Submitted on: 2025, 22th July; revised on: 2025, 17th October; accepted on: 2025, 29th October
Occupancy of artificial shelters by European tree frog (Hyla arborea) in Škocjanski zatok Nature Reserve: A non-invasive sampling method in ecologically sensitive habitats
University of Primorska, Faculty of Mathematics, Science and Information Technology (UP FAMNIT), Glagoljaška 8, 6000 - Koper, Slovenia
*Corresponding author. Email: martina.luznik@upr.si
Abstract. Monitoring amphibians in non-breeding habitats is constrained by low detectability and potential disturbance to sensitive ecosystems. We tested the effectiveness of artificial shelters - PVC pipes - as a non-invasive method for monitoring the European tree frog (Hyla arborea) outside the breeding season in Škocjanski zatok Nature Reserve (Koper, Slovenia). Over three years (2022–2024), we installed 40- and 50-mm diameter shelter pipes at 24 sites and conducted regular inspections. A total of 338 encounters of the focal species were recorded, with frogs of all sizes using the shelters. Overall shelter occupancy declined across years, possibly reflecting a combination of factors including reduced recruitment, vegetation changes, demographic fluctuations, and weather conditions. A statistically significant trend was observed, with narrower (40-mm) shelter pipes having 1.6 to 1.7 times more captures than wider (50-mm) ones, despite no significant difference in the body size of frogs occupying each type. By using PVC artificial shelters, we greatly enhanced the detectability of H. arborea in the Škocjanski zatok Nature Reserve compared to classical monitoring methods such as acoustic surveys or dip-netting. Importantly, a non-invasive approach using artificial shelters minimized the risk of disturbance to breeding and migratory birds, which is essential in areas with high avifaunal conservation value. Our results support the broader use of artificial shelters for monitoring arboreal amphibians in ecologically sensitive areas and suggest future research should explore finer-scale shelter design to improve detection and ecological interpretation across life stages.
Keywords. Hyla arborea, artificial shelters, PVC pipes, amphibian monitoring, protected areas.
Index
Study site and artificial shelters
In recent decades, multi-factorial human-induced changes have contributed to declines in amphibian populations worldwide through different mechanisms, such as habitat loss and fragmentation, emerging infectious disease, toxic substances, and climate change (Alford and Richards, 1999; Houlahan et al., 2000; Falaschi et al., 2019; Luedtke et al., 2023). As a result of their imperilled status, many amphibians are included in monitoring programmes, whereby a major challenge is the imperfect detection of species and the resulting underestimation of a species’ distribution and abundance (Tanadini and Schmidt, 2011; Cruickshank et al., 2021). Most studies of amphibians focus on the reproductive season when they can be found at aquatic habitats in which the species breed, the use of which could be influenced by both landscape-related land-use patterns and site-specific factors. However, much less is known about the use of terrestrial habitats surrounding breeding habitats, these areas being crucial for the long-term survival of amphibian populations (Marsh and Trenham, 2001; Semlitsch and Bodie, 2003; Van Buskirk, 2005).
One of the amphibian species experiencing population declines is the European tree frog Hyla arborea (Linnaeus 1758), which has shown population decreases in parts of its range, particularly in western Europe, despite being listed as Least Concern on the IUCN Red List (Agasyan et al., 2024). Nevertheless, H. arborea is listed as vulnerable and protected by Slovenian national legislation (Ministry of Environment, Spatial Planning and Energy, 2002). Hyla arborea has a prolonged breeding season that lasts from April to June. During the breeding cycle, adults stay in or near the spawning site or in riparian vegetation such as reeds (Ferreira et al., 2012; Pellet et al., 2006) where they can be readily sampled. In the post-breeding season, treefrogs can migrate a few hundred meters and live scattered about in terrestrial habitats (Pellet et al., 2006; Kovács et al., 2007). During this period, they are inconspicuous and very difficult to locate, so knowledge of microhabitat use outside the breeding season is very sparse, although good quality terrestrial microhabitat is crucial for the long-term persistence of this species (Pellet et al., 2004, 2006).
To address sampling this species during the non-breeding season, several approaches have been used. For example, Pellet et al. (2006) employed harmonic direction finders to track individual movements. Others, such as Kovács et al. (2007), searched directly for frogs in shrubs and trees within wetland areas to investigate diet and microhabitat use. However, such methods can be time-consuming or logistically demanding, limiting their applicability for regular monitoring. Artificial shelters – particularly polyvinyl chloride (PVC) or bamboo pipe shelters that mimic natural hiding places of hylids – have been proposed as an efficient and non-invasive alternative (Boughton et al., 2000; Bartareau, 2004; Myers et al., 2007; Johnson et al., 2008; Moulton et al., 2009; do Vale et al., 2018). This method has been proven useful in both ecological (Mahan and Johnson, 2007; Liner et al., 2008; Campbell et al., 2010; McGhee, 2020) and conservation contexts (Schurbon and Fauth, 2003; Wyatt and Forys, 2004; Suriyamongkol et al., 2021). Furthermore, it has also been explored as a tool for microhabitat creation or threat mitigation for amphibians and other vertebrates (Cowan et al., 2021; Suriyamongkol et al., 2021).
To our knowledge, the artificial shelter method has not yet been used in studies of H. arborea, but do Vale et al. (2018) have used this method to study another closely related hylid species of similar size, the Iberian tree frog H. molleri Bedriaga 1889. The purpose of the present study was to test a novel, non-invasive sampling method for H. arborea outside the breeding habitat within one of the last remaining strongholds of this species in Slovenian Istria (Veenvliet and Kus Veenvliet, 2014; Senič et al., 2024). The specific objectives of the study were to: (1) investigate the suitability of polyvinyl chloride (PVC) pipes as shelters for H. arborea monitoring; (2) evaluate the occupancy of PVC shelters of different pipe diameters in relation to the frog body size. Given that the design of the artificial shelters mimics natural hylid refuges, we hypothesized that H. arborea would readily occupy them.
