Submitted on: 2025, 18th June; revised on: 2025, 11th November; accepted on: 2025, 14th November.
Spiny treasures of Gorgona Island: Unveiling the natural history secrets of the lizard Enyalioides heterolepis (Squamata: Hoplocercidae) in Colombia
Grupo de Morfología y Ecología Evolutiva, Instituto de Ciencias Naturales, Universidad Nacional de Colombia. Carrera 45 # 26-85. Postcode: 111321 - Bogotá, Colombia
Email: miamendezga@unal.edu.co
Abstract. Novel insights into the natural history of the spiny woodlizard or spiny dwarf iguana, Enyalioides heterolepis (Bocourt, 1874), are presented based on a distinctive population from Gorgona Island, Cauca department, Colombia. Between July 2023 and August 2024, diurnal and nocturnal field surveys were conducted, and some ecological and morphological data were recorded. E. heterolepis is a diurnal, terrestrial species that primarily uses leaf litter. However, it shifts to arboreal microhabitats at night for sleeping, perching on trunks and leaves of similar heights and diameters across sexes. The species exhibits sexual dimorphism in body size and ventral sexual dichromatism, as well as a low frequency of caudal autotomy in individuals. These findings contrast with patterns observed in other neotropical rainforest iguanian lizards (Anolis, Plica, Uracentron) and partially align with previous reports for this and other Enyalioides species. Additionally, results are discussed in the context of island-mainland differences in lizard population densities. Predation pressure, social behavior, and the ecological and evolutionary bases of sexual dimorphism and dichromatism are proposed as key factors influencing the ecological traits of this species.
Keywords. Density compensation, Chocó biogeographic region, hoplocercid lizards, sleeping site fidelity.
Index
Additional natural history notes
There is a particular phenomenon within island biogeography known as “density compensation” (MacArthur et al., 1972), which postulates that, especially on small or isolated islands, a reduction in the number of species lowers the intensity in limiting factors such as predation, parasitism, or competition, thereby allowing some species – typically less abundant on the mainland – to reach higher population densities (MacArthur et al., 1972; Rodda and Dean‐Bradley, 2002; Buckley and Jetz, 2007). This discrepancy provides an opportunity to better understand various biological and ecological aspects of these species by studying island populations, in order to extrapolate findings to the continent or compare them with those found in mainland populations.
One possible case of density compensation is the spiny woodlizard or spiny dwarf iguana Enyalioides heterolepis (Bocourt, 1874; Fig. 1), a medium-sized lizard (182 mm maximum snout-vent length) that inhabits the lower stratum of primary and secondary tropical humid forests of the Chocó biogeographic region, ranging from 0 to 1150 meters in elevation from Panama to Ecuador, passing through Colombia (Páez et al., 2002; Eisenberg and Pantchev, 2010; MECN, 2010; Rios et al., 2011; Castro-Herrera et al., 2012; Cardona-Botero et al., 2013; Medina-Rangel et al., 2017; Pinto-Erazo et al., 2020). Its abundances are typically low, with most studies reporting findings on the mainland (Eisenberg and Pantchev, 2010; MECN, 2010; Rios et al., 2011; Pinto-Erazo et al., 2020). However, populations of this species are particularly numerous on Gorgona Island, in Colombia (Urbina-Cardona and Londoño-M, 2003; Castro-Herrera et al., 2012).

Some natural history traits of E. heterolepis have been mentioned in the literature, but these reports are often descriptive and provide either insufficient data or involve a low number of individuals (Páez et al., 2002; Eisenberg and Pantchev, 2010; MECN, 2010; Castro-Herrera et al., 2012; Medina-Rangel et al., 2017). This raises several questions: Is this species primarily terrestrial (Castro-Herrera et al., 2012; Medina-Rangel et al., 2017), arboreal in the lower stratum (Páez et al., 2002), or both? What is the range of body size, and what heights do nocturnal perches reach for this species (Eisenberg and Pantchev, 2010)? Does it prefer only fallen branches and trunks for sleeping sites (Páez et al., 2002), or does it also use leaves (MECN, 2010) or other types of perches?
In the present study, I am to clarify and expand the knowledge on some natural history traits of E. heterolepis using data from populations on Gorgona Island, Colombia, including a preliminary quantification of overall and sex-related prevalence of tail loss.
Gorgona National Natural Park, or PNN Gorgona, is located in the Pacific region of Colombia, on Gorgona Island and Gorgonilla islet (2°9’ N, 78°2’ W, 0-338 m a.s.l.), Cauca Department, Guapi Municipality (Fig. 2). The island measures 8.5 km in length and 2.5 km in width, with a maximum elevation of 338 meters above sea level. It is part of the National Natural Parks of Colombia (PNN) system, designated as a protected area. The island is primarily covered by primary and secondary tropical rainforest with some palm groves (Cocos nucifera L.; Sedano-Cruz et al., 2024). The average annual temperature is 26°C, and the relative humidity is 90% (Blanco, 2009), with both showing an almost negligible variation throughout the year, reflecting the highly stable climatic conditions of the island (Rangel-Ch and Rudas-LL, 1990). The protected area covers 617 km², underscoring its ecological significance and conservation value.

