Submitted on: 2025, 27th April; revised on: 2025, 21st October; accepted on: 2025, 22nd October
Hierarchical response to kairomones of predator based on experience acquired through learning
Department of Zoology, Karnatak University, Dharwad-580 003, Karnataka State, India
E-mail:santoshmogali21@gmail.com, santoshmogali@rediffmail.com
Abstract. This study investigated how prior experience modulates the anti-predator behavioral responses of Duttaphrynus melanostictus tadpoles to kairomones from the predatory tadpole Hoplobatrachus tigerinus. I compared the responses of four distinct categories of tadpoles; 1) predator-naïve (laboratory-born), 2) indirect predator-experienced (short-term exposure to caged predator cues), 3) direct predator-experienced (short-term direct encounters), and 4) wild-caught (long-term natural experience). A stimulus solution (kairomones) from the predator was used to simulate predation risk. The results showed that tadpoles of D. melanostictus from all experience groups exhibit antipredator behavioral responses, i.e., overall reduced swimming and less time spent swimming, but with a higher burst speed in response to water-borne kairomonal cues of predators. Crucially, the intensity of these antipredator behavioral responses was strongly dependent on experience, following a clear hierarchical gradient: Wild-caught > direct-predator experienced > indirect-predator experienced > predator-naïve > control. The significant, albeit low-level, response of predator-naïve tadpoles indicates that predator recognition is innate. However, the enhanced antipredator behavior of wild-caught tadpoles compared to predator-naïve or direct or indirect predator-experienced tadpoles suggests that learning and cumulative experience are involved. A combination of both innate and learned behaviors could allow tadpoles of D. melanostictus to calibrate their defensive investment necessary for survival.
Keywords. Anurans, behavioral response, prey-predator interactions, reduced activity, swimming speed, tadpoles.
Index
Behavioral responses of D. melanostictus tadpoles to kairomones of H. tigerinus tadpoles
Intensity of defense behaviors in D. melanostictus tadpoles to kairomones of H. tigerinus tadpoles
Predator-prey interactions play a crucial role in shaping the life-history strategies of animals. In both aquatic and terrestrial ecosystems, predation provides selection pressure that forces prey to maximize their fitness by recognizing and avoiding predators. Predators can impact the behavior, morphology, and life history of prey (Lima and Dill, 1990; Laforsch and Tollrian, 2004; Ferrari et al., 2010; Smith et al., 2010; Gazzola et al., 2024; Saidapur, 2025). For predator recognition, prey animals may use a wide range of cues such as visual, acoustic, electric, tactile, disturbance, chemical, or a combination thereof (Amo et al., 2004; McCormick and Manassa, 2007; Ferrari et al., 2010; Mogali et al., 2011, 2012; Batabyal et al., 2014; Landeira-Dabarca et al., 2019; Saidapur, 2025). In aquatic predator-prey systems, chemical cues are much more efficient in complex, murky ecosystems and are usually detected faster, earlier, and over larger distances than visual cues (Chivers et al., 1996; Mathis and Vincent, 2000; Ferrari et al., 2010).
Anuran larvae are an excellent model system for studying predator-prey interactions because tadpoles are highly vulnerable to aquatic predators (Heyer et al., 1975). Previous studies suggest that most species of anuran tadpoles assess predation risk using chemosensory mechanisms before responding with defense behaviors (Ferrari et al., 2010; Mogali et al., 2012; Saidapur, 2025). Individuals respond to alarm cues released by injured prey, kairomones of predators, and dietary cues that alter behavior to escape predation (Schoeppner and Relyea, 2005, 2009; Scherer and Smee, 2016; Saidapur, 2025). Previous studies have also revealed that anuran larvae exhibit a variety of antipredator responses to chemical cues that can be generally grouped into strategies to avoid predators and/ or strategies to escape from predation (Schmidt and Amezquita, 2001; Hossie and Murray, 2010). These responses include overall decreased activity (Saidapur, 2025), increased hiding and aggregation (Spieler and Linsenmair, 1999; Hossie and Murray, 2010), and defensive behaviors (increased swimming, Mogali et al., 2021).
In aquatic environments, predators may release various types of chemical cues, with kairomones (the odors of predator) being the most common. There is evidence that kairomones trigger antipredator behavior in a wide range of prey animals (Kats and Dill, 1998; Schoeppner and Relyea, 2005; Ferrari et al., 2010). Kairomones are generally considered to be chemical signatures of predators. However, some studies involving starved predators have shown that kairomones may not elicit antipredator responses in some prey taxa (Crowl and Covich, 1990; Stirling, 1995). In contrast, other research focused on larval anuran has demonstrated that kairomones can induce strong antipredator behavioral responses (Petranka and Hayes, 1998; Van Buskirk and Arioli, 2002; Schoeppner and Relyea, 2005, 2009; Gyssels and Stoks, 2006; Mogali et al., 2011).