Study site and artificial shelters
Located in the municipality of Koper (Slovenian Istria), the Škocjanski zatok Nature Reserve is connected to the Adriatic Sea only via a narrow channel. The reserve comprises a shallow, tidally influenced brackish lagoon dominated by halophytic vegetation and a freshwater marsh (Fig. 1) characterised by open water, meadows, shrubland, and extensive reed stands dominated by common reed (Phragmites australis) (Mozetič and Lipej, 2014). Hyla arborea inhabits the freshwater section of the Nature Reserve, where it finds suitable spawning sites and abundant riparian and terrestrial vegetation (Veenvliet and Kus Veenvliet, 2014; Lipej B., 2022, pers. comm.). The population of H. arborea inhabiting the Natural Reserve is thought to be of both local and regional importance for the conservation and future viability of the species in Slovenian Istria (Senič et al., 2024). In 2014, the population of calling males was estimated to be about 50, despite the presence of alien fish species, such as Gambusia holbrookii Girard 1859 and Lepomis gibbosus (Linnaeus 1758) (Veenvliet and Kus Veenvliet, 2014). However, the interior of the freshwater part of the Nature reserve is closed for any non-essential visits. The reserve’s primary focus is bird conservation and is considered as an important nesting, wintering, and migration site (Mozetič and Lipej, 2014). The peak of the nesting season coincides with the breeding season of H. arborea, so it is impossible to sample the frogs without significantly disturbing the nesting of the birds. Therefore, we tested the method with artificial shelters in the transition zone between the freshwater and terrestrial habitats along the naturalistic-educational trail (Fig. 1) to minimize the disturbance.
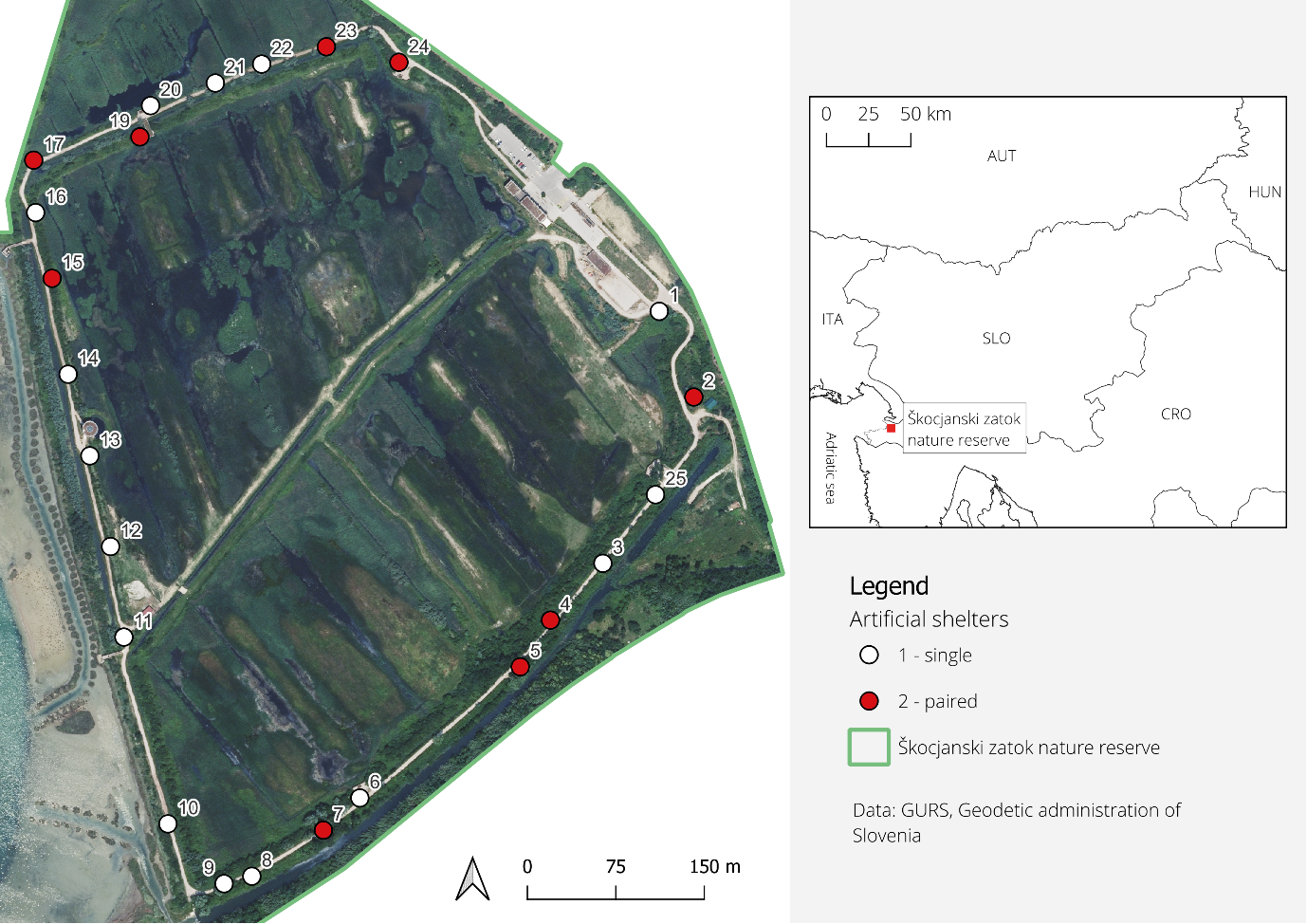
Artificial shelters were designed based on previous research on the genus Hyla (Boughton et al., 2000; Bartareau, 2004; Glorioso and Waddle, 2014; do Vale et al., 2018; Suriyamongkol et al., 2021). We used grey PVC pipes with a white interior, 60 cm in length, and with diameters of either 40 mm or 50 mm. The choice of 50-mm diameter shelters was based on the assumption that H. arborea is relatively large in size among the Hylidae (comparable to Dryophytes cinereus (Schneider, 1799) and H. molleri); following designs of Zacharow et al. (2003) and do Vale et al. (2018). However, 40-mm shelters were also included, as some studies (Bartareau, 2004; Boughton et al., 2000) indicate that Hyla species may occupy narrower pipe diameters (i.e., 13–38 mm).