Data were collected on three different sampling dates: July 2023, January-February 2024, and August 2024 (Table 1). Three observers searched for lizards in the tropical rainforest vegetation cover using the time-constrained Visual Encounter Survey (VES) transect method (Crump and Scott, 1994; Lips, 2001) during two different sampling periods: daytime (06:00 – 18:00) and nighttime (20:00 – 24:00) hours.
| Sampling day | Sampling period | Sampling interval (total hours) | Number of lizards |
|---|---|---|---|
| 5 July 2023 | Day | 11:00-14:00 (3) | 4 |
| 8 July 2023 | Day | 09:00-11:00 (2) | 4 |
| 4 February 2024 | Day | 14:00-18:00 (4) | 11 |
| 5 February 2024 | Day | 06:00-09:00 (3) | 2 |
| 5 July 2023 | Night | 20:30-23:30 (3) | 4 |
| 7 July 2023 | Night | 21:00-23:00 (2) | 7 |
| 9 July 2023 | Night | 21:00-23:00 (2) | 6 |
| 28 January 2024 | Night | 20:00-24:00 (4) | 1 |
| 26 August 2024 | Night | 20:00-23:00 (3) | 5 |
| 30 August 2024 | Night | 21:00-22:30 (1.5) | 2 |
During diurnal samplings, sighting time, microhabitat type (leaf litter, rock, soil, leaf/leaves, branch, fallen trunk, vine, trunk or root; Fig. 2), and, where applicable, perch height (in cm) for each individual were recorded without capturing them. During nocturnal samplings, individuals were captured along the same routes surveyed during the day, although not necessarily covering the exact same distance or duration. For each captured individual, microhabitat type, perch height and perch diameter (in cm), snout-vent length (SVL; in mm), total tail length (TL, in mm), regenerate tail length (regTL; in mm), non-regenerate tail length (no.regTL; in mm), and in case of adult individuals, sex (determined by femoral pores present only in males; Eisenberg and Pantchev, 2010) were recorded. Perch height and perch diameter were measured using a measuring tape, while SVL and TL were measured with a precision analog caliper (precision = 0.05 mm).
To avoid pseudoreplication, diurnal samplings were conducted in different locations for each sampling period, while nocturnal samplings were conducted in distinct locations throughout the entire study.
The encounter rate (relative abundance) was calculated as the total number of lizards observed divided by the total observation hours and the number of observers (individuals h⁻¹ observer⁻¹; Lips, 2001). For microhabitat use, I used the Chi-square test of independence to test differences between diurnal and nocturnal use of terrestrial/ground (leaf litter, rock, soil and root) and arboreal/perch (leaves, branch, fallen trunk, tree trunk and vine) microhabitats. I assessed differences between sexes in snout–vent length (SVL), intact tail length (TL), and perch diameter using an ANOVA or Kruskal-Wallis test. Posteriorly, differences between sexes in perch height were evaluated using a linear model, with perch height as the response variable, sex as a fixed factor, and snout–vent length (SVL) as a covariate to account for potential effects of body size. Prior to analyses, data were inspected for linearity, normality, and homoscedasticity assumptions.
On the other hand, the Chi-square goodness-of-fit test was conducted to determine whether there was an equal number of individuals with regenerated tails compared to those with intact tails. Additionally, I employed the Fisher’s exact test to assess whether there was a significant difference in the proportion of regenerated tails between sexes only in adult individuals (see Results for age class criteria). Finally, the ANOVA or the Kruskal-Wallis test were used to determine if lizards with intact tails exhibited similar tail lengths compared to individuals with regenerated tails. Statistical tests and graphics were conducted using RStudio software v1.3.959 (RStudio Team, 2020).
Over four days, a total of 12 hours of diurnal sampling were conducted, distributed between 06:00 to 18:00 h, amounting to a total of 36 observer hours, and 21 lizards were recorded (Table 1). Similarly, over six days, from 20:00 to 24:00 h, I completed 15.5 hours of nocturnal sampling, totaling 46.5 observer hours and recording 25 sleeping lizards (Table 1). As a result, the lizard encounter rate was 0.58 individuals h⁻¹ observer⁻¹ for diurnal sampling and 0.54 individuals h⁻¹ observer⁻¹ for nocturnal sampling.