The Asian common toad, Duttaphrynus melanostictus (Schneider, 1799), is widely distributed throughout India. In Southern India, during the early monsoon season, D. melanostictus generally breeds in temporary water bodies alongside other coexisting anuran species (Saidapur, 2001; Gramapurohit and Radder, 2012). The ephemeral ponds that host herbivorous tadpoles of D. melanostictus are also habit for a variety of predators. These include invertebrates (e.g., dragonfly and damselfly larvae, beetles, crabs, water boatmen) and both omnivorous tadpoles, Euphlyctis cyanophlyctis and carnivorous tadpoles, Hoplobatrachus tigerinus (Mogali et al., 2023a, b, c). During my regular field visits, I observed that the herbivorous tadpoles of D. melanostictus were primarily preyed upon by H. tigerinus. In studies focused on predator-prey interaction among tadpoles, most researchers have used aquatic insects, fish, or salamanders as predators, often overlooking other aquatic predators, especially anuran tadpole predators (Chivers and Mirza, 2001; Mathis, 2003). Very few studies have investigated the impact of carnivorous tadpole predators on the behavioral responses of herbivorous tadpoles (Saidapur, 2025). Most researchers have studied the behavioral responses of prey tadpoles by using either only laboratory reared (predator-naïve) or laboratory reared tadpoles with short-term direct or indirect experience with predators (predator-experienced) or tadpoles with long-term experience with predators in natural water bodies (wild-caught) (Semilitsch and Reyer, 1992; Laurila et al., 1997; Mogali et al., 2012, 2023c). In this study, I investigated the behavioral responses of different categories of D. melanostictus tadpoles: predator-naïve (PN), indirect predator-experienced (IPE), direct predator-experienced (DPE), and wild-caught (WC) tadpoles. I exposed them to stimulus solutions (kairomones) from a predator, H. tigerinus, as such studies are relatively rare. I hypothesized that all four categories of prey tadpoles would primarily exhibit antipredator behavioral responses to the kairomones of H. tigerinus. Additionally, I expected to find variation in the antipredator behavioral responses among the tadpole categories. Specifically, I expected that wild-caught tadpoles would display the strongest antipredator responses compared to both predator-experienced (direct and indirect) and predator-naïve tadpoles.
Three egg clutches of Duttaphrynus melanostictus were collected from a temporary pond on the Karnatak University Campus, Dharwad, Karnataka State, India (15º27’N, 75º05’E, 750 m a.s.l.), during the early monsoon. Each clutch was placed in a separate 1 L plastic container filled with aged tap water and immediately transported to the laboratory. Here, the clutches were transferred separately in plastic tubs (32 cm in diameter and 14 cm in depth) containing 5 L of aged (dechlorinated) tap water. The eggs from all three clutches hatched synchronously at stage 19 (Gosner, 1960) the following day. Tadpoles from all three clutches, totaling 300 (100 tadpoles per clutch) were mixed and reared together in a glass aquarium (90 × 30 × 15 cm) containing 20 L of aged tap. In this way, two such stocks were maintained, resulting in a total of 600 tadpoles. Five days prior to the experimental trials, predatory tadpoles of H. tigerinus (Gosner stages 32–33; mean total length 37.80 ± 1.26 mm, N = 50) and prey tadpoles of D. melanostictus (Gosner stages 32–33; mean total length 27.32 ± 1.95 mm, N = 50) were collected from the same pond where the D. melanostictus eggs were collected. H. tigerinus tadpoles were reared in plastic tubs (19 cm in diameter and 7 cm in depth) in 0.5 L of aged tap water to avoid cannibalism. The D. melanostictus tadpoles were fed boiled spinach, while H. tigerinus tadpoles were fed tadpoles of D. melanostictus. For experimental categories classification, tadpoles of D. melanostictus reared in the laboratory from the egg stage were designated as predator-naïve (PN); those reared in the laboratory and exposed for a short-term to caged predators were designated as indirect predator-experienced (IPE); tadpoles reared in the laboratory and exposed directly to predators were designated as direct predator-experienced (DPE). Tadpoles of D. melanostictus collected from natural water bodies were designated as wild-caught (WC). The behavioral responses of all four categories of test D. melanostictus tadpoles were studied by exposing them to a “stimulus solution”, which consisted of kairomones of the predatory tadpoles, H. tigerinus.