Shelters were installed at 24 permanent sites spaced 30 to 180 m apart (Fig. 1). They were mounted vertically on trees, in direct contact with the woody surface, with the opening facing upwards and positioned approximately 150 cm above ground level (Fig. 2). Each shelter was sealed at the bottom and filled with water up to a drainage hole located at 10 cm height to ensure high humidity and prevent overflow. At 15 sites, a single 50-mm shelter was installed (referred to as single-shelter sites; Fig. 2A), while at nine sites, two shelters were placed in close proximity (referred to as paired-shelter sites; Fig. 2B): one with a 40-mm and the other with a 50-mm diameter. This study design allowed us to independently assess the suitability of 50-mm shelters for capturing H. arborea as well as to examine shelter diameter occupancy under standardized conditions. Shelters were installed in May 2022 and, following an adaptation period, monitoring was conducted from July 2022 to December 2024. During each inspection, the presence and number of H. arborea individuals in each shelter were recorded (Fig. 2C) and each frog was photographed. Snout–vent length (SVL) of frogs found in paired shelters was measured using digital callipers to the nearest 0.1 mm except during occasional adverse weather conditions. After handling, frogs were returned to their respective shelters. For purpose of this study, frogs were not individually identified; therefore, numbers presented in results refer to frog captures, and not necessarily to individual frogs.

Capture counts of H. arborea were pooled regardless of sex or developmental stage; however, we performed statistical analysis separately for 15 single shelter sites and nine paired shelter sites. We calculated the frequency of frog occurrence (FO) as i) the number of days with at least one frog present in the shelters divided by the number of monitored days, and ii) the number of shelters with frogs present divided by the number of all monitored shelters. For single shelter sites, we calculated the mean number of captures per shelter for all years combined and for each monitoring year separately. Corresponding 95% confidence intervals were derived using the Poisson distribution for count data while ensuring the lower confidence limit did not fall below zero. Given the sufficient sample size (N > 100), the normal approximation method was applied using the formula: (Eq. 1), where λ is the expected value (mean) of the sample Poisson distribution and n is the number of monitored shelters (Bégaud et al., 2005).
For shelter diameter occupancy analysis, we used only the days when at least one frog was present in order to reduce the effect of double-zero observations in the comparison (i.e., no frogs captured in any of the 18 shelters; Zuur et al., 2007). We calculated the mean number of captures in each shelter diameter and corresponding 95% confidence intervals. Confidence intervals were derived using the Poisson distribution for count data using the normal approximation method (Eq. 1; Bégaud et al., 2005). We tested whether the number of frogs captured per shelter and their measured body size follow the normal distribution using Shapiro–Wilk test, with the significance level set at P < 0.05. Both data sets violated normality assumptions, and non-parametric tests were applied (Dytham, 2011). The difference in the number of captures between shelter types was assessed using Wilcoxon matched-pairs signed-rank test due to the parallel placement and simultaneous monitoring of shelter pairs. Due to an extensive period of missing data for 2022, which was covered from July to December, a paired Wilcoxon test was additionally applied to the 2023–2024 data. Furthermore, we assessed shelter selection in relation to frog’s body size using a Wilcoxon signed-rank test. All statistical analyses were performed in R (version 4.4.2; R Core Team, 2024).
A total of 44 monitoring days of all 24 sites were conducted, of which six days (13.6%) were between July and December 2022, 13 (29.5%) between January and December 2023 and 25 (56.8%) between January and December 2024 (Table 1). The total number of captures was 338, with the number of captures exceeding 100 in each year for all shelters combined (Table 1).
| Period | Diam. (mm) | Monitoring occasions | Captures | FO (%) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total (N) | Mean ± SD per shelter | 95% CI | |||||||||||||||||
| Single shelters | |||||||||||||||||||
| 2022 | 50 | 6 | 71 | 0.789 ± 1.590 | 0.605–0.972 | 27.8% | |||||||||||||
| 2023 | 50 | 13 | 47 | 0.241 ± 0.805 | 0.172–0.310 | 12.8% | |||||||||||||
| 2024 | 50 | 25 | 52 | 0.139 ± 0.459 | 0.101–0.176 | 9.9% | |||||||||||||
| All years | 50 | 44 | 170 | 0.258 ± 0.836 | 0.219–0.296 | 13.2% | |||||||||||||
| Paired shelters | |||||||||||||||||||
| All years | 40 | 44 (25*) | 104 | 0.462 ± 1.246 | 0.373–0.551 | 22.2% | |||||||||||||
| 50 | 44 (25*) | 64 | 0.284 ± 0.737 | 0.215–0.354** | 17.3% | ||||||||||||||
| 2023-24 | 40 | 38 (19*) | 83 | 0.485 ± 1.087 | 0.381–0.590 | 24.0% | |||||||||||||
| 50 | 38 (19*) | 49 | 0.287 ± 0.673 | 0.206–0.367** | 19.3% | ||||||||||||||
| * sample size after removal of double-zero occasions, on which the Mean ± SD, 95% CI and FO (%) were calculated (see Materials and methods). | |||||||||||||||||||
| ** statistical significance at p < 0.05. | |||||||||||||||||||
At single shelter sites, frogs were detected on 27 of 44 monitoring days (61.4%) and in 87 of 660 monitored shelters overall (13.2%). Of the 87 occupied shelters, single captures were most numerous (Table 2), that is, in 49 instances (56.3%) only one frog resided in a shelter and in 16 instances (18.4%) two frogs occupied the shelter. Occasions with three or four frogs residing in a single shelter represented 19.5% (17 out of 87), while in the remaining occasions (5 out 87; 5.7%) there were five, six, or seven frogs found in one shelter. Across all years the mean number of frog captures in single shelter sites was 0.258 per shelter with 95 % confidence interval of 0.219 – 0.296 (Table 1). The mean value differed between years, with the 2022 mean frogs captured per shelter (0.789) being 3.2- to 5.7-fold greater than in 2023 (0.241) and 2024 (0.129), respectively (Table 1). Similarly, the frequency of occurrence per shelter was greater in 2022 compared to 2023 and 2024 (Table 1).