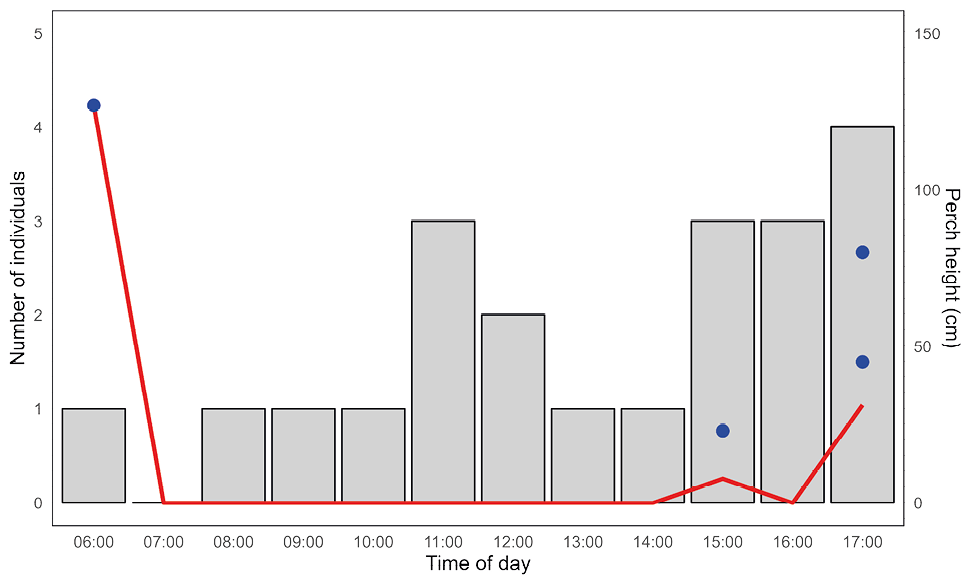
The spiny dwarf iguana Enyalioides heterolepis from Gorgona Island was found exclusively in the lowland tropical forest (mostly secondary) of the island. In general, lizards were observed on the ground, generally under shade or filtered sunlight. However, particularly at 6:00 h and 15:00 to 18:00 h, a few lizards were recorded using perches such as fallen trunks, leaves and branches, positioned between 23 and 127 cm above the ground (Fig. 3).
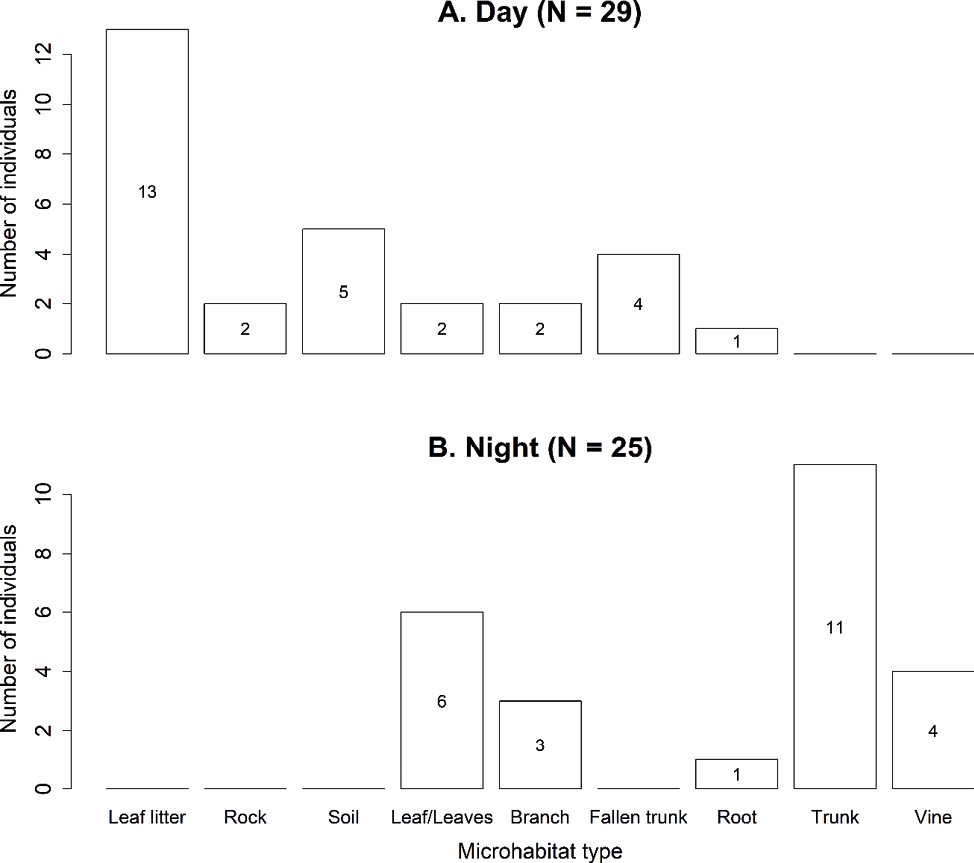
Eight additional diurnal lizard observations were recorded from occasional encounters or brief samplings, completed a total of 29 lizard observations for microhabitat data. During the day, E. heterolepis inhabits principally terrestrial/ground microhabitats (N = 21; 72.4%), mostly on leaf litter (44.8%), followed by bare soil (17.2%) and fallen trunks (13.8%; Fig. 4A). Some of the individuals using fallen trunks, leaves and branches correspond to the arboreal/perch microhabitat observations in Fig. 3 (in blue dots). In contrast, during the night, of the 25 observed lizards, 44% were found sleeping perching on tree trunks, followed by leaves (24%), vines (16%) and branches (12%) (Fig. 4B), with a total of 24 lizards (96%) using arboreal/perch microhabitats to sleep (Fig. 5). Thus, terrestrial and arboreal microhabitat use change significantly between day and night (χ² = 26.028, df = 1, P < 0.05).