Tadpoles of H. tigerinus were placed individually in separate plastic tubs (N = 20 tubs; 19 cm in diameter and 7 cm in depth) containing 200 mL of aged tap water without food for 96 h. This procedure resulted in a solution with only kairomones. After 96 h of starvation period, the predators were removed from the tubs, and the stimulus solution was filtered to remove any small quantities of fecal matter. The filtered solution, rich in kairomones was used immediately for the experimental trials.
(1) Predator-naïve tadpoles (PN): Duttaphrynus melanostictus tadpoles (Gosner stages 32–33; mean total length 27.49 ± 1.80 mm; N = 25) were placed in plastic tubs (32 cm in diameter and 14 cm in depth) with 3 L of aged tap water. At the center of the rearing tub, a small empty plastic tub (19 cm in diameter × 7 cm in height) wrapped with cheese cloth was placed. These tadpoles were raised in the absence of predators from the time of hatching. Two duplicate tubs were maintained.
(2) Indirect predator-experienced tadpoles (IPE): D. melanostictus tadpoles (Gosner stages32–33; mean total length 27.49 ± 1.80 mm; N = 25) were also placed in similar plastic tubs containing 3 L of aged tap water. At the center, a small plastic tub (19 cm in diameter × 7 cm in height) with perforations (1.2 mm2 holes), wrapped with cheese cloth, and housing a single starved tadpole of H. tigerinus (Gosner stages 32–33) was introduced for 8 h. Although the test tadpoles outside the cage had no direct contact with the predators, they were exposed to chemical cues (kairomones) released by the starved predator inside the cage. Two duplicate tubs were maintained.
(3) Direct predator-experienced tadpoles (DPE): the rearing setup for this group was identical to the previous setups, except that a single starved tadpole of H. tigerinus (Gosner stages 32–33) was directly introduced into the rearing tubs from 0900 to 1700 h. On average, the predator consumed 5 ± 0.3 and injured 4 ± 0.5 tadpoles during the 8–h period. After this time, the predator and the injured tadpoles were removed. The surviving uninjured that had experienced a direct predator were then used for trials on the subsequent day. Two duplicate tubs were maintained.
(4) Wild-caught tadpoles (WC): a total of 25 wild-caught tadpoles of D. melanostictus (Gosner stages 32–33; mean total length 27.32 ± 1.95 mm, N = 25) were placed in plastic tubs (32 cm in diameter and 14 cm in depth) containing 3 L of aged tap water with an empty cage at the center. In their natural temporary water bodies, D. melanostictus tadpoles have lived more than 15 days with various aquatic predators, potentially including H. tigerinus tadpoles. Two duplicate tubs were maintained.
The behavioral responses to predator kairomones of the four tested categories of tadpoles were recorded by placing single test subjects in a rectangular glass test tank (28 × 15 × 15 cm) containing 600 mL of aged tap water. A handycam (Sony, DCR-SR300/E, Japan) was fixed above the test tank to record the entire area. The handycam was connected to a computer running the Ethovision Video Tracking System (Noldus Information Technology, The Netherlands) to track movements of the tadpole before and after the addition of the stimulus solution (predator kairomones). The Ethovision system recorded the maximum swimming speed (Vmax), distance traversed by the tadpole, number of swimming spurts and time spent swimming during the entire trial. For each trial, a new test tadpole was introduced into the tank and left undisturbed for 5 min (acclimation). A burette was placed ~1 cm above the water level and 50 mL of aged tap water (chemical blank) was added at the rate of ~1 mL/s to simulate the disturbance created by the subsequent chemical cue. The burette was then gently removed. The movement of the tadpole was recorded for 5 min using Ethovision to determine its baseline activity in the absence of any cues. Following this period, 50 mL of stimulus solution containing predator kairomones was added as described above. Movement of the tadpole was then recorded for another 5 min to determine the activity pattern after exposure to the kairomonal cues.
A total of 25 trials were carried out for each tadpole category (100 trials in total). A new test tadpole was used for each trial. The test tank was cleaned and replenished with aged tap water between each trials.
