimental and meta-analytical approaches show that cystacanth infection has no effect on intermediate host size (Benesh and Tellervo Valtonen, 2007; Fayard et al., 2020). Even, a positive relationship is observed between helminth larvae (cystacanths and metacercariae) and intermediate host size in natural marine environments (Poulin et al., 2003). These patterns contrast with our results; cystacanths and other larval helminths sharing intermediate hosts had positive correlations among them, along with negative correlations with serotonin concentrations. This have been suggested as evidence of host manipulation and the removal of heavily infected hosts by predation (Poulin et al., 2003). However, this also contrasts with our results.
This could suggest that in this study area, larval stages could not present refined mechanisms of host exploitation, possibly due to a recent relation between hosts and parasites, in evolutionary terms. Warburton (2020) consider drylands as ‘natural laboratories’, where hypothesis on virulence evolution, host spectra, and life cycle strategies could be tested, because extreme conditions and low population density represent examples within extremes in understand parasite communities diversity, structure and the transmission dynamic.
Studies in amphibians and reptiles as intermediate or paratenic hosts in these environments are scarce. In this sense, our comparison with previous research has to be as conservative as possible. At the same time, we suggest the design of more field experimental studies in this area in order to tap the potential of these environments, unraveling the challenges and knowledge gaps in the subjacent mechanisms of these parasite-host relationships.
This study provides new insights into the parasitic fauna of snakes, contributing valuable data on the ecology of ME infracommunities and component community of E. p. caesius, as well as their relationship with the host’s biological characteristics. Its helps fill the research gap in area, specifically within the Dipsadidae family in Neotropics. However, most Neotropical research remains largely descriptive, unlike the more quantitative Holarctic studies.
The sampling effort and sample size, combining scarce field material with museum specimens from different origins and times periods, have provide a valuable approach to parasite communities in areas where knowledge is still limited, as Neotropical zones (Carlson et al., 2020). Nevertheless, these methods have limitations that need to be addressed. The underestimation of certain factors may exclude measurable aspects that must be considered. It is important to account for the heterogeneity of each study area, including environmental conditions and intraspecific host differences.
Evidence has shown that ME in communities associated to amphibians and reptiles belong to generalist species with a broad host range and low specificity. Considering this, studies on the parasitic fauna of amphibian and reptiles species provide valuable insights into the parasitic communities of related species with similar habitat use and trophic ecology. This could be especially relevant for species that are more vulnerable or have restricted distributions due to population declines, often caused by environmental degradation, such as the one the American Chaco has been enduring for decades (Nori et al., 2016).
We want to thank deeply to Andres Ulibarrie for provide us photographs of the snake host for illustrate this work and, to Dr. Franco Rosso (Dirección de Recursos Naturales y Gestión – Ministerio de la Producción y Ambiente, Provincia de Formosa, Argentina), for authorizing the capture of the snakes sampled in this study (Dispositions 108/21 and 091/22). We acknowledge an anonymous reviewer whose comments contributed to improving the manuscript significantly.
Financial support was received from Universidad Nacional del Nordeste (Grant number PI 21Q001 to C.E.G.), from CONICET (PIP 11220200101582CO to C.E.G.), from Universidad Nacional del Litoral (CAID- UNL 2020), and from Agencia Nacional de Promoción de la Investigación, el Desarrollo Tecnológico y la Innovación (PICT-2020-SERIEA-01341).
Supplementary material associated with this article can be found at: https://oaj.fupress.net/index.php/ah/article/view/17983/14574.
Aho, J. (1990): Helminth communities of amphibians and reptiles: comparative approaches to understanding patterns and processes. In: Parasite communities: patterns and processes, pp. 157-195. Esch, G., Bush, A., Aho, J., Eds, London, Chapman and Hall.
Amaral, A. (1978): Serpentes do Brasil: Iconografia Colorida. São Paulo. Editora da Universidade de São Paulo.
Amato, J.F.R., Amato, S.B. (2010): Técnicas gerais para coleta e preparação de helmintos endoparasitos de aves. In: Ornitologia e conservação: ciência aplicada, técnicas de pesquisa e levantamento, pp. 367-394. Von Matter, S., Costa Straube, F., Almeida Accordi, I., Queiroz Piacentini, V., Cândido Jr., J.F., Eds, Rio de Janeiro, Technical Books Editora.