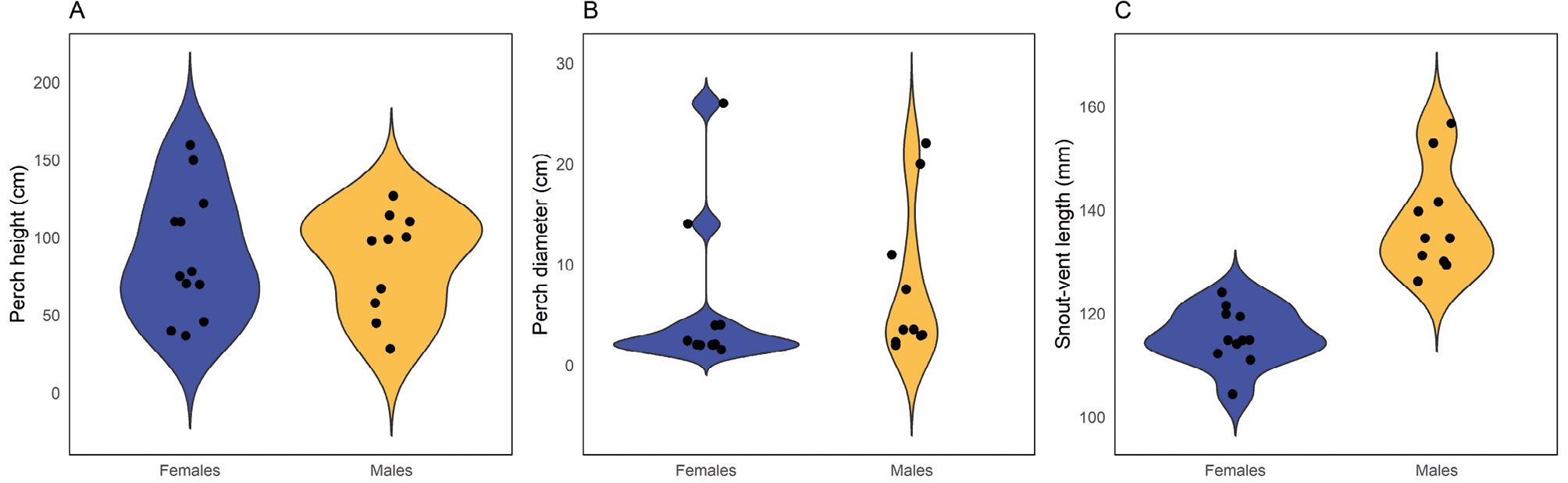
Adult males were found to be significantly larger than adult females in terms of the snout-vent length (SVL) (F1,19 = 39.5, P < 0.05; Fig. 6C; Table 2) and intact tail length (TL) (F1,12 = 24.1, P < 0.05; Table 2). During nocturnal sampling, only two juveniles were captured, while a third juvenile was occasionally captured during diurnal sampling. Although there is no available evidence about the sexual maturation size for males or females in this species, the largest juvenile reached 84.4 mm SVL, while the shortest adult, a female, measured 104.4 mm SVL, with a gap of 20 mm between the two sizes. This difference is larger than any SVL variation among adult measurements (Fig. 6C), suggesting that this gap could serve as an alternative criterion to differentiate between adults from juveniles in this dataset. Additionally, the smallest gravid female found had an SVL of 111 mm, providing additional support for the established adult size threshold.
| Perch height (cm) |
Perch diameter (cm) |
Snout-vent length (SVL) (mm) |
Tail length (TL) (mm) |
Non-regenerate tail portion (no.regTL) (mm) |
Regenerate tail portion (regTL) (mm) |
regTL + no.regTL (mm) |
|
|---|---|---|---|---|---|---|---|
| Males | 84.6 ± 32.9 (28-127) N = 10 |
7.7 ± 7.5 (2-22) N = 10 |
137.6 ± 10.3 (126-156.8) N = 10 |
199.2 ± 13.1 (179.5-214.1) N = 8 |
40.5 ± 13 (31.25-49.7) N = 2 |
70.8 ± 14.8 (60.4-81.3) N = 2 |
111.3 ± 1.7 (110.1-112.5) N = 2 |
| Females | 88.9 ± 41.1 (37-159) N = 12 |
5.4 ± 7.3 (1.5-26) N = 12 |
115.5 ± 5.5 (104.4-124.1) N = 11 |
159.9 ± 16.9 (134.8-175.7) N = 6 |
57.7 ± 37.7 (21.2-121) N = 5 |
39.8 ± 16 (19.3-58) N = 5 |
87.4 ± 39.2 (44-140.3) N = 5 |
| Juveniles | 45.5 ± 16 (34-57) N = 2 |
12.2 ± 2.5 (10.5-14) N = 2 |
62.2 ± 23 (38.5-84.4) N = 3 |
87.65 ± 38.5 (59.5 - 131.6) N = 3 |
- | - | - |
| Total | 83.5 ± 37.1 (28-159) N = 24 |
6.9 ± 7.2 (1.5-26) N = 24 |
118.1 ± 26 (38.5-156.8) N = 24 |
165.6 ± 45.4 (59.5-214.1) N = 17 |
52.8 ± 32.3 (21.2-49.7) N = 7 |
48.7 ± 20.9 (19.3-81.3) N = 7 |
94.2 ± 34.1 (44-140.3) N = 7 |
Perch diameter (H = 2.14, df = 1, P = 0.14) of nocturnal arboreal microhabitats was quite similar between adult males and females (Fig. 6A-B; Table 2). However, it is necessary to highlight the presence of four outliers above 12 cm (two for each sex), which corresponds to perches used in leaf microhabitats (Fig. 6B). Excluding these data points revealed a marginally tendency (H = 3.69, df = 1, P = 0.055) for males to use slightly broader perches (Mean ± SD (Min-Max) N: 4.5 ± 3.1 (2-11) N = 8) compared to females (Mean ± SD (Min-Max) N: 2.5 ± 0.8 (2-4) N = 9). On the other hand, the linear regression model including sex and snout–vent length (SVL) as predictors indicated no significant effects on perch height (overall model: F₂,₁₈ = 1.46, P = 0.26, R² = 0.14). Although males tended to use slightly lower perches than females (β = −44.76 ± 27.36, P = 0.12) and perch height tended to increase with body size (β = 1.65 ± 1.02, P = 0.12), neither effect was substantial.