After checking for normality, the data on the behavioral responses of tadpoles of D. melanostictus before and after the addition of the stimulus solution were compared separately by using the paired-samples t–test. The data were analyzed using a General Linear Model for testing the overall effects of tadpole tested category, treatment type (exposure to chemical blank water or stimulus solution), and their interactions. The response variables were the different swimming activities (i.e., maximum swimming speed, frequency of swimming spurts, time spent swimming, and total distance travelled). In addition, the data on the behavioral responses between different tested categories of tadpoles of D. melanostictus to the stimulus solution of a predator were analyzed using a one-way ANOVA followed by a Tukeys’ HSD post-hoc test. All these tests were performed using SPSS ver. 22.0.
Behavioral responses of D. melanostictus tadpoles to kairomones of H. tigerinus tadpoles
Upon exposure to the predator kairomones, the four tadpoles categories of D. melanostictus showed a significant increase in Vmax and a significant decline in the number of swimming spurts, time spent swimming, and in the total distance moved when compared to their baseline activities in chemical blank water (Table 1).
| Treatment/ Tadpole Category | Maximumc swimming speed (Vmax; cm/s) | Frequency of swimming spurts | Time spent swimming (s) | Distance travelled (cm) |
|---|---|---|---|---|
| Predator-naïve tadpoles (PN) | ||||
| Chemical blank solution | 11.79 ± 0.08 | 65.92 ± 1.40 | 63.08 ± 1.30 | 398.68 ± 8.72 |
| Stimulus solution | 18.49 ± 0.20 | 31.00 ± 0.55 | 29.37 ± 0.54 | 256.39 ± 3.89 |
| t values | t24 = -30.68, | t24 = 20.81, | t24 = 21.06, | t24 = 14.27, |
| p values | P < 0.05 | P < 0.05 | P < 0.05 | P < 0.05 |
| Indirect predator-experienced tadpoles (IpE) | ||||
| Chemical blank solution | 11.83 ± 0.08 | 67.20 ± 1.39 | 65.31 ± 1.31 | 403.76 ± 10.16 |
| Stimulus solution | 19.68 ± 0.32 | 26.60 ± 0.81 | 25.16 ± 0.83 | 229.02 ± 6.33 |
| t values | t24 = -22.21, | t24 = 26.89, | t24 = 28.05, | t24 = 14.18, |
| p values | P < 0.05 | P < 0.05 | P < 0.05 | P < 0.05 |
| Direct predator-experienced tadpoles (DpE) | ||||
| Chemical blank solution | 11.75 ± 0.07 | 65.56 ± 1.47 | 63.53 ± 1.46 | 398.98 ± 8.52 |
| Stimulus solution | 21.15 ± 0.22 | 22.28 ± 0.70 | 20.99 ± 0.67 | 195.91 ± 3.80 |
| t values | t24 = -38.46, | t24 = 27.97, | t24 = 27.71, | t24 = 27.72, |
| p values | P < 0.05 | P < 0.05 | P < 0.05 | P < 0.05 |