| Ind / shelter (N) | 50-mm single | 40-mm paired | 50-mm paired | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Freq (N) | % | Freq (N) | % | Freq (N) | % | ||||||||||||||
| 0 | 573 | 86.8 | 346 (175*) | 87.4 (77.8*) | 357 (186*) | 90.2 (82.7*) | |||||||||||||
| 1 | 49 | 7.4 | 28 | 7.1 (12.4*) | 23 | 5.8 (10.2*) | |||||||||||||
| 2 | 16 | 2.4 | 10 | 2.5 (4.4*) | 10 | 2.5 (4.4*) | |||||||||||||
| 3 | 9 | 1.4 | 4 | 1.0 (1.8*) | 4 | 1.0 (1.8*) | |||||||||||||
| 4 | 8 | 1.2 | 3 | 0.8 (1.3*) | 1 | 0.3 (0.4*) | |||||||||||||
| 5 | 1 | 0.2 | 4 | 1.0 (1.8*) | 1 | 0.3 (0.4*) | |||||||||||||
| 6 | 3 | 0.5 | |||||||||||||||||
| 7 | 1 | 0.2 | |||||||||||||||||
| 12 | 1 | 0.3 (0.4*) | |||||||||||||||||
| Total | 660 | 100 | 396 (225*) | 100 | 396 (225*) | 100 | |||||||||||||
| * results after removal of double-zero occasions (see Materials and methods). | |||||||||||||||||||
The overall frequency of occurrence in paired shelter sites was 56.8% (25 out of 44 days, Table 1). There were 19 occasions (43.2%) with no frogs present in any of the paired shelters, i.e., double-zero occasions, that were removed from further analysis. The frequency of occurrence per shelter was 22.2% (50 out of 225) in 40-mm and 17.3% (39 out of 225) in 50-mm shelters (Table 1). Both shelter types commonly captured one or two individuals (76.0% in 40-mm, 84.6% in 50-mm shelters), followed by three, four or five (22.0% in 40-mm, 15.4% in 50-mm shelters). Additionally, on one occasion (2.0%) in 2022, there were 12 individuals captured in a 40-mm shelter. The number of frogs captured per shelter deviated from normality both for pooled paired shelter sites (W = 0.410, P < 0.001), as well as separated by the shelter diameter (40-mm: W = 0.417, P < 0.001; 50-mm: W = 0.446, P < 0.001). Overall, the total number of frog captures was 1.6-fold higher in 40-mm shelters (N = 104) compared to the 50-mm shelters (N = 64). A similar pattern was observed for the 2023–2024 only data, with the difference amounting to 1.7-fold (N = 83 in 40-mm, N = 49 in 50-mm shelters). This difference is statistically significant across all years (V = 1223.5, P < 0.05) and for 2023–2024 data (V = 870.5, P < 0.05) and is reflected in mean values and corresponding 95% confidence intervals (Table 1).
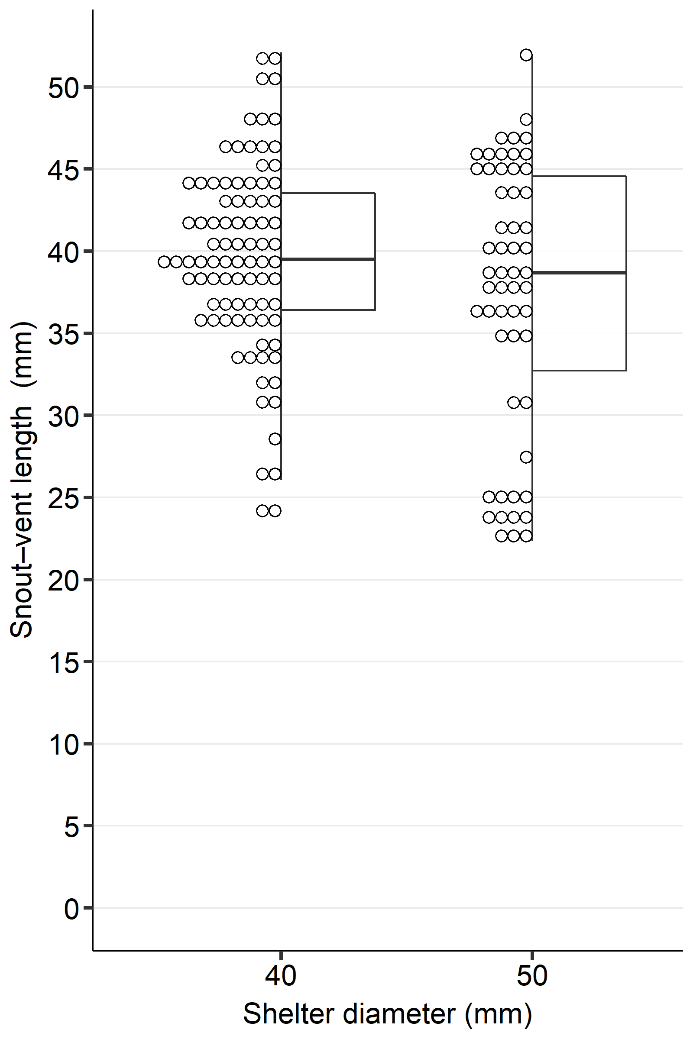
Snout–vent length (SVL) was measured for a total of 142 frog captures at paired shelter sites, while 26 captures were not measured (see Materials and methods). In 40-mm diameter shelters (N = 87), SVL ranged from 24.1 mm to 52.1 mm (mean ± SD = 39.6 ± 5.8 mm; median SVL 39.5 mm; Fig. 3). In 50-mm diameter shelters (N = 55), SVL ranged from 22.4 mm to 51.9 mm (mean ± SD = 37.3 ± 8.3 mm; median = 38.7 mm; Fig. 3). Snout-vent length data deviated from a normal distribution (Shapiro–Wilk test: W = 0.951, P < 0.001), and no significant difference in body size was found between shelter diameters (Wilcoxon rank-sum test: W = 2652, P = 0.278).