A total of 17 individuals were found to have intact tails, while 7 individuals (representing 29% of the total individuals) exhibited regenerated tails (Table 2). This discrepancy in the proportions between the two groups was statistically significant (χ² = 4.167, df = 1, P < 0.05). All captured juveniles had intact tails. Among adults, 8 males and 6 females had intact tails while 2 males and 5 females had regenerated tails. There was no significant association between tail condition and sex (Fisher’s exact test, P = 0.36).
Among the 7 individuals with regenerated tails, the average proportion of the regenerated section was 0.5 ± 0.19, ranging from 0.14 to 0.72. Additionally, these individuals had shorter total tail lengths (regTL + no.regTL) compared to the total tail lengths (TL) of the 14 individuals with intact tails (F1,19 = 48.26, P < 0.05).
Unlike previous studies on the mainland (Eisenberg and Pantchev, 2010; MECN, 2010; Rios et al., 2011; Pinto-Erazo et al., 2020), a substantial abundance of the lizard Enyalioides heterolepis on Gorgona Island was found, observing 21 diurnal, 25 nocturnal, and 8 occasional encounters, with a total 54 lizard observations. Moreover, this value is similar to the 46 individuals found by Urbina-Cardona and Londoño-M (2003) with a sampling effort of 32 transects of 200 m each, and contrast with the 17 individuals recorded by Rios et al. (2011) in 448 h⁻¹ observer⁻¹ (0.038 individuals h⁻¹ observer⁻¹) or the two 2 individuals reported by Pinto-Erazo et al. (2020) in 1604 hours.person-1 (0.001 individuals h⁻¹ observer⁻¹) at other continental sites in the Pacific region of Colombia. This mainland-island difference in abundance of E. heterolepis agrees with the “density compensation” phenomenon (MacArthur et al., 1972) of higher populations densities of species in islands vs. continent, which also has robust evidence in lizards (Novosolov et al., 2015).
These authors suggest that lizard population densities are higher in snake-free islands. In this regard, our findings are even more notable given the large snake species richness of Gorgona Island, hence the name of the place (Urbina-Cardona and Londoño-M, 2003; Urbina-Cardona et al., 2008; Castro-Herrera et al., 2012). However, only a better comparison of the richness, abundance and composition of snake diversity between E. heterolepis continental vs. Gorgona Island populations could outperform our understanding of island-mainland snake predatory pressure on this species.
Studies on natural history and ecology of hoplocercid lizards are scarce (but also see Eisenberg and Pantchev, 2010 and Thomas et al., 2021), compared with studies of other neotropical rainforest diurnal lizards such as Anolis (Vitt et al., 2003 a, c), tropidurids (Vitt et al., 1997; Ellinger et al., 2001), teiids (Biázquez, 1996; Mesquita et al., 2006), and microteiids (Vitt and Avila-Pires, 1998; Vitt et al., 1998, 2003 b, 2007). Specifically, E. heterolepis is a terrestrial species that uses principally leaf litter during the day, contrasting to arboreal habits of other iguanian lizards such as Anolis, Plica, and Uracentron (Vitt et al., 1997; 2003 a, c; Ellinger et al., 2001), and showing some ecological resemblance to microteiids such as Potamites, Cercosaura and Alopoglossus, although these lizards are more cryptic and live under the leaf litter (Vitt and Avila-Pires, 1998; Vitt et al., 1998, 2003 b, 2007). Terrestriality was also observed twice in the hoplocercid E. groi (Corredor et al., 1985; Vásquez-Restrepo, 2021). On the other hand, teiids prefer the open ground (Biázquez, 1996; Mesquita et al., 2006), with species of the genus Crocodilurus associated with water streams, while Dracaena guianensis is found perched on shrubs (Mesquita et al., 2006). Microteiids of the genus Potamites are also associated with water streams, in contrast to E. heterolepis.
Microhabitat use switches completely between day and night in E. heterolepis, changing from a leaf litter terrestrial habit to arboreal perches, particularly trunks and leaves. Sleeping perches have been previously reported in this species (Eisenberg and Pantchev, 2010) as well as in E. laticeps, although E. laticeps is mainly arboreal during its diurnal activity period (Thomas et al., 2021). The number of observed individuals and perch height agree with this circadian change of microhabitat use, with an afternoon-crepuscular peak of observations between 15:00 and 18:00 h and three individuals with perch heights of 23, 45, and 80 cm within this period, suggesting active searching and climbing to sleeping perches. In contrast, Vásquez-Restrepo (2021) found a female of E. groi at 19 h in a burrow, suggesting a different sleep microhabitat for this Enyalioides species.