| Wild-caught tadpoles (WC) | ||||
| Chemical blank solution | 11.82 ± 0.07 | 67.00 ± 1.36 | 65.45 ± 1.37 | 394.81 ± 6.14 |
| Stimulus solution | 24.53 ± 0.30 | 14.44 ± 0.52 | 13.42 ± 0.47 | 148.11 ± 3.77 |
| t values | t24 = -46.24, | t24 = 39.80, | t24 = 39.66, | t24 = 37.10, |
| p values | P < 0.05 | P < 0.05 | P < 0.05 | P < 0.05 |
Intensity of defense behaviors in D. melanostictus tadpoles to kairomones of H. tigerinus tadpoles
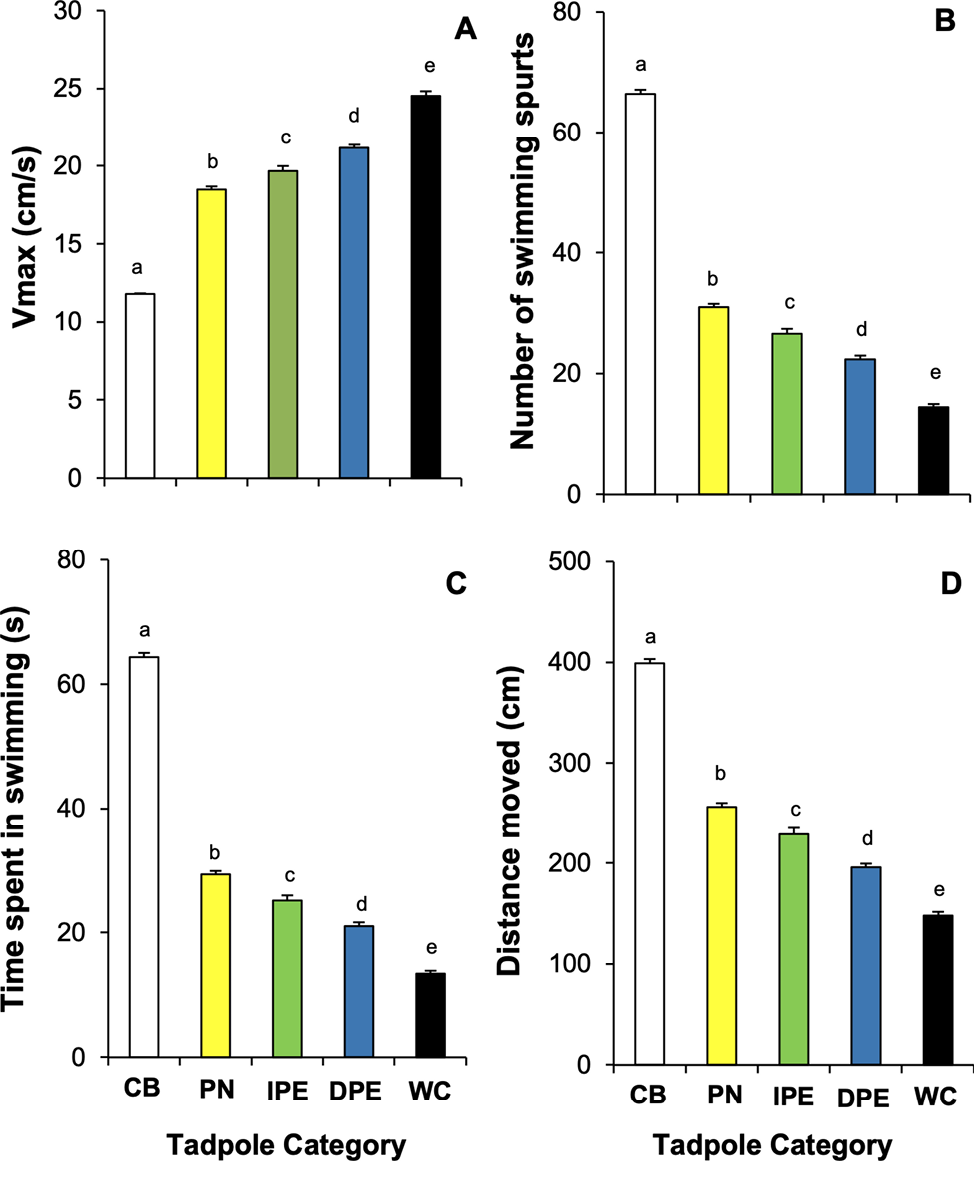
General Linear Model showed that both the tadpole’s experience level (category) and the presence of the cue (treatment), as well as their interaction, had a significant influence on the swimming activities of D. melanostictus tadpoles (Table 2). Tadpoles with more experience with predator cues showed significantly stronger defensive reactions. Results of ANOVA also showed a significant difference in the Vmax (F4,195 = 1206.0, P < 0.05), number of swimming spurts (F4,195 = 792.89, P < 0.05), time spent swimming (F4,195 = 803.69, P < 0.05), and total distance moved (F4,195 = 432.50, P < 0.05) among prey tadpoles in the different tadpole category groups (Figure 1). The intensity of the observed defense behaviors varied significantly with the prey’s level of prior experience with the predator cue (Figure 1). Wild-caught (WC) tadpoles exhibited the strongest response, displaying significantly higher Vmax (P < 0.05), significantly reduced time swimming (P < 0.05), number of swimming spurts (P < 0.05), and moved for a shorter distance (P < 0.05) compared to all other groups (Figure 1). The DpE tadpoles also exhibited significantly higher Vmax (P < 0.05) and spent significantly less time swimming (P < 0.05) with a reduced number of swimming spurts (P < 0.05) and moved only a short distance (P < 0.05) compared to the IpE, predator-naïve, and control groups (Figure 1). The IpE tadpoles also showed significantly higher Vmax (P < 0.05) and reduced swimming metrics (P < 0.05) compared to the predator-naïve and control groups (Figure 1). The predator-naïve tadpoles displayed the lowest-level defense, but still showed significantly higher Vmax (P < 0.05), spent less time in swimming (P < 0.05) with a reduced number of swimming spurts (P < 0.05), and moved only a short distance (P < 0.05) compared to the control group (Figure 1). The hierarchy of intensity of defense behaviors was as follows: wild-caught tadpoles > direct predator-experienced tadpoles > indirect predator-experienced tadpoles > predator-naïve tadpoles > control group.