Amin, O. (1987): Key to the families and subfamilies of Acanthocephala, with the erection of a new class (Polyacanthocephala) and a new order (Polyacanthorhynchida). J. Parasitol. 73: 1216–1219.
Amin, O. (2013): Classification of Acanthocephala. Folia Parasitol. (Praha). 60: 273-305.
Anderson, R.C., Chabaud, A.G., Willmontt, S. (2009): Keys to the Nematode parasites of vertebrates. Archival Volume, Wallingford. CAB International.
Anderson, R.C. (2000): Nematode parasites of vertebrates: Their development and transmission. Wallingford, CAB International.
Andrade, H., da Costa, S.M., dos Santos, M.A., dos Reis Dias, E.J. (2020): Diet review of Erythrolamprus poecilogyrus (Wied-Neuwied, 1825) (Serpentes: Dipsadidae), and first record of Dermatonotus muelleri (Boettger, 1885) (Anura: Microhylidae) as a prey item in Sergipe State, northeastern Brazil. Herpetol. Notes 13: 1065-1068.
Andrade-Díaz, M.S., Sarquis, J.A., Loiselle, B.A., Giraudo, A.R., Díaz-Gómez, J.M. (2019): Expansion of the agricultural frontier in the largest South American Dry Forest: Identifying priority conservation areas for snakes before everything is lost. PLoS One 14: e0221901.
Ávila, R., Morais, D., Anjos, L., Almeida, W., Silva, R. (2013): Endoparasites infecting the semiaquatic coral snake Micrurus surinamensis (Squamata: Elapidae) in the Southern Amazonian region, Mato Grosso state, Brazil. Braz. J. Biol. 73: 645-647.
Barton, D., Riley, J. (2004) Raillietiella indica (Pentastomida) from the lungs of the giant toad, Bufo marinus (Amphibia), in Hawaii, U.S.A. Comp. Parasitol. 71: 251-254.
Batista, S.F., Gomes Facure, K., Marques, O.A.V. (2019): First record of mammal consumption by Erythrolamprus miliaris. How wide is the diet of this water snake? Herp. Notes 12: 713-715.
Beaupre, SJ., Jacobson, ER., Lillywhite, HB., Zamudio, K. (2004): Guidelines for use of live amphibians and reptiles in field and laboratory research. Herpetological Animal Care and Use Committee (HACC). American Society of Ichthyologists and Herpetologists.
Bellini, G.P., Giraudo, A.R., Arzamendia, V., Etchepare, E.G. (2015): Temperate snake community in South America: is diet determined by phylogeny or ecology? PLoS One 10: e0123237.
Benesh, D.P., Tellervo Valtonen, E. (2007): Effects of Acanthocephalus lucii (Acanthocephala) on intermediate host survival and growth: implications for exploitation strategies. J. Parasitol. 93: 735-741.
Biserkov, V., Kostadinova, A. (1997): Development of the pleroceroid I of Ophiotaenia europaea in reptiles. Int. J. Parasitol. 27: 1513-1516.
Bolek, M., Gustafson, K.D., Langford, G.J. (2024): Parasites in relation to other organisms: Hosts, reservoirs, and vectors. In: Concepts in parasitology, pp. 40-46. Gardner, S.L., Gardner, S.A., Eds, Lincoln Nebraska, Zea Books.
Bray, R.A., Gibson, D.I., Jones, A. (2008): Keys to the Trematoda. Volume 3. Wallingford. CAB International.
Brown, G.P., Madsen, T.R.L., Shine, R. (2017): Resource availability and sexual size dimorphism: differential effects of prey abundance on the growth rates of tropical snakes. Funct. Ecol. 31: 1592-1599.
Bursey, C.R., Goldberg, S.R., Parmalee, J.R. (2001): Gastrointestinal helminths of 51 species of anurans from Reserva Cuzco Amazónico, Peru. Comp. Parasitol. 68: 21-35.
Bush, A., Holmes, J. (1986): Intestinal helminths of lesser scaup ducks: Patterns of association. Can. J. Zool. 64: 132-141.