This study is the first in which we test the use of artificial shelters for occupancy by H. arborea. So far, in Europe artificial shelters have been used only in one study of the closely related H. molleri in north-western Portugal (do Vale et al., 2018). Other known studies using similar shelters for capturing arboreal hylid species were from North America (for species such as Dryophytes cinereus (Schneider, 1799), D. squirellus (Bosc, 1800), D. femoralis (Bosc, 1800), D. gratiosus (LeConte, 1856), and Osteopilus septentrionalis (Duméril et Bibron, 1841); see Boughton et al., 2000; Campbell et al., 2010; Suriyamongkol et al., 2021) and from South America for genera such as Osteocephalus, Scinax, Trachycephalus, and Aparasphenodon (Ferreira et al., 2012; Pereira-Ribeiro et al., 2017). Trimble and van Aarde (2014) reported the usefulness of this method for capturing arboreal species of the family Hyperoliidae (such as Hyperolius sp. and Afrixalus spp.) from South Africa.
The size of the H. arborea population in Škocjanski zatok Nature Reserve is currently unknown, although it was estimated to be around 50 calling males indicating the presence of a population multiple times larger (Veenvliet and Kus Veenvliet, 2014). In our study, the frequency of frog occurrence per shelter varied between years in single shelters (see Table 1). We hypothesize several reasons underlying this distribution.
Availability of natural shelters provided by the surrounding vegetation may reduce the attractiveness of artificial refuges and thereby influence occupancy (Pittman et al., 2008; Hoffmann et al., 2009; Trimble and van Aarde, 2014). Although we did not quantify vegetation density in different years of the study period, vegetation management, such as cutting along the trails within the Nature Reserve, may have influenced habitat structure and consequently affected the presence or behaviour of the species. There are several anecdotal reports of sightings of H. arborea in the vegetation surrounding the trails, especially in leaf axils of teasel plants Dipsacus fullonum (Lipej B., 2022, pers. comm.).
Notably, a high number of small individuals were recorded in July 2022, suggesting that newly metamorphosed frogs readily occupied the artificial shelters. These individuals made up more than half of all frogs detected in that year (Lužnik, in preparation), indicating a likely successful breeding season and recruitment. In contrast, no such individuals were observed in 2023 and only one in 2024, when mostly larger, adult-sized frogs were recorded. However, it is important to note that there is no clear-cut size threshold distinguishing juveniles from adults in H. arborea. For example, Moravec (1990) reported juveniles reaching up to 33 mm in the season of metamorphosis, but that sexually active males were also of a similar size at breeding sites. In our study, individuals ranged from 22.4 to 52.1 mm in snout–vent length (see below). Frogs captured after the breeding season with sizes between 30.0 and 35.0 mm could not always be reliably assigned to a specific age class. Despite this uncertainty, our findings indicate that the method is suitable for monitoring both juvenile and adult H. arborea. With further development, such as incorporating a mark–recapture approach with regular sampling, it could potentially allow estimation of interannual variation in population structure and abundance (Carlson and Edenhamn, 2000), which remains to be tested in the studied population. One approach to consider is mark-recapture with photo identification, as used in Lukanov et al. (2024). Some of the juveniles recorded early on (in 2022) may have continued using the shelters in subsequent seasons as adults, suggesting a potential habituation to these artificial refuges. Such behavioural adjustment could explain continued but reduced occupancy over time. However, this remains speculative, as we currently lack direct data to confirm long-term individual shelter use; further analyses on this topic are in preparation.
The last possible reason is the interference of researchers and handling of animals (getting them out of the shelter), which could have affected the use of artificial shelters by the species and reduced their numbers (Perry et al., 2011). Frogs tend not to return to the same refuge following disturbances caused by researchers, such as toe-clipping, measuring, or removal from shelter (Boughton et al., 2000). Researchers should consider the varying degrees of disturbance associated with these procedures. Toe-clipping, for example, constitutes a substantial disturbance due to the physical injury it inflicts (Waddle et al., 2008). In contrast, we argue that brief and careful handling for the purpose of photographing and measuring SVL is considerably less invasive. Therefore, we do not consider this type of handling to be a likely explanation for the observed decrease in shelter use (Pittman et al., 2008, do Vale et al., 2018; Hutton et al., 2024).
For artificial shelters, several parameters have been suggested to influence occupancy, such as pipe colour (Ferreira et al., 2012), pipe design (Granatosky and Krysko, 2011), pipe placement (Pittman et al., 2008), pipe orientation (Bartareau, 2004; Ferreira et al., 2012), and pipe diameter (Bartareau, 2004). For this study we used grey PVC pipes with white interior because Ferreira et al. (2012) reported that shelter colour (grey, white or black) did not result in statistical differences in occupancy between pipe shelters. Among environmental conditions, moisture retention and temperature are one of the main factors influencing habitat selection by the study species (Goin, 1958; Boughton et al., 2000; Granatosky and Krysko, 2011). With caps at the bottom of pipes and providing starting level of water to 10 cm height in our pipe design we enabled more suitable environment as suggested by Granatosky and Krysko (2011), while the seasonal variability of ambient/air temperature may have contributed to the observed occupancy patterns and should be examined in future studies.