Thomas et al. (2021) noted that sleeping site selection could play an anti-predation role, provide greater stability (also related to predation) when sleep, or offer thermoregulatory advantages in the morning. This could also explain the abundance of E. heterolepis on Gorgona Island despite its richness of snake species. In addition, a midday peak of observations between 11:00 to 13:00 h occurs, which could be more related with thermoregulatory activity of diurnal lizard species (Ellinger et al., 2001; Biázquez, 1996; Mesquita et al., 2006; Vitt and Avila-Pires, 1998; Vitt et al., 1997, 1998, 2003 a, b, c, 2007), in which case predation or stability seems to be better explanations of sleeping site selection. However, additional studies on predation risk and thermoregulation are necessary to test these hypotheses.
Both males and females used sleeping perches of similar heights and diameters. In contrast, there was evident sexual dimorphism in SVL, with males being larger than females. This pattern differs from other studies on neotropical rainforest diurnal lizards, where there is no sexual dimorphism (Vitt et al., 1997, 2003 b, 2007, Mesquita et al., 2006, Ellinger et al., 2001) or when it exist, females are larger than males (Vitt and Avila-Pires, 1998; Vitt et al., 1998, 2003 c). Additionally, not only size, but also coloration differed between sexes. In all captured adults, distinct blackish blotches were observed only in males, a color trait previously noted by Eisenberg and Pantchev (2010). This type of ventral sexual dichromatism has been documented in other lizard genera, such as Sceloporus (Putman et al., 2025) and Platysaurus (Whiting 1999; Whiting et al., 2003), and is associated with conspecific interactions, for example, during male agonistic ventral displays in the latter.
Sexual dimorphism is commonly attributed to sexual selection, in terms of male competition or female choice, or natural selection related to male-female resource partitioning, for instance, in Anolis lizards (Rodríguez-Rodríguez and Calderón-Espinosa, 2024). Based on the above, it seems that habitat source does not play a role in the sexual dimorphism and dichromatism observed in E. heterolepis, and other processes such as sexual selection, male status signaling, territorial defense, or individual recognition may be more plausible explanations. However, further studies are needed to clarify the evolutionary basis of sexual difference of these traits, as well as additional animal behavior research on this species.
The proportion of individuals exhibiting caudal autotomy was moderate to low (29%), which may reflect low predation pressure or efficiency, or a generally low susceptibility to predator attacks (Bateman and Fleming, 2009). Alternatively, this pattern may result from the use of other effective antipredator strategies, such as crypsis or early flight initiation (Vitt and Caldwell, 2014). For instance, individuals usually flew away from observers, some with a flight initiation distance greater than ten meters. Likewise, juveniles with intact tails may be more cryptic than adults, which could also explain their low abundance in this study, but the small sample size makes it difficult to assert this. Intraspecific aggression has also been proposed as an explanation for caudal autotomy rates, which are related to sex differences, with males having a higher rate than females due to their agonistic behavior (Bateman and Fleming, 2009). However, this is not the case for E. heterolepis, where these differences were not found. In fact, I found more adult females than males with caudal autotomy. Regardless, shorter tails in E. heterolepis individuals with caudal autotomy vs. individuals with intact tails seem to reflect the energetic cost of tail regeneration, but the time of regeneration is needed to confirm this (Bateman and Fleming, 2009).
Additional natural history notes
Similar to E. laticeps (Thomas et al., 2021), sleeping site fidelity in further surveys was observed: three adult individuals, one male and two females, were found 1.5, 4, and 7 meters away, respectively, from their original sleeping perches three nights after their first detection on 28 January 2024. Other natural history observations regarding sleeping sites include: (1) on 8 June 2023, a pair was observed sharing the same trunk perch, with the male and female positioned at perch heights of 98 cm and 159 cm, respectively; (2) on 28 January 2024, an adult female was found sleeping on a trunk perch that was subsequently used by an adult male three and eight nights later; and (3) homing behavior was documented on 27 August 2024, when an adult female translocated 50 meters from its original perch site returned within two days to a location approximately 4 meters away from it. Male-female interactions also occurred during diurnal activity, on 8 July 2023, when a pair was observed on the leaf litter at 11:00 h.
Gorgona Island provided an opportunity to study key ecological traits of the spiny woodlizard or spiny dwarf iguana (Enyalioides heterolepis). Present results indicate that this species is diurnal and terrestrial, primarily using leaf litter as its main substrate. At night, it shifts to arboreal microhabitats for sleeping, perching on trunks and leaves of similar heights and diameters in both sexes. I also found clear sexual dimorphism in body size and ventral sexual dichromatism, as well as low rates of caudal autotomy. Further research on predation pressures in island versus mainland populations, thermal ecophysiology, social behavior, and ecological and evolutionary bases of sexual differences is needed to better understand the patterns observed in this study.