Bush, A.O., Lafferty, K.D., Lotz, J.M., Shostak, A.W. (1997): Parasitology meets ecology on its own terms: Margolis et al. revisited. J. Parasitol. 83: 575–583.
Cabral, H., Bueno-Villafañe, D., Romero-Nardelli, L. (2017): Comments on the diet of juvenile Erythrolamprus poecilogyrus caesius (Serpentes: Dipsadidae) in the Paraguayan Chaco. Phyllomedusa 16: 299-302.
Cabral, H., Carrillo, J.F.C., Bejarano Coronel, D., Caballero Gini, A.N., Santana, D.J. (2022): Report of aggregation behavior in Erythrolamprus poecilogyrus caesius (Cope, 1862) (Serpentes: Dipsadidade) in the Dry Chaco. Rev. latinoam. Herpetol. 5: 25-30.
Cabral, H., Piatti, L., Santana, D. (2024): Impacts of climate change in taxonomic, phylogenetic and functional diversity in snakes in largest dry forest ecoregion, the Gran Chaco. J. Arid Environ. 224: 105214.
Campião, K.M., Ribas, A.C.A, Morais, D.H., Silva, R.J., Tavares, L.E.R. (2015): How many parasites species a frog might have? Determinants of parasite diversity in South American anurans. PLoS One 10: e0140577.
Caraballo, J., Ulibarrie, A., Hamann, M., Guerrero, R., Arzamendia, V., González, C. (2024): Metazoan endoparasites of snakes from Argentina: review and checklist with distributional notes and remarks. J. Helminthol. 98: 1-19.
Carlson, C.J., Dallas, T.A., Alexander L.W., Phelan, A.L., Phillips, A.J. (2020a): What would it take to describe the global diversity of parasites? Proc. R. Soc. B. 287: 20201841.
Carlson, C.J., Hopkins, S., Bell, K.C., Doña, J., Godfrey, S.S., Mackenzie, L.K., Lafferty, K.D., Moir, M.L., Speer, K.A., Strona, G., Torchin, M., and Wood, C.L. (2020b): A global parasite conservation plan. Biol. Conserv. 250: 108596.
Carvalho, E.F.F., Silva-Neta, A.F.D., Silva, C.S., Oliveira, C.R., Nunes, J.D.C.X., Souza, T.G., Ávila, R.W. (2018): Helminths infecting the cat-eyed snake Leptodeira annulata Linnaeus 1758 (Squamata: Dipsadidae) in a semiarid region of Brazil. Helminthologia 55: 281-285.
Castellari-González, R., Marcial de Castro, T., Silva-Soares, T. (2016): Predation of the water snake Erythrolamprus miliaris (Serpentes: Dipsadidae) by the crab-eating fox Cerdocyon thous (Carnivora: Canidae). Bol. Mus. Biol. Mello Leitão, Nova Sér. 38: 315-323.
Caubisens Poumarau, E.M. (1968): Trematodes de ofídios de la Argentina. Rev. Mus. Argent. 1: 1-129.
Cawthorn, R.J., Anderson, R.C. (1976): Development of Physaloptera maxillaris (Nematoda: Physalopteroidea) in skunk (Mephitis mephitis) and the role of paratenic and other hosts in its life cycle. Can. J. Zool. 54: 313-323.
Chen, H.W., Liu, W.C., Davis, A.J., Jordán, F., Hwang, M.J., Shao, K.T. (2008): Network position of hosts in food webs and their parasite diversity. Oikos 117: 1847-1855.
Christoffersen, M.L., De Assis, J.E. (2013): A systematic monograph of the Recent Pentastomida, with a compilation of their hosts. Zool. Meded. 87: 1–206.
Colwell, R.K., Mao, C.X., Chang, J. (2004): Interpolating, extrapolating, and comparing incidence-based species accumulation curves. Ecology. 85: 2717-2727.
Combes, C. (2005): The art of being a parasite. Chicago, University of Chicago Press.