| Source | Wilks’ Lambda | F | P |
|---|---|---|---|
| Tadpole category | 0.342 | 20.825 | < 0.001* |
| Treatment type | 0.027 | 1676.0 | < 0.001* |
| Tadpole category × treatment type | 0.359 | 19.694 | < 0.001* |
In aquatic environments, the survival of prey like anuran tadpoles is contingent upon effective anti-predator defenses (Schmidt and Amezquita, 2001; Relyea, 2007; Gazzalo et al., 2024; Saidapur, 2025). In such systems, various types of chemical cues (alarm cues of damaged conspecifics, dietary metabolites of predators, disturbance cues, and kairomones of predators) trigger the behavioral responses of prey (Schoeppner and Relyea, 2005, 2009; Saidapur, 2025).
The results of the present study showed that all four categories of prey tadpoles of D. melanostictus regardless of origin, identify the kairomones of the syntopic predator, H. tigerinus as a significant threat and decreased their activity levels during the trial period. When tadpoles of D. melanostictus moved in the stimulus solution (kairomones), their spurt speed (Vmax) was higher than in the stimulus blank solution, indicating their effort to escape from the perceived predator kairomones. This strong, specific response aligns with previous findings on tadpoles of D. melanostictus (Mogali et al., 2011) and Polypedates maculatus (Mogali et al., 2023a). The intensity of this reaction is likely linked to the co-evolutionary history and the hunting strategy of the predator. H. tigerinus tadpoles are active, visually-orienting predators that pose a constant threat (Mogali et al., 2023 a, b). In contrast, tadpoles of Indosylvirana temporalis (Mogali et al., 2012) and D. melanostictus (Mogali et al., 2020) did not alter their behavior in response to kairomones of an insect predator, the larvae of a dragonfly, Pantala flavescens. Larvae of dragonflies are sit-and-wait predators that move slowly and usually wait for prey to come near before attacking (Miller et al., 2014) and may be perceived by the prey as less dangerous and pose a lower predation threat. Sit-and-wait predators, such as dragonfly larvae, exert different selective pressures and may suppress their chemical cues to attract prey (Miller et al., 2015). D. melanostictus exhibited strong antipredator behavioral responses to tadpoles of H. tigerinus because they are active predators that visually locate prey, including syntopic anuran tadpoles (Mogali et al., 2023 a, b; Saidapur, 2025). Thus tadpoles of H. tigerinus pose a serious predation threat to tadpoles of D. melanostictus. The long ecological co-existence of tadpoles of D. melanostictus with sympatric carnivorous tadpoles such as H. tigerinus may have led to the evolution of antipredator defense strategies in response to kairomones of these predators (Mogali et al., 2011, 2023 a, b).
The most significant finding of this study is the clear experience-dependent gradient in the intensity of this defensive behavior. The strong reaction of wild-caught tadpole is consistent with their long-term, cumulative experience in a high-risk natural environment (Mogali et al., 2023 c). Crucially, the significant (though low-level) defensive response of the predator-naïve tadpoles demonstrates that the recognition of H. tigerinus kairomones is fundamentally innate. This provides a vital baseline defense for tadpoles encountering this predator for the first time (Mogali et al., 2011). However, the results show that this innate response is strongly amplified by learning and experience. The IPE group confirms that exposure to the predator’s scent is sufficient to enhance the response, while the DPE group’s stronger reaction suggests that direct, multi-sensory contact with the predator provides an even more powerful reinforcement (Mogali et al., 2012). The hierarchy of antipredator behavioral responses observed in tadpoles of D. melanostictus, wild-caught > direct predator-experienced > indirect predator-experienced > predator-naïve, illustrates this plasticity. This graded response, has also seen in P. maculatus (Mogali et al., 2023 c).
In summary, this study shows that all prey categories, i.e., predator-naïve, direct predator-experienced, indirect predator-experienced and wild-caught tadpoles of D. melanostictus, exhibit a highly plastic anti-predator strategy in response to predator kairomones of H. tigerinus. This behavioral defense is innately recognized, but its intensity is significantly modulated by experience. The observed hierarchy, from a low-level innate response to a maximal response in wild-caught individuals, highlights a mechanism for fine-tuning antipredator behavior based on experience acquired through learning.