In addition to shelter characteristics and environmental conditions, several studies have suggested that characteristics of study animals such as sex, life stage (e.g., adult versus juvenile) and body size may influence the use of shelters by tree frogs (Zacharow et al., 2003; Myers et al., 2007; Johnson et al., 2008; Hoffmann et al., 2009). In our study, we did not differentiate individuals by sex or life stage in the analysis. While the presence of vocal sacs is a reliable external trait distinguishing adult males from females in H. arborea, it is not possible to confidently classify all captured individuals, particularly those within the intermediate size range (30.0–35.0 mm), which may include both small adult females and large juveniles (Moravec, 1990; Gvoždík et al., 2008). To eliminate this uncertainty, we would have had to exclude a significant portion of our dataset, which would have further reduced the already limited sample size. However, our results demonstrate that individuals of all sizes used the shelters (see above). We therefore opted to retain the full dataset to better reflect the overall size distribution of shelter occupants.
We acknowledge that capture success varies across studies using different pipe designs or placements (Zacharow et al., 2003; Bartareau, 2004;), suggesting that tree frogs exhibit selectivity in shelter use. Bartareau (2004), who tested three pipe diameters (13 mm / 25 mm / 38 mm), found that juveniles and smaller adults preferred the narrower shelters (13 mm /25 mm). Similarly, Zacharow et al. (2003) tested 19-mm, 51-mm, and 77-mm pipes and observed that smaller frogs (D. squirellus, mean SVL: 28.7 mm; range: 24.0 mm–35.0 mm) favoured 19-mm pipes, while larger individuals (D. cinereus, mean SVL: 45.0 mm; range: 37.0 mm–66.0 mm) also used 51-mm pipes, but only half as often as 19-mm pipes. Interestingly, in Bartareau’s (2004) study, D. cinereus did not use 38-mm pipes at all. do Vale et al. (2018) reported H. molleri occupying 44-mm pipes, with most individuals measuring 35.0 mm–45.0 mm. Hyla arborea is comparable in body size to H. molleri and D. cinereus, and in our study, frogs showed a consistent and statistically significant preference for narrower shelters (40 mm) over wider ones (50 mm), with 1.6 times greater use across three years (2022–2024), and 1.7 times greater use in 2023–2024 when monitoring was conducted year-round. However, no statistically significant relationship was found between body size and shelter diameter occupancy, as SVL ranges were similar between the two pipe types (24.1 mm–52.1 mm in 40-mm shelters vs. 22.4 mm–51.9 mm in 50-mm shelters). We hypothesise that the observed occupancy in narrow shelters could reflect a general tendency toward microhabitats with more favourable microclimatic conditions, particularly improved moisture retention (Granatosky and Krysko, 2011). Additionally, narrower shelters may offer more enclosed space, potentially reducing visibility and access to predators (Johnson, 2005). Forks (2015) suggested that PVC shelters may serve as effective refuges for juveniles shortly after metamorphosis, which presents an interesting direction for future research. Based on our observations, we also propose testing whether H. arborea would use artificial shelters with even smaller pipe diameters, as this could further clarify microhabitat preferences across developmental stages.
Our results demonstrate that PVC shelters offer a practical and effective non-invasive method for monitoring H. arborea in sensitive habitats such as wetlands with high conservation value, for both juvenile and adult individuals. Compared to traditional more invasive techniques, this passive sampling approach significantly improves detectability – especially outside the breeding season – and minimizes disturbance to non-target species, such as protected bird populations or sensitive vegetation. These advantages make it particularly suitable for long-term monitoring in ecologically sensitive or physically less accessible areas.
Throughout three consecutive years, we observed consistent occupancy in narrower (40-mm) shelters over wider (50-mm) ones, although frog body size did not differ significantly between shelter types. The observed pattern may reflect opportunistic use by juveniles and a potential preference for more enclosed microhabitats among adults – possibly due to favourable microclimatic conditions or lower predation risk. Despite an overall decrease in occupancy over time, this trend may be influenced by a combination of factors, including sampling biases, vegetation dynamics, demographic fluctuations typical of H. arborea populations, and possibly habituation or disturbance tolerance. Notably, artificial shelters provided refuge to both juvenile and adult individuals, allowing us to detect shifts in population structure among years. Future research should explore shelter use across a wider range of pipe diameters to clarify age- or size-based preferences, and integrate demographic and environmental data to improve the ecological interpretation of occupancy trends. Given the success and low invasiveness of this method, we recommend its application in other protected or sensitive amphibian habitats where traditional methods may be impractical or undesirable.
We would like to thank the Škocjanski zatok Nature Reserve (DOPPS – BirdLife Slovenia) for granting access to the study area and for supporting our work throughout the monitoring period. Special thanks go to the reserve staff for their logistical support and field assistance. We are especially grateful to Elena Shklyarnik, Marija Jakovljević, Maša Možina, Nina Lončarević, and Katja Adam for their invaluable help with fieldwork and data collection. We also thank Nina Lončarević for preparing the map used in this study. The research was conducted under the permission of the Slovenian Environment Agency (ARSO), permit no. 35601-40/2021-5, and Ministry of the Environment and Spatial Planning, permits no. 35606-32/2023-2550-4, 35606-22/2024-2. We thank C. Kenneth Dodd Jr. and an anonymous reviewer for their constructive comments that improved the manuscript.
Agasyan, A., Avci, A., Tuniyev, B., Andrén, C., Cogălniceanu, D., Crnobrnja-Isailović, J., Poboljšaj, K., Halpern, B., Wilkinson, J., Ananjeva, N.B., Üzüm, N., Orlov, N.L., Lymberakis, P., Podloucky, R., Litvinchuk, S. (2024): Hyla arborea. In The IUCN Red List of Threatened Species 2024.
Alford, R.A., Richards, S.J. (1999): Global amphibian declines: A problem in applied ecology. Annu. Rev. Ecol. Evol. Syst. 30: 135-165.
Bartareau, T.M. (2004): PVC pipe diameter influences the species and sizes of treefrogs captured in a Florida coastal scrub community. Herpetol. Rev. 35: 150-152.