Corrêa, D.N., Quintela, F.M., Loebmann, D. (2016): Feeding ecology of Erythrolamprus jaegeri jaegeri (Günter, 1858) and Erythrolamprus poecilogyrus sublineatus (Cope, 1860) in the coastal zone of Subtropical Brazil (Serpentes, Dipsadidae). An. Acad. Bras. Ciênc. 88: 293-308.
Dallas, T.A., Laine, A.L., Ovaskainen, O. (2019): Detecting parasite associations within multi-species host and parasite communities. Proc. R. Soc. B. 286: 20191109.
De Chambrier, A., Alves, P.V., Schuster, R.K., Scholz, T. (2021): Ophiotaenia echidis n. sp. (Cestoda: Proteocephalidae) from the saw-scaled viper, Echis carinatus sochureki Stemmler (Ophidia: Viperidae), one of the world’s deadliest snakes, from the United Arab Emirates. Int. J. Parasitol. Parasites Wildl. 14: 341-354.
De Marzo, T., Gasparri, N.I., Lambin, E.F., Kuemmerle, T. (2022): Agents of forest disturbance in the Argentine Dry Chaco. Remote Sens. 14: 1-19.
De Souza, E., Lima-Santos, J., Entiauspe-Neto, O.M., dos Santos, M.M., de Moura, P.R., Hingst-Zaher, E. (2022): Ophiophagy in Brazilian birds: a contribution from a collaborative platform of citizen science. Ornithol. Res. 30: 15-24.
Detterline, J.L., Jacob, J.S., Wilhelm, W.E. (1984): A comparison of helminth endoparasites in the cottonmouth (Agkistrodon piscivorus) and three species of water snakes (Nerodia). Trans. Am. Micros. Soc. 103: 137-143.
Di Pietro, D.O., Williams, J.D., Cabrera, M.D., Alcalde, L., Cajade, R., Kacoliris, F.P. (2020): Resource partitioning in a snake assemblage from east central Argentina. An. Acad. Bras. Ciênc. 92: 1-22.
Drago, F.B., Lunaschi, L. (2015): Update of checklist of digenean parasites of wild birds from Argentina, with comments about the extent of their inventory. Neotrop. Helminthol. 9: 325-350.
Eva, H.D., Belward, A.S., De Miranda, E.E., Di Bella, C.M., Gond, V., Huber, O., Jones, S., Sgrenzaroli, M., Fritz, S. (2004): A land cover map of South America. Glob. Chang. Biol. 10: 731-744.
Fain, A. (1966): Pentastomida of snakes. Their parasitological role in man and animals. Mem. Inst. Butantan 33: 167-174.
Falk, B.G., Snow, R.W., Reed R.N. (2017): A validation of 11 body-condition indices in a giant snake species that exhibits positive allometry. PLoS One 12: e0180791.
Fayard, M., Dechaume-Moncharmont, F.X., Wattier, R., Perrot-Minnot, M.J. (2020): Magnitude and direction of parasite-induced phenotypic alterations: a meta-analysis in acanthocephalans. Biol. Rev. 95: 1233-1251.
Fernández-Reinoso, R., Acosta, J.C., Acosta, R., Corrales, L.A. (2022): Primer registro de depredación de Erythrolamprus sagittifer (Squamata: Dipsadidae) por Geranoaetus albicaudatus (Accipitriformes: Accipitridae) en la Provincia de Neuquén, Argentina. Rev. latinoam. herpetol. 5: e544.
Fredes, F., Raffo, E. (2005): Hallazgo de Raillietiella sp. en culebra chilena de cola larga (Philodryas chamissonis) de un zoológico de la región metropolitana. Parasitol. latinoam. 60: 189-191.
Fontenot, L.W., Font, W. (1996): Helminth parasites of four species of aquatic snakes from two habitats in southeastern Louisiana. Proc Helminthol Soc Wash. 63: 66-75.
Giraudo, A.R., Arzamendia, V., Bellini, G.P., Bessa, C.A., Calamante, C.C., Cardozo, G., Chiaraviglio, M., Costanzo, M.B., Etchepare, E.G., Di Cola, V., Di Pietro, D.O., Kretzschmar, S., Palomas, S., Nenda, S.J., Rivera, P.C., Rodríguez, M.E., Scrocchi, G.J., Williams, J.D. (2012): Categorización del estado de conservación de las Serpientes de la República Argentina. Cuad. herpetol. 26: 303-326.