I am grateful to the staff of Parques Nacionales Naturales de Colombia: Diego Grajales and Robinson Dias for their invaluable assistance with fieldwork and logistics on the island, and Geraldine Núñez, Álvaro Fierro, and Sofia Gomez for their help in obtaining permits and supporting field activities. I am also deeply grateful to the Gorgona field team, especially Alejandra Pinto-Erazo, Philipp Böning, Martha Lucía Calderón-Espinosa, Adriana Jerez, Darío Alarcón-Naforo, Camilo Yara, Marthin Rozo and Daniela Ortegón, for their invaluable assistance, unwavering commitment, and hard work throughout the fieldwork, and for the camaraderie that made the fieldwork both productive and truly enjoyable. This study was conducted under the authorization granted by Parques Nacionales Naturales de Colombia (Permit No. Memorando 20242000001553), whose support in logistics and accommodation was also essential.
Bateman, P.W., Fleming, P.A. (2009): To cut a long tail short: A review of lizard caudal autotomy studies carried out over the last 20 years. J. Zool. 277: 1-14.
Biázquez, M.C. (1996): Activity and habitat use in a population of Ameiva ameiva in southeastern Columbia. Biotropica: 714-719.
Blanco, J.F. (2009): The hydroclimatology of Gorgona Island: seasonal and ENSO-related patterns. Actual. Biol. 31: 111-121.
Buckley, L.B., Jetz, W. (2007): Insularity and the determinants of lizard population density. Ecol. Lett. 10: 481-489.
Cardona-Botero, V.E., Viáfara-Vega, R.A., Valencia-Zuleta, A., Echeverry-Bocanegra, A., Hernández-Córdoba, O.D., Jaramillo-Martinez, A.F., Galvis-Cruz, R., Gutiérrez-Zúñiga, J.A., Castro-Herrera, F. (2013): Diversidad de la herpetofauna en el Valle del Cauca (Colombia): un enfoque basado en la distribución por ecorregiones, altura y zonas de vida. Biota Colomb. 14.
Castro-Herrera, F., Valencia, A., Villaquirán, D. (2012): Diversidad de anfibios y reptiles del Parque Nacional Natural Isla Gorgona, 112 pp. Cali, Colombia, Feriva Impresores S.A.
Corredor, V., Renjifo, J.M., Ayala, S. (1985): Discovery of Morunasaurus groi Dunn (Sauria: Iguanidae) in northwestern Colombia. J. Herpetol. 19: 162-164.
Crump, M.L., Scott, N.J. Jr. (1994): Visual encounter surveys. In: Measuring and monitoring biological diversity: Standard methods for amphibians, pp. 84-91. Heyer, W. R., Donnelly, M. A., McDiarmid, R. W., Hayek, L. C., Foster, M. S., Eds, Washington, D.C., Smithsonian Institution Press.
Eisenberg, T., Pantchev, N. (2010): Notes on body size and natural history of Enyalioides heterolepis (Bocourt 1874) in its northernmost population in Panama. Herpetol. Bull. 111: 12-14.
Ellinger, N., Schlatte, G., Jerome, N., Hödl, W. (2001): Habitat use and activity patterns of the neotropical arboreal lizard Tropidurus (=Uracentron) azureus werneri (Tropiduridae). J. Herpetol. 35: 395-402.
Lips, K. R., Reaser, J. K., Young, B. E., & Ibáñez, R. (2001): Amphibian monitoring in Latin America: A protocol manual / Monitoreo de anfibios en América Latina: Manual de protocolos (Herpetological Circular No. 30). Society for the Study of Amphibians and Reptiles.
MacArthur, R.H., Diamond, J.M., Karr, J.R. (1972): Density compensation in island faunas. Ecology 53: 330-342.
MECN (2010): Serie Herpetofauna del Ecuador: El Chocó Esmeraldeño. Monografía 5: 1-232. Museo Ecuatoriano de Ciencias Naturales. Quito-Ecuador.
Medina-Rangel, G.F., Cárdenas-Arevalo, G., Rentería-Moreno, L.E. (2017): Herpetofauna del Cerro Tacarcuna, Serranía del Darién, Unguía, Chocó, Colombia. Guía de campo. ILAP-Instituto de Investigaciones Ambientales del Pacífico y Expedición Colombia-Bio 2017-2017-COLCIENCIAS. Quibdó, Chocó, Colombia.
Mesquita, D.O., Colli, G.R., Costa, G.C., França, F.G., Garda, A.A., Péres, A.K. (2006): At the water’s edge: Ecology of semiaquatic teiids in Brazilian Amazon. J. Herpetol. 40: 221-229.
Novosolov, M., Rodda, G.H., Feldman, A., Kadison, A.E., Dor, R., Meiri, S. (2016): Power in numbers. Drivers of high population density in insular lizards. Glob. Ecol. Biogeogr. 25: 87-95.
Páez, V.P., Bock, B.C., Estrada, J.J., Ortega, A.M., Daza, J.M., Gutiérrez-C., P.D. (2002): Guía de campo de algunas especies de anfibios y reptiles de Antioquia. Medellín, Colombia, Colciencias, Universidad de Antioquia y Universidad Nacional.
Pinto-Erazo, M.A., Calderón-Espinosa, M.L., Medina-Rangel, G.F., Méndez-Galeano, M.Á. (2020): Herpetofauna from two municipalities of southwestern Colombia. Biota Colomb. 21: 41-57.