Bégaud, B., Martin, K., Abouelfath, A., Tubert-Bitter, P., Moore, N., Moride, Y. (2005): An easy to use method to approximate Poisson confidence limits. Eur. J. Epidemiol. 20: 213-216.
Boughton, R.G., Staiger, J., Franz, R. (2000): Use of PVC pipe refugia as a sampling technique for hylid treefrogs. Am. Midl. Nat. 144: 168-177.
Campbell, K.R., Campbell, T.S., Johnson, S.A. (2010): The use of PVC pipe refugia to evaluate spatial and temporal distributions of native and introduced treefrogs. Fla. Sci. 73: 78-88.
Carlson, A., Edenhamn, P. (2000): Extinction dynamics and the regional persistence of a tree frog metapopulation. Proc. R. Soc. B Biol. Sci. 267: 1311-1313.
Cowan, M.A., Callan, M.N., Watson, M.J., Watson, D.M., Doherty, T.S., Michael, D.R., Dunlop, J.A., Turner, J.M., Moore, H.A., Watchorn, D.J., Nimmo, D.G. (2021): Artificial refuges for wildlife conservation: What is the state of the science? Biol. Rev. 96: 2735-2754.
Cruickshank, S.S., Bergamini, A., Schmidt, B.R. (2021): Estimation of breeding probability can make monitoring data more revealing: A case study of amphibians. Ecol. Appl. 31.
do Vale, R.L., Torres, I., Gomes, S., Fonseca, C., Ferreira, E. (2018): Ecological preferences of Hyla molleri in the colonisation of arboreal refuges in a human-shaped wetland. Amphib.-Reptil. 39: 51-61.
Dytham, C. (2011): Choosing and using statistics: A biologist’s guide (3rd ed.). Wiley-Blackwell.
Falaschi, M., Manenti, R., Thuiller, W., Ficetola, G.F. (2019): Continental-scale determinants of population trends in European amphibians and reptiles. Glob. Change Biol. 25: 3504-3515.
Ferreira, E., Gomes Rocha, R., Malvasio, A., Fonseca, C. (2012): Short notes pipe refuge occupancy by herpetofauna in the Amazonia/Cerrado ecotone. Herpetol. J. 22: 59-62.
Forks, K. (2015): Use of PVC pipes of different diameters as artificial refuges by green tree frogs (Hyla cinerea) in Texas. Unpublished Undergraduate Research Scholars Thesis. Texas A&M University.
Glorioso, B.M., Waddle, J.H. (2014): A review of pipe and bamboo artificial refugia as sampling tools in anuran studies. Herpetol. Conserv. Biol. 9: 609-625.
Goin, O.B. (1958): A comparison of the nonbreeding habits of two treefrogs, Hyla squirella and Hyla cinerae. Q. J. Fla. Acad. Sci. 21: 49-60.
Granatosky, M.C., Krysko, K.L. (2011): Ontogenetic behavioral shifts in habitat utilization of treefrogs (Hylidae) in North-central Florida. Reptil. Amphib. 18: 194-201.
Gvoždík, V., Moravec, J.Í., Kratochvíl, L. (2008): Geographic morphological variation in parapatric Western Palearctic tree frogs, Hyla arborea and Hyla savignyi: Are related species similarly affected by climatic conditions? Biol. J. Linn. Soc. 95: 539-556.
Hoffmann, K.E., Johnson, S.A., McGarrity, M.E. (2009): Interspecific variation in use of polyvinyl chloride (PVC) pipe refuges by hylid treefrogs: A potential source of capture bias. Herpetol. Rev. 40: 423-426.
Houlahan, J.E., Findlay, C.S., Schmidt, B.R., Meyer, A.H., Kuzmin, S.L., Kuzmink, S.L. (2000): Quantitative evidence for global amphibian population declines. Nature, 404: 752-755.
Hutton, J.M., Warne, R., Macedo, A.D. (2024): Factors influencing the occupancy and detection of nonbreeding Hyla chrysoscelis within artificial polyvinyl chloride refugia. Herpetologica, 80: 221-233.
Johnson, J.R. (2005): A novel arboreal pipe-trap designed to capture the gray treefrog (Hyla versicolor). Herpetol. Rev. 36: 274-276.
Johnson, J.R., Mahan, R.D., Semlitsch, R.D. (2008): Seasonal terrestrial microhabitat use by gray treefrogs (Hyla versicolor) in Missouri oak-hickory forests. Herpetologica, 64: 259-269.
Kovács, É.-H., Sas, I., Covaciu-Marcov, S.-D., Hartel, T., Cupsa, D., Groza, M. (2007): Seasonal variation in the diet of a population of Hyla arborea from Romania. Amphib.-Reptil. 28: 485-491.
Liner, A.E., Smith, L.L., Golladay, S.W., Castleberry, S.B., Gibbons, J.W. (2008): Amphibian distributions within three types of isolated wetlands in Southwest Georgia. Am. Midl. Nat. 160: 69-81.
Luedtke, J.A., Chanson, J., Neam, K., Hobin, L., Maciel, A.O., Catenazzi, A., Borzée, A., Hamidy, A., Aowphol, A., Jean, A., Sosa-Bartuano, Á., Fong G.A., de Silva, A., Fouquet, A., Angulo, A., Kidov, A.A., Muñoz Saravia, A., Diesmos, A.C., Tominaga, A., … Stuart, S.N. (2023): Ongoing declines for the world’s amphibians in the face of emerging threats. Nature, 622: 308-314.
Lukanov, S., Kolev, A., Dyugmedzhiev, A., Slavchev, M. (2024): The lateral stripe - a reliable way for software assisted individual identification for Hyla arborea. Acta Herpetol. 19: 69-73.
Mahan, R.D., Johnson, J.R. (2007): Diet of the gray treefrog (Hyla versicolor) in relation to foraging site location. J. Herpetol. 41: 16-23.
Marsh, D. M., Trenham, P.C. (2001): Metapopulation dynamics and amphibian conservation. Conserv. Biol. 15: 40-49.