Guerrero, R. (1996): Streblidae (Diptera: Pupipara) parásitos de los murciélagos de Pakitza, Parque Nacional Manu, Perú. In: Manu, la biodiversidad del sureste del Perú, pp. 627-641, Wilson, D., Sandoval, A., Eds, Washington DC, Smithsonian Institution.
Hamann, M.I., Kehr, A.I. (2003): Ecological aspects of parasitism in the tadpole of Pseudis paradoxa from Argentina. Herpetol. Rev. 34: 336-341.
Hamann, M.I., Kehr, A.I., González, C.E., Duré, M.I., Schaefer, E.F. (2009): Parasite and reproductive features of Scinax nasicus (Anura: Hylidae) from a South American subtropical area. Interciencia 34: 214-218.
Hamann, M.I., González, C.E. (2009): Larval digenetic trematodes in tadpoles of six amphibian species from northeastern Argentina. J. Parasitol. 95: 623-628.
Hamann, M.I., Fernández, M.V., González, C.E. (2019): Metacercariae of Strigeidae parasitizing amphibians of the Chaco region in Argentina. An. Acad. Bras. Ciênc. 91: e20180044.
Hamann, M.I., Fernández, M.V., González, C.E. (2022 a): Larval digenean parasitizing amphibian hosts from the Argentinian Chaco region. Acta Parasitol. 68: 194-207.
Hamann, M.I., González, C.E., Duré, M.I., Palomas, Y.S. (2022 b): Helminth community in the Llanos frog, Lepidobatrachus llanensis (Ceratophryidae), from the Dry Chaco. South Am J Herpetol. 25: 12-17.
Hammer, Ø., Harper, D.A.T., Ryan, P.D. (2001): PAST: Paleontological statistics software package for education and data analysis. Palaeontol electron. 4: 1-9.
Jiménez-Ruiz, FA, García-Prieto L, Pérez-Ponce de León, G. (2002): Helminth infracommunity structure of the sympatric garter snakes Thamnophis eques and Thamnophis melanogaster from the Mesa Central of Mexico. J. Parasitol. 88: 454-460.
Jones, A., Bray, R.A., Gibson, D.I. (2005): Keys to the Trematoda. Volume 2. Wallingford, CAB International.
Kennedy, C.R. (2006): Ecology of the Acanthocephala. Cambridge, Cambridge University Press.
Khalil, L., Jones, A., Bray, R. (1994): Key to the cestode parasites of vertebrates. Wallingford, CAB International.
Lescano, J.N., Bellis, L.M., Hoyos, L.E., Leynaud, G.C. (2015): Amphibian assemblages in dry forests: multi-scale variables explain variations in species richness. Acta Oecol. 65–66: 41-50.
Lester, R.J.G., Blomberg, S.P. (2021): Three methods to measure parasite aggregation using examples from Australian fish parasites. Methods Ecol Evol. 12: 1999-2007.
Lettoof, D., von Takach, B., Bateman, P.W., Gagnon, M.M., Aubret F. (2019): Investigating the role of urbanisation, wetlands and climatic conditions in nematode parasitism in a large Australian elapid snake. Int. J. Parasitol. Parasites Wildl. 3: 32-39.
Leung, T.L.F., Koprivnikar, J. (2019): Your infections are what you eat: How host ecology shapes the helminth parasite communities of lizards. J. Anim. Ecol. 88: 416-426.
Lunaschi, L.I., Drago, F.B. (2002): Primer registro de Catadiscus uruguayensis Freitas y Lent, 1939 (Digenea: Diplodiscidae) como parásito de reptiles. Neotropica 48: 65-67.
Lunaschi, L.I., Cremonte, F., Drago, F.B. (2007): Checklist of digenean parasites of birds from Argentina. Zootaxa 1403: 1-36.
Madsen, T., Shine, R. (2000): Silver spoons and snake body sizes: prey availability early in life influences long-term growth rates of free-ranging pythons. J. An. Ecol. 69: 952-958.
Magurran, A.L. (1988): Ecological diversity and its measuring. New Jersey, Princeton University Press.