I thank my teachers Prof. Bhagyashri A. Shanbhag and Prof. Srinivas K. Saidapur for their support and encouragement to undertake research on behavioral ecology of anuran tadpoles. The study was supported by a grant from the Department of Science and Technology (No. SP-SO/AS-38/2009) and University Grants Commission, New Delhi. All procedures were conducted in accordance with the ethical guidelines of CPCSEA, New Delhi, India (registration no. 639/02/a/CPCSEA).
Amo, L., Lopez, P., Martin, J. (2004): Wall lizards combine chemical and visual cues of ambush snake predators to avoid overestimating risk inside refuges. Anim. Behav. 67: 647-653.
Batabyal, A., Gosavi, S.M., Gramapurohit, N.P. (2014): Determining sensitive stages for learning to detect predators in larval bronzed frogs: Importance of alarm cues in learning. J. Biosci. 39: 701-710.
Chivers, D.P., Mirza, R.S. (2001): Importance of predator diet cues in responses of larval wood frogs to fish and invertebrate predators. J. Chem. Ecol. 27: 45-51.
Chivers, D.P., Brown, G.E., Smith, R.J.F. (1996): The evolution of chemical alarm signals: Attracting predators benefits alarm signal senders. Am. Nat. 148: 649-659.
Crowl, T.A., Covich, A.P. (1990): Predator-induced life-history shifts in a freshwater snail. Science 247: 949-951.
Ferrari, M.C.O., Wisenden, B.D., Chivers, D.P. (2010): Chemical ecology of predator-prey interactions in aquatic ecosystems: A review and prospectus. Can. J. Zool. 88: 698-724.
Gazzola A., Ratto, D., Perrucci, F., Occhinegro, A., Leone, R., Giammello, F., Balestrieri, A., Rosa, D., Rossi, P., Brandalise, F. (2024): Predation cues induce predator specific changes in olfactory neurons encoding defensive responses in agile frog tadpoles. PLOS One 19: e0302728.
Gosner, K.L. (1960): A simplified table for staging anuran embryos and larvae with notes on identification. Herpetologica 16: 183-190.
Gramapurohit, N.P., Radder, R.S. (2012): Mating pattern, spawning behavior, and sexual size dimorphism in the tropical toad Bufo melanostictus (Schn,). J. Herpetol. 46: 412-416.
Gyssels, F., Stoks, R. (2006): Behavioural responses to fish kairomones and autotomy in a damselfly. J. Ethol. 24: 79-83.
Heyer, W.R., McDiarmid, R.W., Weigmann, D.L. (1975): Tadpoles, predation and pond habitats in the tropics. Biotropica 7: 100-111.
Hossie, T.J., Murray, D.L. (2010): You can’t run but you can hide: Refuge use in frog tadpoles elicits density-dependent predation by dragonfly larvae. Oecologia 163: 395-404.
Kats, L.B., Dill, L.M. (1998): The scent of death: Chemosensory assessment of predation risk by prey animals. Ecoscience 5: 361-394.
Laforsch, C., Tollrian, R. (2004): Inducible defenses in multimodal environments: Cyclomorphosis in Daphnia cucullata. Ecology 85: 2302-2311.
Landeira-Dabarca, A., Näslund, J., Johnsson, J.I., Álvarez, M. (2019): Cue recognition and behavioural responses in the three-spined stickleback (Gasterosteus aculeatus) under risk of fish predation. Acta Ethol. 22: 209-221.
Laurila, A., Kujasalo, J., Ranta, E. (1997): Different antipredatory behaviour in two anuran tadpoles: Effects of predator diet. Behav. Ecol. Sociobiol. 40: 329-336.
Lima, S.L., Dill, L.M. (1990): Behavioral decisions made under the risk of predation: A review and prospectus. Can. J. Zool. 68: 619-640.
Mathis, A. (2003): Use of chemical cues in detection of conspecific predators and prey by newts, Notophthamus viridescens. Chemoecology 13: 193-197.
Mathis, A., Vincent, F. (2000): Differential use of visual and chemical cues in predator recognition and threat-sensitive predator avoidance responses by larval newts (Notophthalmus viridescens). Can. J. Zool. 78: 1646-1652.
McCormick, M.I., Manassa, R. (2007): Predation risk assessment by olfactory and visual cues in a coral reef fish. Coral Reefs 27: 105-113.
Miller, J.R., Ament, B.J.M., Schmitz, O.J. (2014): Fear on the move: Predator hunting mode predicts variation in prey mortality and plasticity in prey spatial response. J. Anim. Ecol. 83: 214-222.