McGhee, J.D. (2020): Assessing summer pond and lake inlet use by gray treefrogs (Hyla versicolor/chrysoscelis complex) using PVC pipe traps in Northwest Missouri. Trans. Mo. Acad. Sci. 48: 17-21.
Ministry of Environment, Spatial Planning and Energy (2002): Pravilnik o uvrstitvi ogroženih rastlinskih in živalskih vrst v rdeči seznam. Official Gazette of Republic of Slovenia, 82/2002. https://www.uradni-list.si/_pdf/2002/Ur/u2002082.pdf [Rules on the inclusion of endangered plant and animal species in the Red List. In Slovenian]
Moravec, J. (1990): Postmetamorphic growth in the European tree frog (Hyla arborea). Acta Univ. Carolin. Biol. 34, 359-370.
Moulton, C.A., Fleming, W.J., Nerney, B.R. (2009): The use of PVC pipes to capture hylid frogs. Herpetol. Rev. 27: 186-187.
Mozetič, B., Lipej, B. (2014): Zeleno srce Kopra - vodnik po Naravnem rezervatu Škocjanski zatok (2nd ed.). Društvo za opazovanje in proučevanje ptic Slovenije DOPPS. (The Green Heart of Koper - A Guide to the Škocjanski Zatok Nature Reserve (2nd ed.). Society for the Observation and Study of Birds of Slovenia (DOPPS)).
Myers, C.H., Eigner, L., Harris, J. A., Hilman, R., Johnson, M.D., Kalinowski, R., Muir, J.J., Reyes, M., Tucci, L.E. (2007): A Comparison of ground-based and tree-based polyvinyl chloride pipe refugia for capturing Pseudacris regilla in Northwestern California. Northwest. Nat. 147-154.
Pellet, J., Hoehn, S., Perrin, N. (2004): Multiscale determinants of tree frog (Hyla arborea L.) calling ponds in western Switzerland. Biodivers. Conserv. 13: 2227-2235.
Pellet, J., Rechsteiner, L., Skrivervik, A.K., Zürcher, J.-F., Perrin, N. (2006): Use of the harmonic direction finder to study the terrestrial habitats of the European tree frog (Hyla arborea). Amphib.-Reptil., 27: 138-142.
Pereira-Ribeiro, J., Ferreguetti, Á.C., Bergallo, H.G., Rocha, C.F.D. (2017): Use of polyvinyl chloride pipes (PVC) as potential artificial shelters for amphibians in a coastal plain forest of southeastern Brazil. J. Coast. Conserv. 21: 327-331.
Perry, G., Wallace, M.C., Perry, D., Curzer, H., Muhlberger, P. (2011): Toe clipping of amphibians and reptiles: Science, ethics, and the law. J. Herpetol. 45: 547-555.
Pittman, S.E., Jendrek, A.L., Price, S.J., Dorcas, M.E. (2008): Habitat selection and site fidelity of Cope’s Gray Treefrog (Hyla chrysoscelis) at the aquatic-terrestrial ecotone. J. Herpetol. 42: 378-385.
R Core Team. (2024): R: A language and environment for statistical computing. R Foundation for Statistical Computing.
Schurbon, J. M., Fauth, J. E. (2003): Effects of prescribed burning on amphibian diversity in a Southeastern U.S. National forest. Conserv. Biol. 17: 1338-1349.
Semlitsch, R. D., Bodie, J. R. (2003): Biological criteria for buffer zones around wetlands and riparian habitats for Amphibians and Reptiles. Conserv. Biol. 17: 1219-1228.
Senič, M., Adam, K., Jakovljević, M., Jugovic, J., Lončarević, N., Možina, M., Rečnik, K., Shklyarnik, E., Zupan, S., & Lužnik, M. (2024): Using artificial shelters and photo identification for monitoring the European tree frog (Hyla arborea) in Škocjanski zatok nature reserve. In: Book of abstracts: 2nd Life Amphicon International Conference: amphibian conservation and habitat restoration, 2024.Bolčina, A., Poboljšaj, K., Levstek, S., Vertič, Ž., Kotnik, T., Eds, Podsreda, Kozjanski park.
Suriyamongkol, T., Forks, K., Villamizar-Gomez, A., Wang, H. H., Grant, W. E., Forstner, M. R. J., Mali, I. (2021): A simple conservation tool to aid restoration of amphibians following high-severity wildfires: Use of PVC pipes by green tree frogs (Hyla cinerea) in central Texas, USA. Diversity, 13.
Tanadini, L. G., Schmidt, B. R. (2011): Population size influences amphibian detection probability: Implications for biodiversity monitoring programs. PLOS ONE, 6.
Trimble, M. J., van Aarde, R. J. (2014): A note on polyvinyl chloride (PVC) pipe traps for sampling vegetation-dwelling frogs in South Africa. Afr. J. Ecol. 52: 381-383.
Van Buskirk, J. (2005): Local and landscape influence on amphibian occurrence and abundance. Ecology, 86: 1936-1947.
Veenvliet, P., Kus Veenvliet, J. (2014): Monitoring dvoživk in plazilcev v Naravnem rezervatu Škocjanski zatok-končno poročilo. Nova vas, Zavod Symbiosis.
Waddle, J. H., Rice, K. G., Mazzotti, F. J., Percival, H. F. (2008): Modelling the effect of toe clipping on treefrog survival: Beyond the return rate. J. Herpetol. 42: 467-473.
Wyatt, J. L., Forys, E. A. (2004): Conservation implications of predation by Cuban treefrogs (Osteopilus septentrionalis) on native hylids in Florida. Southeast. Nat. 3: 695-700.
Zacharow, M., Barichivich, W. J., Dodd, C. K., Jr. (2003): Using ground-placed PVC pipes to monitor hylid treefrogs: Capture biases. Southeast. Nat. 2: 575-590.
Zuur, A. F., Ieno, E. N., Smith, G. M. (2007): Analysing ecological data. Springer New York, NY.