Maldonado, P., Hohne, E., Naumann, M. (2006): Atlas del Gran Chaco Sudamericano. Buenos Aires, Sociedad Alemana de Cooperación Técnica (GTZ) ErreGé and Asoc.
Margalef, R. (1972): Homage to E. Hutchison, or why is there an upper limit to diversity. Trans. Conn. Acad. Arts Sci. 44: 21-235.
Mati, V.L.T., Pinto, H.A., Melo, A.L. (2015): Helminths of Liophis miliaris (Squamata, Dipsadidae): a list of species and new records. Helminthologia 52: 159-166.
Matias, C.S., Silva, C., Sousa, J.G., Ávila, R. (2018): Helminths infecting the black false boa Pseudoboa nigra (Squamata: Dipsadidae) in northeastern Brazil. Acta Herpetol. 13: 171-175.
McVinish R., Lester R.J.G. (2020): Measuring aggregation in parasite populations. J. R. Soc. Interface. 17: 20190886.
Möhl, K., Grosse, K., Hamedy, A., Wüste, T., Kabelitz, K., Lücker, E. (2009): Biology of Alaria spp. and human exposition risk to Alaria mesocercariae – a review. Parasitol. Res. 105: 1-15.
Montes, M.M., Furlán, E.O., Barneche, J.A., Vercellini, C., Acuña Gonzalez, T., Ibañez Shimabukuro, M., Arzamendia, V. (2025): New information on adults pentastomids (Crustacea: Pentastomida) found in ophidians from Argentina: Insights from 28S rDNA and COI mtDNA. Syst. Parasitol. 102: 25.
Morales, G., Pino, L. (1987): Parasitología cuantitativa. Caracas, Fundación Fondo Editorial Acta Científica Venezolana.
Niewiadomska, K., Pojmańska, T. (2011): Multiple strategies of digenean trematodes to complete their life cycles. Ann. Parasitol. 57: 233-241.
Nogueira, C., Williams, J., Giraudo, A., Fitzgerald, L., Arzamendia, V., Scrocchi, G., Schargel, W., Rivas, G. (2020): Erythrolamprus poecilogyrus. In: The IUCN Red List of Threatened Species. e.T50956250A50956261.
Nori, J., Torres, R.M., Lescano, J.N., Cordier, J.M., Periago, M.E., Baldo, J.D. (2016): Protected areas and spatial conservation priorities for endemic vertebrates of the Gran Chaco, one of the most threatened ecoregions of the world. Divers. distrib. 22: 1212-1219.
Oliveira, M.C., Ferreira-Silva, C., Silva, R.J., França, F.G.R., Lorenço-de-Moraes, R. (2024): Water snakes have a high diversity of parasites in anthropized environments. Urban Ecosyst. 27: 1435-1449.
Pence, D.B., Eason, S. (1980): Comparison of the helminth faunas of two sympatric top carnivores from the rolling plains of Texas. J. Parasitol. 66: 115-120.
Pereira, J.V., Mascarenhas, C.S., Müller, G. (2022): Digenea parasitizing snakes in Pampa Biome, southern Brazil. Rev. Mex. Biodiv. 93: e934112.
Pérez-Ponce de León, G., Poulin, R. (2016): Taxonomic distribution of cryptic diversity among metazoans: not so homogeneous after all. Biol. Lett. 12: 20160371.
Pérez-Ponce de León, G., Poulin, R. (2018): An updated look at the uneven distribution of cryptic diversity among parasitic helminths. J Helminthol 92: 197-202.
Pinto, R.R., Fernandes, R. (2004): Reproductive biology and diet of Liophis poecilogyrus poecilogyrus (Serpentes, Colubridae) from southeastern Brazil. Phyllomedusa 3: 9-14.
Poulin, R. (1993): The disparity between observed and uniform distributions: A new look at parasite aggregation, Int. J. Parasitol. 23: 937-944.
Poulin, R. (1997): Species richness of parasite assemblages: Evolution and patterns. Annu Rev Ecol Evol Syst. 28: 341-358.
Poulin, R. (1999): The functional importance of parasites in animal communities: Many roles at many levels? Int. J. Parasitol. 29: 903-914.