Miller, A.K., Martiz, B., Mckay, S., Glaudas, X., Alexander, G.J. (2015): An ambusher’s arsenal: Chemical crypsis in the puff adder (Bitis arietans). Proc. R. Soc. B. 282: 20152182.
Mogali, S.M., Saidapur, S.K., Shanbhag, B.A. (2011): Levels of predation modulate antipredator defense behavior and metamorphic traits in the toad Bufo melanostictus. J. Herpetol. 45: 428-431.
Mogali, S.M., Saidapur, S.K., Shanbhag, B.A. (2012): Tadpoles of the bronze frog (Rana temporalis) assess predation risk before evoking antipredator defense behavior. J. Ethol. 30: 379-386.
Mogali, S.M., Saidapur, S.K., Shanbhag, B.A. (2020): Behavioral responses of tadpoles of Duttaphrynus melanostictus (Anura: Bufonidae) to cues of starved and fed dragonfly larvae. Phyllomedusa 19: 93-98.
Mogali, S.M., Shanbhag, B.A., Saidapur, S.K. (2021): Comparative vulnerability of Indosylvirana temporalis and Clinotarsus curtipes (Anura: Ranidae) tadpoles to water scorpions: Importance of refugia and swimming speed in predator avoidance. Phyllomedusa 20: 159-164.
Mogali, S.M., Shanbhag, B.A., Saidapur, S.K. (2023 a): Antipredator responses of Polypedates maculatus tadpoles to kairomones from the carnivorous tadpoles of Hoplobatrachus tigerinus. Salamandra 59: 83-86.
Mogali, S.M., Shanbhag, B.A., Saidapur, S.K. (2023 b): Similar antipredator behaviour in tadpoles of two closely associated anuran species Duttaphrynus melanostictus and Sphaerotheca breviceps (Anura) to the common predator Pantala flavescens (Odonata). Salamandra 59: 78-82.
Mogali, S.M., Shanbhag, B.A., Saidapur, S.K. (2023 c): Behavioral responses of laboratory-reared and wild-caught Polypedates maculatus (Anura: Rhacophoridae) tadpoles to dietary cues from the carnivorous tadpoles of Hoplobatrachus tigerinus (Anura: Dicroglossidae). Phyllomedusa 22: 3-10.
Petranka, J., Hayes, L. (1998): Chemically mediated avoidance of a predatory odonate (Anax junius) by American toad (Bufo americanus) and wood frog (Rana sylvatica) tadpoles. Behav. Ecol. Sociobiol. 42: 263-271.
Relyea, R.A. (2007): Getting out alive: How predators affect the decision to metamorphose. Oecologia 152: 389-400.
Saidapur, S.K. (2001): Behavioral ecology of anuran tadpoles: Indian scenario. Proc. Indian Nat. Sci. Acad. 67: 311-322.
Saidapur, S.K. (2025): Behavioral ecology and developmental plasticity in Indian anuran tadpoles. Behav. Ecol. Sociobiol. 79: 46.
Scherer, A.E., Smee, D.L. (2016): A review of predator diet effects on prey defensive responses. Chemoecology 26: 83-100.
Schmidt, B.R., Amezquita, A. (2001): Predator-induced behavioral responses: Tadpoles of the neotropical frog Phyllomedusa tarsius do not respond to all predators. Herpetol. J. 11: 9-15.
Schoeppner, N.M., Relyea, R.A. (2005): Damage, digestion, and defense: The role of alarm cues and kairomones for inducing prey defenses. Ecol. Lett. 8: 505-512.
Schoeppner, N.M., Relyea, R.A. (2009): When should prey respond to consumed heterospecifics? Testing hypotheses of perceived risk. Copeia 2009: 190-194.
Semilitsch, R.D., Reyer, H.O. (1992): Modifications of antipredator defenses in tadpoles by environmental conditioning. J. Anim. Ecol. 61: 353-360.
Smith, G.R., Boyd, A., Dayer, C.B., Ogle, M.E., Terlecky, A.J., Dibble, C.J. (2010): Effects of sibship and the presence of multiple predators on the behavior of green frog (Rana clamitans) tadpoles. Ethology 116: 213-217.
Spieler, M., Linsenmair, K.E. (1999): Aggregation behaviour of Bufo maculates tadpoles as an antipredator mechanism. Ethology 105: 665-686.
Stirling, G. (1995): Daphnia behaviour as a bioassay of fish presence or predation. Funct. Ecol. 9: 778-784.
Van Buskirk, J., Arioli, M. (2002): Dosage response of an induced defense: How sensitive are tadpoles to predation risk? Ecology 83: 1580-1585.