Putman, B.J., Stevens, B., Fresco, N.A., Urquidi, E.R. (2025): Effects of urbanization on ventral patch size and phenotypic correlates of patch expression in male Western Fence Lizards (Sceloporus occidentalis). Ecol. Evol. 15: e70915.
Rangel-Ch., J. O., & Rudas-Ll., A. (1990): Macroclima de Gorgona y region costera aledana. In: Biota y ecosistemas de Gorgona, pp. 13-41. Aguirre, En J. C., Rangel Ch., J. O., Eds, Colombia, Fondo FEN.
Rios, E.E., Hurtado, C.F., Rengifo, J.T., Castro-Herrera, F. (2011): Lagartos en comunidades naturales de dos localidades en la región del Chocó de Colombia. Herpetotropicos 5: 85-92.
Rodda, G.H., Dean-Bradley, K. (2002): Excess density compensation of island herpetofaunal assemblages. J. Biogeogr. 29: 623-632.
Rodríguez-Rodríguez, J.D., Calderón-Espinosa, M.L. (2024): Sexual dimorphism and female ecomorphology in Anolis (Squamata: Anolidae): Knowledge of female morphology increases the understanding of Anolis diversification. Biol. J. Linn. Soc. 143: 1-14.
RStudio Team (2020): R Studio: Integrated development environment for R. Boston, MA, RStudio, PBC. URL http://www.rstudio.com/.
Sedano-Cruz, R.E., Pérez-Amaya, N., Rivera-Gutierrez, H.F. (2024): Vocal and genetic variation between a land-bridge island and mainland populations of the Black-crowned Antshrike (Thamnophilus atrinucha). Behav. Ecol. Sociobiol. 78: 42.
Thomas, O., Mangini, G.G., Gualinga, J. (2021): Sleeping site fidelity in three neotropical species of herpetofauna. Herpetol. Bull. 155: 8-11.
Urbina-Cardona, J.N., Londoño, M.C. (2003): Distribución de la comunidad de herpetofauna asociada a cuatro áreas con diferente grado de perturbación en la Isla Gorgona, Pacífico colombiano. Rev. Acad. Colomb. Ciencias 27: 105-114.
Urbina-Cardona, J.N., Londoño-Murcia, M.C., García-Ávila, D.G. (2008): Dinámica espacio-temporal en la diversidad de serpientes en cuatro hábitats con diferente grado de alteración antropogénica en el Parque Nacional Natural Isla Gorgona, Pacífico colombiano. Caldasia 30: 479-493.
Vásquez-Restrepo, J.D. (2021): 35 years behind the scenes: Range extension of the rare Gro’s manticore, Morunasaurus groi (Squamata, Hoplocercidae), in Colombia. Papéis Avulsos Zool. 61: e20216163.
Vitt, L.J., Avila-Pires, T.C. (1998): Ecology of two sympatric species of Neusticurus (Sauria: Gymnophthalmidae) in the western Amazon of Brazil. Copeia: 570-582.
Vitt, L.J., Caldwell, J.P. (2014): Herpetology: An introductory biology of amphibians and reptiles. 4th ed. San Diego, CA, Academic Press.
Vitt, L.J., Avila-Pires, T.C.S., Espósito, M.C., Sartorius, S.S., Zani, P.A. (2003a): Sharing Amazonian rain-forest trees: Ecology of Anolis punctatus and Anolis transversalis (Squamata: Polychrotidae). J. Herpetol. 37: 276-285.
Vitt, L.J., Ávila-Pires, T.C.S., Espósito, M.C., Sartorius, S.S., Zani, P.A. (2007): Ecology of Alopoglossus angulatus and A. atriventris (Squamata, Gymnophthalmidae) in Western Amazonia. Phyllomedusa 6: 11-21.
Vitt, L.J., Avila-Pires, T.C.S., Zani, P.A., Espósito, M.C., Sartorius, S.S. (2003b): Life at the interface: Ecology of Prionodactylus oshaughnessyi in the western Amazon and comparisons with P. argulus and P. eigenmanni. Can. J. Zool. 81: 302-312.
Vitt, L.J., Avila-Pires, T.C.S., Zani, P.A., Sartorius, S.S., Espósito, M.C. (2003c): Life above ground: Ecology of Anolis fuscoauratus in the Amazon rain forest, and comparisons with its nearest relatives. Can. J. Zool. 81: 142-156.
Vitt, L.J., Sartorius, S.S., Avila-Pires, T.C.S., Espósito, M.C. (1998): Use of time, space, and food by the gymnophthalmid lizard Prionodactylus eigenmanni from the western Amazon of Brazil. Can. J. Zool. 76: 1681-1688.
Vitt, L.J., Zani, P.A., Avila-Pires, T.C.S. (1997): Ecology of the arboreal tropidurid lizard Tropidurus (=Plica) umbra in the Amazon region. Can. J. Zool. 75: 1876-1882.
Whiting, M.J. (1999): When to be neighbourly: differential agonistic responses in the lizard Platysaurus broadleyi. Behav. Ecol. Sociobiol. 46: 210-214.
Whiting, M.J., Nagy, K.A., Bateman, P.W. (2003): Evolution and maintenance of social status-signaling badges. In: Lizard Social Behavior, pp. 47-82. Fox, S.F., McCoy, J.K., Baird, T.A., Eds, Baltimore, Johns Hopkins University Press.