Poulin, R. (2001): Interactions between species and the structure of helminth communities. Parasitology. 122: S3-S11.
Poulin, R., Morand, S. (2000): The diversity of parasites. Q. Rev. Biol. 75: 277-293.
Poulin, R., Nichol, K., Latham, A.D. (2003): Host sharing and host manipulation by larval helminths in shore crabs: Cooperation or conflict? Int. J. Parasitol. 33: 425-433.
Prieto, Y.A., Giraudo, A.R., López M.S. (2012): Diet and sexual dimorphism of Liophis poecilogyrus (Serpentes, Dipsadidae) from the wetland regions of northeast Argentina. J. Herpetol. 46: 402-406.
Quintela, F.M., Iob, G., Artioli, L.G.S. (2014): Diet of Procyon cancrivorus (Carnivora, Procyonidae) in restinga and estuarine environments of southern Brazil. Iheringia Sér. Zool. 104: 143-149.
Reiczigel, J., Marozzi, M., Ibolya, F., Rózsa, L. (2024): Biostatistics for parasitologists: A painless introduction. In: Concepts in Parasitology, pp. 84-91. Gardner, S.L., Gardner, S.A., Eds, Lincoln Nebraska, Zea Books.
Richardson, D.J. (2006): Life cycle of Oligacanthorhynchus tortuosa (Oligacanthorhynchidae), an acanthocephalan of the Virginia opossum (Didelphis virginiana). Comp. Parasitol. 73: 1-6.
Rios, F.M., Zimmerman, L.M. (2015): Immunology of Reptiles. In: eLS. John Wiley & Sons, Ltd: Chichester. UK.
Rodgers, M.L., Bolnick, D.I. (2024): Opening a can of worms: A test of the co-infection facilitation hypothesis. Oecologia 204: 317-325.
Santoro, M., Aznar, F.J., Mattiucci, S., Kinsella, J.M., Pellegrino, F., Cipriani, P., Nascetti, G. (2013): Parasite assemblages in the western whip snake Hierophis viridiflavus carbonarius (Colubridae) from southern Italy. J. Helminthol. 87: 277-285.
Schad, G.A. (1956): Studies on the genus Kalicephalus (Nematoda: Diaphanocephalidae). I. On the life histories of the North American Species K. parvus, K. agkistrodontis and K. rectiphilus. Can. J. Zool. 34: 425-452.
Scholz, T., De Chambrier, A. (2003): Taxonomy and biology of proteocephalidean cestodes: Current state and perspectives. Helminthologia 40: 65-77.
Silva-Trejo, F.S., Sánchez-Nava, P., Sunny, A., Venegas-Barrera, C.S., Manjarrez, J. (2025): Parasite fauna of the blackbelly garter snake (Thamnophis melanogaster) in Central México. Diversity: 17: 11.
Thul, J.E., Forrester, D.J., Abercrombie, C.L. (1985): Ecology of parasitic helminths of wood ducks Aix sponsa In the Atlantic flyway. Proc. Helminthol. Soc. Wash. 52: 297-310.
TNC (2005): Evaluación Ecorregional del Gran Chaco Americano. Buenos Aires, Fundación Vida Silvestre Argentina (FVSA).
Warburton, E.M. (2020): Untapped potential: The utility of drylands for testing eco-evolutionary relationships between hosts and parasites. Int. J. Parasitol. Parasites Wildl. 12: 291-299.
Williams, J., Vera, D., Di Pietro, D. (2021): Lista comentada de las serpientes de la Argentina, con referencias a su sistemática, distribución geográfica, dieta, reproducción, potencial peligrosidad y etimologías. Revista Mus La Plata 6: 26-124.
William, J., Vera, D. (2023): Serpientes de la Argentina. Buenos Aires, Ediciones LBN.
Wood, C.L., Johnson, P.T. (2015): A world without parasites: Exploring the hidden ecology of infection. Front. Ecol. Evol. 13: 425-434.
Yamaguti, S. (1975): A synoptical review of life histories of digenetic trematodes of vertebrates: With special reference to the morphology of their larval forms. Tokyo, Yugaku-sha.